Chapter: Clinical Anesthesiology: Anesthetic Management: Anesthesia for Patients with Respiratory Disease
Anesthesia : Chronic Obstructive Pulmonary Disease
CHRONIC OBSTRUCTIVE PULMONARY DISEASE
Preoperative Considerations
COPD is the most common pulmonary
disorder encountered in anesthetic practice, and its preva-lence increases with
age. The disorder is strongly associated with cigarette smoking and has a male
predominance. COPD is currently defined as a disease state characterized by
airflow limitation that is not fully reversible. The chronic airflow limitation
of this disease is due to a mixture of small and large airway disease (chronic
bronchitis/bron-chiolitis) and parenchymal destruction (emphy-sema), with
representation of these two components varying from patient to patient.Most
patients with COPD are asymptomatic or only mildly symptomatic, but show
expiratory air-flow obstruction upon PFTs. In many patients, the obstruction
has an element of reversibility, presum-ably from bronchospasm (as shown by
improvement in response to administration of a bronchodilator). With advancing
disease, maldistribution of both ventilation and· pulmonary· blood flow results
in areas of low (V/Q) ratios· (intrapulmonary· shunt), as well as areas of high
(V/Q) ratios (dead space).
A. Chronic Bronchitis
The clinical diagnosis of chronic
bronchitis is defined by the presence of a productive cough on most days of 3
consecutive months for at least 2 consecutive years. In addition to cigarette
smok-ing, air pollutants, occupational exposure to dusts, recurrent pulmonary
infections, and familial fac-tors may be responsible. Secretions from
hyper-trophied bronchial mucous glands and mucosal edema from inflammation of
the airways produce airflow obstruction. The term “chronic asthmatic
bronchitis” may be used when
bronchospasm is a major feature. Recurrent pulmonary infections (viral and
bacterial) are common and often associ-ated with bronchospasm. RV is increased,
but TLC is often normal. Intrapulmonary shunting is promi-nent, and hypoxemia
is common.
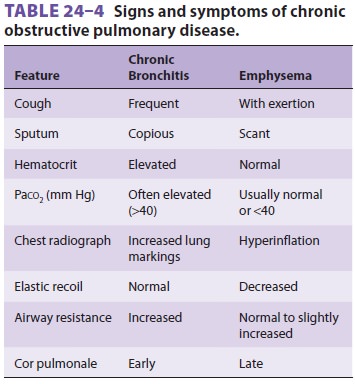
In patients with COPD, chronic hypoxemia
leads to erythrocytosis, pulmonary hypertension, and eventually right
ventricular failure (cor pulmo-nale); this combination of findings is often
referred to as the blue bloater syndrome, but <5% of patients with COPD fit this
description (Table24–4).
In the course of disease progression, patients gradually develop chronic CO2 retention; the normal venti-latory drive becomes
less sensitive to arterial CO2 tension and may be depressed by oxygen
adminis-tration (below).
B. Emphysema
Emphysema is a pathological disorder
character-ized by irreversible enlargement of the airways distal to terminal
bronchioles and destruction of alveo-lar septa. The diagnosis can be reliably
made with computed tomography (CT) of the chest. Mild api-cal emphysematous
changes are a normal, but clini-cally insignificant, consequence of aging.
Significant emphysema is more frequently related to cigarette smoking. Less
commonly, emphysema occurs at an early age and is associated with a homozygous
deficiency of α1-antitrypsin.
This is a protease inhibitor that prevents excessive activity of proteo-lytic
enzymes (mainly elastase) in the lungs; these enzymes are produced by pulmonary
neutrophils and macrophages in response to infection and pol-lutants. Emphysema
associated with smoking may similarly be due to a relative imbalance between
protease and antiprotease activities in susceptible individuals.
Emphysema may exist in a centrilobular
or panlobular form. The centrilobular (or centriaci-nar) form results from
dilatation or destruction of the respiratory bronchioles, is more closely
associ-ated with tobacco smoking, and has predominantly an upper lobe
distribution. The panlobular (or pan-acinar) form results in a more even
dilatation and destruction of the entire acinus, is associated with α1-antitrypsin deficiency, and has
predominantly alower lobe distribution.
Loss of the elastic recoil that normally
supports small airways by radial traction allows premature collapse during
exhalation, leading to expiratory flow limitation with air trapping and
hyperinfla-tion. Patients characteristically have increases in RV, FRC, TLC,
and the RV/TLC ratio. The FRC is shifted rightward along the compliance curve
of the lungs, toward the flat portion of the curve, in detriment of the
pulmonary mechanics.
Disruption of the alveolar–capillary
structure and loss of the acinar structure leads to decreased diffusion lung
capacity (DLCO), V/Q mismatch, and impairment of gas exchange. Also, normal
parenchyma may become compressed by the hyperinflated portions ·of· the lung,
resulting in a further increase in the V/Q mismatch. Due to the higher
diffusibility of CO2its elimination is well preserveduntilV/Qabnormalities
become severe. ChronicCO2 retention occurs slowly and generally
results in a compensated respiratory acidosis on blood gas analysis. Arterial
oxygen tension is usually normal or slightly reduced. Acute CO2 retention is a sign of impending respiratory
failure.
Destruction of pulmonary capillaries in
the alveolar septa leads to the development of pul-monary hypertension.
However, the degree of pulmonary hypertension is usually low to moderate,
rarely exceeding 35-40 mm Hg.
When dyspneic, patients with emphysema
often purse their lips to delay closure of the small airways, which accounts
for the term “pink puffers” that is often used (Table 24–4). However, as
mentioned above, most patients diagnosed with COPD have a combination of
bronchitis and emphysema.
C. Treatment
TreatmentforCOPDisprimarilysupportive.Cessation
of smoking is the long-term inter-vention that has been shown to reduce the
rateof decline in lung function. Patients demonstrating a reversible element in
airway obstruction (>15% improvement in FEV1
following administration of a bronchodilator) should be started on long-term
bronchodilator therapy. Inhaled β2-adrenergic ago-nists, glucocorticoids, and
ipratropium are very use-ful; ipratropium may play a more important role in the
management of these patients than in patients with asthma. Even patients who do
not show improvement in their PFTs from the use of broncho-dilators may improve
clinically with bronchodilator therapy. Treatment with systemic corticosteroids
may be required in patients with acute exacerbations of COPD. However, systemic
corticosteroids in patients with stable COPD is discouraged due to the lack of
added benefit and the potential for systemic side effects. COPD exacerbations
may be related to bouts of bronchitis, heralded by a change in sputum; frequent
treatment with broad-spectrum antibiotics may be necessary. Hypoxemia should be
treated carefully with supplemental oxygen. Patients with chronic hypoxemia
(Pao2<55
mm Hg) and pulmo-nary hypertension require low-flow oxygen therapy (1–2 L/min).
Oxygen treatment during acute COPD exacerbations to a Pao2 above 60 mm Hg may lead toCO retention, most likely
due to an inhibition of the hypoxic vasoconstriction in areas with low V/Q
andthe Haldane effect.
When cor pulmonale is present, diuretics
are used to control peripheral edema; beneficial effects from vasodilators are
inconsistent. Pulmonary reha-bilitation may improve the functional status of
the patient by improving physical symptoms and exer-cise capacity. Some studies
suggest that the ability to increase oxygen consumption during exercise is
inversely related to postoperative complications.
Anesthetic Considerations
A. Preoperative Management
Patients
with COPD should be prepared prior to elective surgical procedures in the same
way as patients with asthma (above). They should be ques-tioned about recent
changes in dyspnea, sputum, and wheezing. Patients with an FEV 1
less than 50% of predicted (1.2–1.5 L) usually have dyspnea on exertion,
whereas those with an FEV 1 less than 25% (<1
L in men) typically have dyspnea with minimal activity. The latter finding, in
patients with predomi-nantly chronic bronchitis, is also often associated with
CO2 retention and pulmonary hypertension. PFTs, chest radiographs,
and arterial blood gas mea-surements, if available, should be reviewed
carefully. The presence of bullous changes on the radiograph should be noted.
Many patients have concomitant cardiac disease and should also receive a
careful car-diovascular evaluation.
In
contrast to asthma, only limited improve-ment in respiratory function may be
seen after a short period of intensive preoperative preparation.Nonetheless,
preoperative interventions in patients with COPD aimed at correctinghypoxemia,
relieving bronchospasm, mobilizing and reducing secretions, and treating
infections may decrease the incidence of postoperative pulmonary complications.
Patients at greatest risk of complica-tions are those with preoperative
pulmonary func-tion measurements less than 50% of predicted. The possibility
that postoperative ventilation may be necessary in high-risk patients should be
discussed with both the patient and the surgeon.
Smoking
should be discontinued for at least 6–8 weeks before the operation to decrease
secre-tions and to reduce pulmonary complications.Cigarette smoking increases mucus
production and decreases clearance. Both gaseous and particulate phases of
cigarette smoke can deplete glutathione and vitamin C and may promote oxidative
injury to tissues. Cessation of smoking for as little as 24 hr has theoretical
beneficial effects on the oxygen-carrying capacity of hemoglobin; acute
inhalation of cigarette smoke releases carbon monoxide, which increases
carboxyhemoglobin levels, as well as nitric oxide, and nitrogen dioxide, which
can lead to formation of methemoglobin.
Long-acting
bronchodilators and mucolytics should be continued, including on the day of
surgery. COPD exacerbations should be treated aggressively.
Preoperative
chest physiotherapy and lung expansion interventions with incentive spirometry,
deep breathing exercises, cough, chest percussion, and postural drainage may be
beneficial in decreas-ing postoperative pulmonary complications.
B. Intraoperative Management
Although
regional anesthesia is often considered preferable to general anesthesia, high
spinal or epi-dural anesthesia can decrease lung volumes, restrict the use of
accessory respiratory muscles, and pro-duce an ineffective cough, leading to
dyspnea and retention of secretions. Loss of proprioception from the chest and
positions such as lithotomy or lateral decubitus may accentuate dyspnea in
awake patients.
Concerns
about diaphragmatic paralysis may make interscalene blocks a less attractive
option in the lung disease patient.
Preoxygenation
prior to induction of general anesthesia prevents the rapid oxygen desaturation
often seen in these patients. The selection of anes-thetic agents and general
intraoperative manage-ment must be tailored to the specific needs and goals of
every patient. Unfortunately, the use of broncho-dilating anesthetics improves
only the reversible component of airflow obstruction; significant expi-ratory obstruction
may still present, even under deep anesthesia. Expiratory airflow limitation,
espe-cially under positive pressure ventilation, may lead to air trapping,
dynamic hyperinflation, and elevated intrinsic positive end-expiratory pressure
(iPEEP). Dynamic hyperinflation may result in volutrauma to the lungs,
hemodynamic instability, hypercap-nia, and acidosis. Interventions to mitigate
air trap-ping include: (1) allowing more time to exhale by decreasing both the
respiratory rate and I:E ratio;allowing permissive hypercapnia; (3) applying
low levels of extrinsic PEEP; and (4) aggressively treating bronchospasm.
Intraoperative
causes of hypotension include pneumothorax, and right heart failure due to
hypercapnia and acidosis. A pneumothorax may manifest as hypoxemia, increased
peak airway pressures, decreasing tidal volumes, and abrupt cardiovascular
collapse unresponsive to fluid and vasopressor administration.
Nitrous
oxide should be avoided in patients with bullae and pulmonary hypertension.
Inhibition of hypoxic pulmonary vasoconstriction by inhala-tion anesthetics is
usually not clinically significant at the usual doses. However, due to
increased dead space, patients with severe COPD have unpredict-able uptake and
distribution of inhalational agents, and the end-tidal volatile anesthetic
concentration is inaccurate.
Measurement
of arterial blood gases is desir-able for extensive intraabdominal and thoracic
procedures. Although pulse oximetry accurately detects significant arterial
desaturation, direct measurement of arterial oxygen tensions may be necessary
to detect more subtle changes in intrapul-monary shunting. Moreover, arterial
CO 2 measure-ments should be used to guide ventilation because
increased dead space widens the normal arterial-to-end-tidal CO2
gradient. Moderate hypercapnia with a Paco2 of up to 70 mm Hg may be
well tolerated in the short term, assuming a reasonable cardiovas-cular
reserve. Hemodynamic support with inotro-pic agents may be required in more
compromised patients. Hemodynamic monitoring should be dic-tated by any
underlying cardiac dysfunction, as well as the extent of the surgery. In
patients with pulmo-nary hypertension, measurements of central venous pressure
reflect right ventricular function rather than intravascular volume.
At
the end of surgery, the timing of extubation should balance the risk of
bronchospasm with that of respiratory failure, but evidence suggests that early
extubation (in the operating room) is beneficial. Successful extubation at the
end of the procedure depends on multiple factors: adequate pain control,
reversal of neuromuscular blockade, absence of sig-nificant bronchospasm and
secretions, absence of significant hypercapnia and acidosis, and absence of
respiratory depression due to residual anesthetic agents. Patients with an FEV 1
below 50% may require a period of postoperative ventilation, particularly
following upper abdominal and thoracic operations.
Related Topics