Chapter: Environmental Engineering : Water Treatment
Water Tretment: Hot Process Softening
WATER TRETMENT: HOT PROCESS SOFTENING
Hot
process softening is usually carried out under pressure at temperatures of
227-240 o F (108-116 o C). At the operating temperature, hot process softening
reactions go essentially to completion. This treatment method involves the same
reactions described above, except that raw water CO2 is vented and
does not participate in the lime reaction. The use of lime and soda ash permits
hardness reduction down to 0.5 gr/gal, or about 8 ppm, as calcium carbonate.
Magnesium
is reduced to 2-5 ppm because of the lower solubility of magnesium hydroxide at
the elevated temperatures.
Silica Reduction
Hot
process softening can also provide very good silica reduction. The silica
reduction is accomplished through adsorption of the silica on the magnesium
hydroxide precipitate. If there is insufficient magnesium present in the raw
water to reduce silica to the desired level, magnesium compounds (such as
magnesium oxide, magnesium sulfate, magnesium carbonate, or dolomitic lime) may
be used. Figure 7-3 is a plot of magnesium oxide vs. raw water silica (in ppm),
which may be used to estimate the quantity of magnesium oxide required to
reduce silica to the levels indicated . Magnesium oxide is the preferred
chemical because it does not increase the dissolved solids concentration of the
water.
Good
sludge contact enhances silica reduction. To ensure optimum contact, sludge is
frequently recirculated back to the inlet of the unit.
Cold
or warm process softening is not as effective as hot process softening for
silica reduction. However, added magnesium oxide and good sludge contact will
improve results.
Predicted
analyses of a typical raw water treated by various lime and lime-soda softening
processes are presented in Table 7-1.
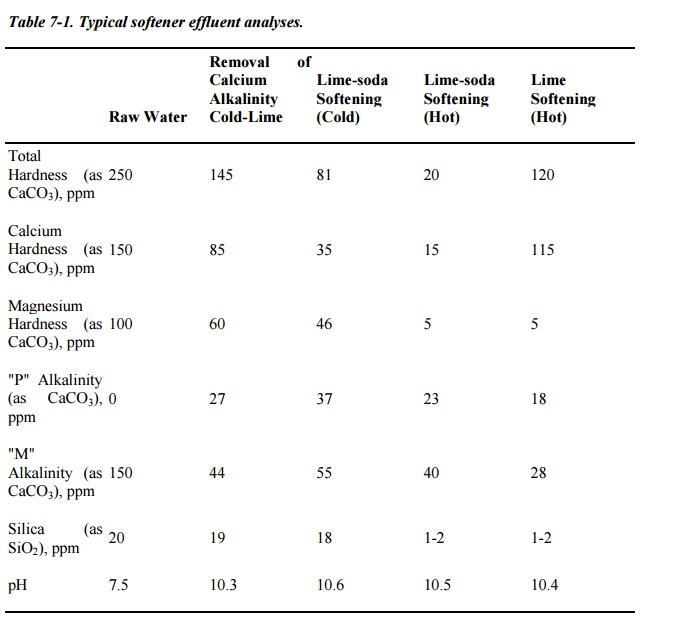
Table 7-1.
Typical softener effluent analyses.
Removal of
Calcium Lime-soda Lime-soda Lime
Alkalinity Softening Softening Softening
Raw Water Cold-Lime (Cold) (Hot) (Hot)
Total
Hardness (as 250 145 81 20 120
CaCO3), ppm
Calcium
Hardness (as 150 85 35 15 115
CaCO3), ppm
Magnesium
Hardness (as 100 60 46 5 5
CaCO3), ppm
"P" Alkalinity
(as CaCO3), 0 27 37 23 18
ppm
"M"
Alkalinity (as 150 44 55 40 28
CaCO3), ppm
Silica (as 20 19 18 1-2 1-2
SiO2), ppm
pH 7.5 10.3 10.6 10.5 10.4
Reduction of Other
Contaminants
Treatment
by lime precipitation reduces alkalinity. However, if the raw water alkalinity
exceeds the total hardness, sodium bicarbonate alkalinity is present. In such
cases, it is usually necessary to reduce treated water alkalinity in order to
reduce condensate system corrosion or permit increased cycles of concentration.
Treatment
by lime converts the sodium bicarbonate in the raw water to sodium carbonate as
follows:
2NaHCO3
+ Ca(OH)2= CaCO3 Ż
+ Na2CO3+ 2H2O
Sodium
bicarbonate + calcium hydroxide = calcium carbonate + sodium carbonate + water
Calcium sulfate
(gypsum) may be added to reduce the carbonate to required levels. The reaction
is as follows:
Na2CO3
+ CaSO4= CaCO3 Ż
+ Na2SO4
sodium
carbonate + calcium sulfate = calcium carbonate + sodiumsulfate
This
is the same reaction involved in the reduction of noncarbonate calcium hardness
previously discussed.
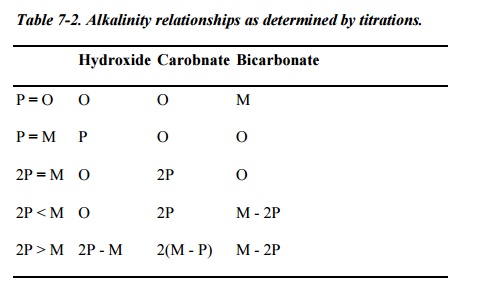
Table
7-2. Alkalinity relationships as determined by titrations.
Hydroxide Carobnate Bicarbonate
P = O O O M
P = M P O O
2P = M O 2P O
2P < M O 2P M - 2P
2P > M 2P - M 2(M - P)M - 2P
Reduction of Other
Contaminants
Lime
softening processes, with the usual filters, will reduce oxidized iron and
manganese to about 0.05 and 0.01 ppm, respectively. Raw water organics
(color-contributing colloids) are also reduced.
Turbidity,
present in most surface supplies, is reduced to about 1.0 NTU with filtration
following chemical treatment. Raw water turbidity in excess of 100 NTU may be
tolerated in these systems; however, it may be necessary to coagulate raw water
solids with a cationic polymer before the water enters the softener vessel to
assist liquid-solids separation.
Oil
may also be removed by adsorption on the precipitates formed during treatment.
However, oil in concentrations above about 30 ppm should be reduced before lime
treatment because higher concentrations of oil may exert a dispersing influence
and cause floc carryover.
Precipitation Process
(Chemical) Control
Lime or lime-soda
softener control is usually based on treated water alkalinity and hardness.
Samples are tested to determine the alkalinity to the P (phenolphthalein, pH
8.3) and M (methyl orange or methyl purple, pH 4.3) end points. The following
relationships apply:
P (ppm as CaCO3)
= OH-( Hydroxyl) + ˝ CO 3
2-(carbonate)
M (ppm CaCO3)=
OH- (Hydroxyl)+ CO3 2- (carbonate)+ HCO3(bicarbonate)
In
the presence of hydroxyl ion (OH-), bicarbonate concentration is so
low that it may be assumed to be zero.
In the precipitation
process, it is advisable to ensure that all of the bicarbonate has been
converted to carbonate (the least soluble form of the calcium); therefore, a
slight excess of hydroxyl ion should be maintained in the treated water. When
the equations above are combined, it can be shown that when 2P - M is positive,
hydroxyl ion is present. The usual control range is:
2P - M = 5-15
ppm
This corresponds to a
pH of approximately 10.2.
If
soda ash is also used, the control is on the excess carbonate ion. As shown in
Figure 7-1 (above), excess carbonate will depress the calcium to the level
desired. The usual control range for hot lime-soda units is:
M
(alkalinity) - TH (total hardness) = 20-40 ppm
For
cold lime-soda softening, where effluent magnesium hardness is significantly
greater than in hot lime or soda, the control range above may be inappropriate.
For cold lime-soda units, soda ash can be controlled such that:
2(M
- P) - Calcium hardness = 20-40 ppm
Care
must be exercised in the specification of soda ash control ranges. If the
softened water is to be used as boiler feedwater, hardness removal by the
addition of soda ash may not be worth the cost of the resulting increase in
steam condensate system corrosion. This corrosion is caused by the higher
levels of carbon dioxide in the steam resulting from the higher carbonate
alkalinity of the feedwater.
Coagulants/Flocculants/Sludge
Conditioners
Organic
polymer flocculants and coagulants are preferred over inorganic salts of
aluminum or iron. Polymers add minimal dissolved solids to the water and their
use results in reduced sludge quantity compared to the use of inorganic
coagulants. Inorganic coagulants must react with raw water alkalinity to form
the metallic precipitate that aids in clarification and sludge bed conditioning.
For example, alum reacts as follows:
3Ca(HCO3)2
+ Al2(SO4)3= 3CaSO4+2Al(OH)3
Ż+6CO2
calcium
bicarbonate + aluminum sulfate = calciumsulfate
+ aluminum hydroxide + carbon
dioxide
The
precipitated aluminum hydroxide is incorporated within the sludge produced by
the softening reactions. This increases the fluidity of the softener sludge,
which allows for increased solids contact, improving softening and effluent
clarity.
Waters
producing high calcium-to-magnesium precipitation ratios usually need sludge
bed conditioning chemical feed for proper operation. Specialized organic
polymers are available for proper conditioning of the sludge bed without the
use of inorganic salts.
Four potentially
adverse effects of using inorganic salts may be noted:
·
The inorganic salt reduces the
alkalinity. This converts the hardness to
noncarbonate hardness, which is not affected by lime. As a result,
inorganic salts increase hardness in water that is naturally deficient in
bicarbonate alkalinity.
·
When the water is to be treated
further by ion exchange, regenerant consumption is increased. This
is due to the higher hardness and the added soluble sulfate/chloride load.
·
The carbon dioxide generated by the
reaction has a lime demand which is twice that of the bicarbonate. Therefore,
increased chemical addition is required.
·
soluble aluminum in the softener
effluent interferes with softened water alkalinity titrations, even when very
low levels of soluble aluminum exist. This interference, which necessitates
an increase in lime feed, causes falsely low (2P - M) readings and may be
partly responsible for the additional removal of magnesium seen when aluminum
salts are used.
H+ + CO32- = HCO3-
hydrogen ion + carbonateion
= bicarbonate ion
A typical cold lime
softener system is shown in Figure 7-6 .
Hot
Process
Two hot process softener designs are illustrated in
Figures 7-7 and 7-8 . The former, the simplest in design and fabrication, is
referred to as a "downflow" unit. The latter, which incorporates
additional features, is referred to as an "upflow" unit. Many
variations in design of both units exist, but the principle of operation is
quite similar.
In each unit, water is admitted to the top of the
vessel designed to operate at 5-15 psig saturated steam pressure (227-240 o F,
108-116 o C). An inlet valve is used to control the inlet water flow as a
function of the operating level of the vessel. The water is sprayed into the
steam space of the unit and is heated to within 2 or 3 degrees of the
saturation temperature of the steam. Heating reduces the noncondensible gas
content of the water. Oxygen and carbon dioxide are released and vented to the
atmosphere with a controlled loss of heating steam. Although they are not
deaerators, hot process units reduce oxygen to about 0.3 ppm (0.21 cmł/L) and
carbon dioxide to 0.
This residual oxygen level in the high-temperature
water is aggressive and will attack downstream equipment such as filters and
zeolites. Therefore, users should consider feeding a chemical oxygen scavenger
to the effluent of hot process softeners.
Treatment chemicals are introduced into the top of
the vessel as a function of flow and raw water analysis. Although the reactions
go essentially to completion quite rapidly, a minimum of 1 hr of retention is
designed into the unit. Also, flow rate through the unit is limited to 1.7-2.0
gpm/ft˛. Filter backwash water may be withdrawn from the outlet of the unit,
from the filtered water header, or from internal or external storage. Internal
storage compartments are illustrated in Figure 7-8. Filter backwash water is
usually returned to the unit for recovery.
In the downflow design, the water leaves the vessel
after reversing direction and enters the internal hood. Precipitates separate
from the water at the hood and continue downward into the cone for removal by
blowdown. Sludge blowdown is proportioned to raw water flow. For improved
silica reduction, sludge is recirculated from the cone back to the top of the
unit.
For optimum silica reduction, a sludge-contact unit
(shown in Figure 7-8) is used. Water and chemicals enter the top of the unit
and flow to the bottom of the softener through a downcomer. The sludge level is
maintained in such a way that the downcomer always discharges into the sludge
bed. This ensures good contact with the sludge, which is rich in magnesium
hydroxide. Also, the sludge bed acts as a filter, entrapping finer solids before
the water exits near the top of the vessel. Sludge recycle may also be used.
The upflow design also lends itself to easier
incorporation of internal compartments for filter backwash storage and return,
and condensate or treated water deaeration.
Limitations
Given proper consideration of raw water quality and
ultimate end use of the treated water, the application of precipitation
processes has few limitations. However, operational difficulties may be
encountered unless the following factors are controlled:
Temperature.
Cold and warm units are subject to carryover if the temperature varies more than
4 o F/hr (2 o C/hr). Hot process units are less sensitive to slight temperature
variations.
However,
a clogged or improper spray pattern can prevent proper heating of the water,
and carryover can result.
·
Hydraulics.
In any system, steady-state operation within design limi ts optimizes the performance
of the equipment. Rapid flow variations can cause severe system upsets.
Suitable treated water storage capacity should be incorporated into the total
system design to minimize load swings on the softener.
·
Chemical Control.
This should be as precise as possible to prevent poor water quality. Because
of the comparatively constant quality of most well waters, changes in chemical
feed rates are largely a function of flow only. However, surface water quality
may vary hourly. Therefore, for proper control, it is imperative that users
perform frequent testing of the raw water as well a s the treated effluent, and
adjust chemical feed accordingly.
Iron
Exchange
History
![]()
Classifications
of ion exchang e resins
![]()
Sodium zeolite
softening
![]()
Hot zeolite
softening
![]()
Demineralization
![]()
Dealkalization
![]()
Counterflow and
mixed bed deionization
![]()
Other
demineralization processses
![]()
Condensate
polishing
![]()
Common ion
exchange systemm problems
![]()
Resin fouling
and degradation
![]()
Resin testing
and analysis
![]()
All natural waters contain, in various concentrations,
dissolved salts which dissociate in water to form charged ions. Positively c
harged ions are called cations; negatively charg ed ions are called anions.
Ionic impurities can seriously affect the reliability and operating efficie ncy
of a boiler or process system. Overheating caused by the buildup of scale or
deposits formed by these impurities can lead to catastro phic tube failures,
costly production losses, and unscheduled downtime. Hardness ions, such as
calcium and magnesium, must be remove d from the water supply before it can be
used as boiler feedwater. For high-pressure boiler feedw ater systems and many
process systems, nearly c omplete removal of all ions, including carbon dioxide
and silica, is required. Ion exchange system s are used for efficient removal
of dissolved io ns from water.
Ion exchangers exchange
one ion for another, hold it temporarily, and then release it to a regenerant
solution. In an ion exchange system, undesirable ions in the water supply are replaced
with more acceptable ions. For example, in a sodium zeolite softener,
scale-forming calcium and magnesium ions are replaced with sodium ions.
Related Topics