Chapter: Clinical Anesthesiology: Anesthetic Management: Renal Physiology & Anesthesia
Anesthesia: Loop Diuretics
LOOP DIURETICS
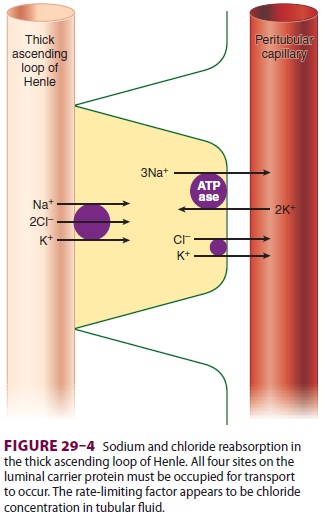
The loop diuretics include furosemide (Lasix), bumetanide (Bumex), ethacrynic acid (Edecrin), and torsemide (Demadex). All loop diuretics inhibit Na+ and Cl − reabsorption in the thick ascending limb. Sodium reabsorption at that site requires that all four sites on the Na +–K+–2Cl− luminal car-rier protein be occupied. Loop diuretics compete with Cl− for its binding site on the carrier protein (see Figure 29–4). With a maximal effect, they can promote excretion of 15–20% of the filtered sodium load. Both urinary concentrating and urinary dilut-ing capacities are impaired. The large amounts of Na+ and Cl − presented to the distal nephron over-whelm its limited reabsorptive capability. The result-ing urine remains hypotonic, probably due to rapid urinary flow rates that prevent equilibration with the hypertonic renal medulla or due to interference with the action of ADH on the collecting tubules. A marked increase in diuresis may occur when a loop diuretic is combined with a thiazide diuretic, espe-cially metolazone.
Loop diuretics also increase urinary calcium and magnesium excretion. Ethacrynic acid is the only loop diuretic that is not a sulfonamide deriv-ative, and thus may be the diuretic of choice in patients allergic to sulfonamide drugs. Torsemide may have an antihypertensive action independent of its diuretic effect.
Uses
A. Edematous States (Sodium Overload)
These disorders include heart failure, cirrhosis, the nephrotic syndrome, and renal insufficiency. When given intravenously, these agents can rapidly reverse cardiac and pulmonary manifestations of fluid overload.
B. Hypertension
Loop diuretics may be used as adjuncts to other hypotensive agents, particularly when thiazides (below) alone are ineffective.
C. Evaluation of Acute Oliguria
The response to a small dose (10–20 mg) of furo-semide may be useful in differentiating between oliguria resulting from hypovolemia and oliguria resulting from redistribution of RBF to juxtamed-ullary nephrons. Little or no response is seen with hypovolemia, whereas resumption of normal uri-nary output occurs with the latter. However, the optimal initial approach to evaluation of acute oli-guria is to correct hypovolemia and optimize cardiac output and renal perfusion.
D. Conversion of Oliguric Kidney Failure to Nonoliguric Failure
As with mannitol, discussed earlier, many clinicians continue to administer loop diuretics for renal pro-tectn and to convert oliguric acute kidney failure to nonoliguric kidney failure, despite lack of evi-dence that such use provides renal protection, less-ens the severity of AKI, or lessens the morbidity or mortality associated with AKI, when compared with correction of hypovolemia and preservation of ade-quate renal perfusion alone.
E. Treatment of Hypercalcemia
F. Rapid Correction of Hyponatremia
Intravenous Dosages
The intravenous doses are furosemide, 10–100 mg; bumetanide, 0.5–1 mg; ethacrynic acid, 50–100 mg; and torsemide 10–100 mg.
Side Effects
Increased delivery of Na+ to the distal and collect-ing tubules increases K+ and H+ secretion at those sites and can result in hypokalemia and metabolic alkalosis. Marked Na+ losses will also lead to hypo-volemia and prerenal azotemia; secondary hyper-aldosteronism often accentuates the hypokalemia and metabolic alkalosis. Urinary calcium and magnesium loss promoted by loop diuretics may result in hypocalcemia or hypomagnesemia, or both. Hypercalciuria can result in stone formation. Hyperuricemia may result from increased urate reabsorption and from competitive inhibition of urate secretion in the proximal tubule. Reversible and irreversible hearing loss has been reported with loop diuretics, especially furosemide and ethacrynic acid.
Related Topics