Chemistry, Properties of Water - Water - Biomolecules | 11th Botany : Chapter 8 : Biomolecules
Chapter: 11th Botany : Chapter 8 : Biomolecules
Water - Biomolecules
Water
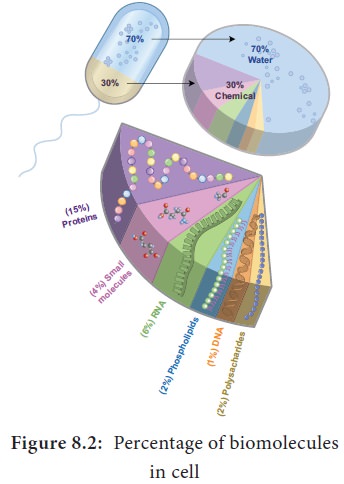
Water is the most abundant component in living
organisms. Life on earth is inevitably linked to water. Water makes up 70% of human cell and upto 95% of mass of a plant cell (Figure
8.2).
1. Chemistry of Water
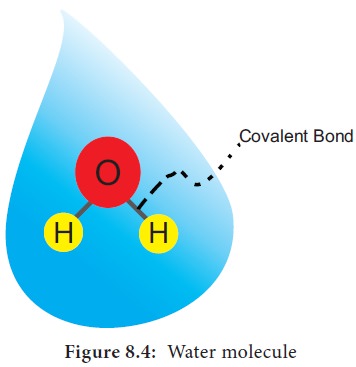
Water is a tiny polar molecule and can readily pass
through membranes. Two electronegative atoms of oxygen share a hydrogen bonds
of two water molecule. Thus, they can stick together by cohesion and results in
lattice formation (Figure 8.4).
2. Properties of Water
•
Adhesion and cohesion property
•
High latent heat of vaporisation
•
High melting and boiling point
•
Universal solvent
•
Specific heat capacity
Related Topics