Properties, Lock and Key Mechanism, Factors Affecting, Nomenclature, Classification, Uses - Enzymes - Biomolecules | 11th Botany : Chapter 8 : Biomolecules
Chapter: 11th Botany : Chapter 8 : Biomolecules
Enzymes - Biomolecules
Enzymes
Enzymes are globular proteins that catalyse the
many thousands of metabolic reactions taking place within cells and organism.
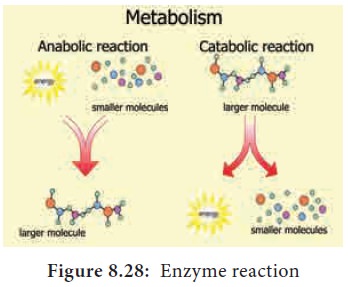
The molecules involved in such reactions are metabolites. Metabolism consists
of chains and cycles of enzyme-catalysed reactions, such as respiration,
photosynthesis, protein synthesis and other pathways. These reactions are
classified as
•
anabolic (building
up of organic molecules). Synthesis
of proteins from amino acids and synthesis of polysaccharides from simple
sugars are examples of anabolic reactions.
•
catabolic
(breaking
down of larger molecules). Digestion
of complex foods and the breaking down of sugar in respiration are examples of
catabolic reactions (Figure 8.28).
Enzymes can be extracellular
enzyme as secreted and work externally exported from cells. Eg. digestive
enzymes; or intracellular enzymes that remain within cells and
work there. These are found inside
organelles or within cells. Eg. insulin
1. Properties of Enzyme
•
All are globular proteins.
•
They act as catalysts and effective even in small
quantity.
•
They remain unchanged at the end of the reaction.
•
They are highly specific.
•
They have an active site where the reaction takes
place.
•
Enzymes lower activation energy of the reaction
they catalyse.
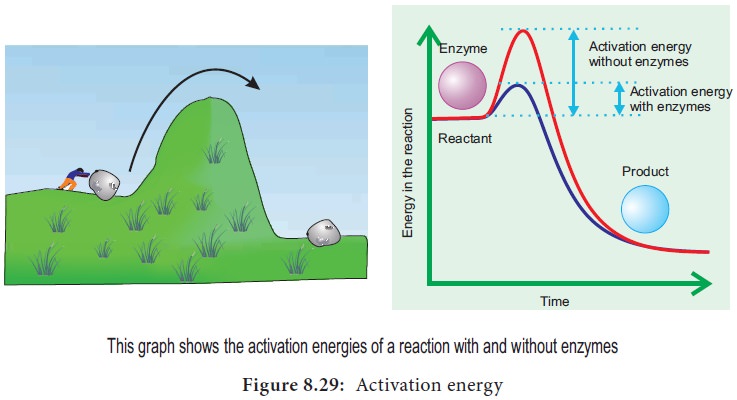
As molecules react they become unstable, high
energy intermediates, but they are in this transition state only momentarily.
Energy is required to raise molecules to this transition state and this minimum
energy needed is called the activation
energy. This could be explained schematically by ‘boulder on hillside’
model of activation energy (Figure 8.29).
2. Lock and Key Mechanism of Enzyme
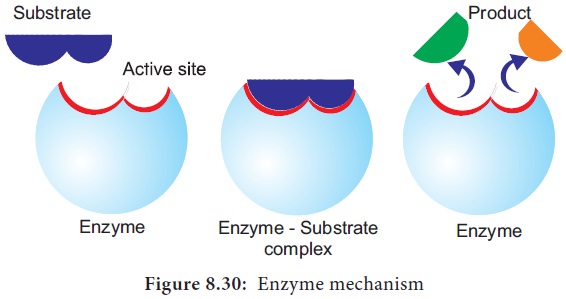
In a enzyme catalysed reaction, the starting
substance is the substrate. It is converted to the product. The substrate binds
to the specially formed pocket in the enzyme – the active site, this is called lock and key mechanism of enzyme action. As the enzyme and substrate form a ES complex, the
substrate is raised in energy to a
transition state and then breaks down into products plus unchanged enzyme
(Figure 8.30).
3. Factors Affecting the Rate of Enzyme Reactions
Enzymes are sensitive to environmental condition.
It could be affected by temperature, pH, substrate concentration and enzyme
concentration.
The rate of enzyme reaction is measured by the
amount of substrate changed or amount of product formed, during a period of
time.
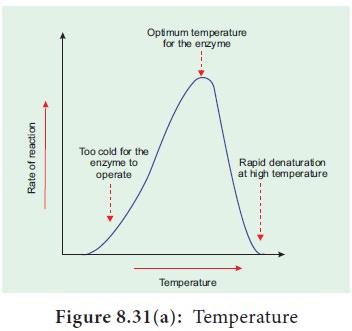
4. Temperature
Heating increases molecular motion. Thus the
molecules of the substrate and enzyme move more quickly resulting in a greater
probability of occurence of the reaction. The temperature that promotes maximum
activity is referred to as optimum temperature (Figure 8.31a).
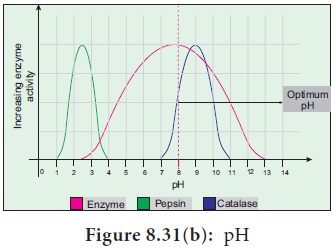
5. pH
The optimum pH is that at which the maximum rate of
reaction occurs. Thus the pH change leads to an alteration of enzyme shape,
including the active site. If extremes of pH are encountered by an enzyme, then
it will be denatured (Figure 8.31b).
6. Substrate Concentration
For a given enzyme concentration, the rate of an
enzyme reaction increases with increasing substrate concentration (Figure
8.32).
7. Enzyme Concentration
The rate of reaction is directly proportional to the enzyme concentration.
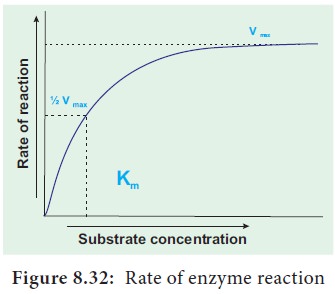
8. Introducing the Michaelis-Menton Constant (Km) and Its Significance
When the initial rate of reaction of an enzyme is measured over a range of substrate concentrations (with a fixed amount of enzyme) and the results plotted on a graph. With increasing substrate concentration, the velocity increases – rapidly at lower substrate concentration.
However the rate increases
progres-sively, above a certain concentration of the substrate the curve
flattened out. No further increase in rate occurs.
This shows that the enzyme is working at maximum
velocity at this point. On the graph, this point of maximum velocity is shown
as Vmax.
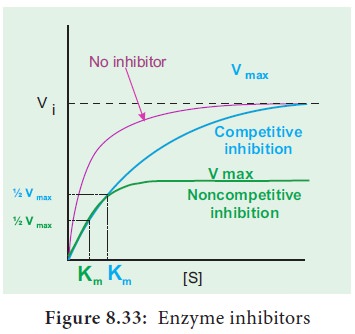
9. Inhibitors of Enzyme
Certain substances present in the cells may react
with the enzyme and lower the rate of reaction. These substances are called inhibitors. It is of two types competitive and non-competitive (Figure
8.33).
10. Competitive Inhibitor
![]()
![]()
![]()
Molecules that resemble the shape of the substrate
and may compete to occupy the active site of enzyme are known as competitive inhibitors. For Example:
the enzyme that catalyses the
reaction between carbon di oxide and the CO2 acceptor molecule in
photosynthesis, known as ribulose
biphosphate carboxylase oxygenase
(RUBISCO) is competitively inhibited
by oxygen/carbon-di-oxide in the
chloroplast. The competitive inhibitor is malonate
for succinic dehydrogenase (Figure
8.34).
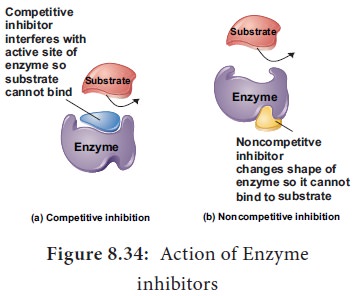
11. Non-competitive Inhibitors
There are certain inhibitors which may be unlike
the substrate molecule but still combines with the enzyme. This either blocks
the attachment of the substrate to active site or change the shape so that it
is unable to accept the substrate. For example the effect of the amino acids
alanine on the enzyme pyruvate kinase in the final step of glycolysis.
Certain non-reversible/irreversible
inhibitors bind tightly and permanently
to an enzyme and destroy its catalytic properties entirely. These could
also be termed as poisons . Example
– cyanide ions which blocks cytochrome
oxidase in terminal oxidation in cell aerobic respiration, the nerve gas sarin blocks a
neurotransmitter in synapse transmission.
12. Allosteric Enzymes
They modify enzyme activity by causing a reversible
change in the structure of the enzyme active site. This in turn affects the
ability of the substrate to bind to the enzyme. Such compounds are called allosteric
inhibitors. Eg. The enzyme hexokinase
which catalysis glucose to glucose-6 phosphate in glycolysis is inhibited by
glucose 6 phosphate. This is an example for feedback allosteric inhibitor.
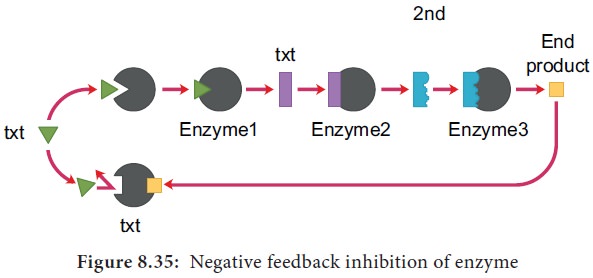
13. End Product Inhibition (Negative Feedback Inhibition)
When the end product of a metabolic pathway begins
to accumulate, it may act as an allosteric inhibitor of the enzyme controlling
the first step of the pathway. Thus the product starts to switch off its own
production as it builds up. The process is self – regulatory. As the product is
used up, its production is switched on once again. This is called end-product inhibition (Figure 8.35).
14. Enzyme Cofactors
Many enzymes require non-protein components called cofactors for their efficient activity.
Cofactors may vary from simple inorganic ions to complex organic molecules.
They are of three types: inorganic ions,
prosthetic groups and coenzymes (Figure
8.36).
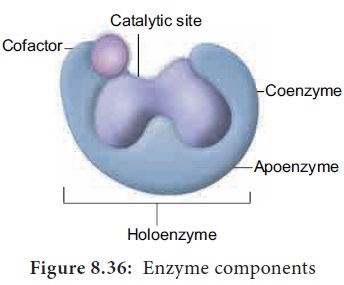
• Holoenzyme
– active
enzyme with its non protein
component.
•
Apoenzyme
– the
inactive enzyme without its non
protein component.
•
Inorganic
ions help to increase the rate of reaction
catalysed by enzymes. Example: Salivary amylase activity is increased in the
presence of chloride ions.
•
Prosthetic
groups are organic molecules that
assist in catalytic function of an enzyme. Flavin adenine dinucleotide (FAD)
contains riboflavin (vit B2), the function of which is to accept hydrogen.
‘Haem’ is an iron-containing prosthetic group with an iron atom at its centre.
•
Coenzymes
are
organic compounds which act as
cofactors but do not remain attached to the enzyme. The essential chemical
components of many coenzymes are vitamins. Eg. NAD, NADP, Coenzyme A, ATP
15. Nomenclature of Enzymes
![]()
![]()
![]()
Most of the enzymes have a name based on their
substrate with the ending –ase. For
example lactase hydrolyses lactose and amylase hydrolyses amylose. Other
enzymes like renin, trypsin do not depict any relation with their function.
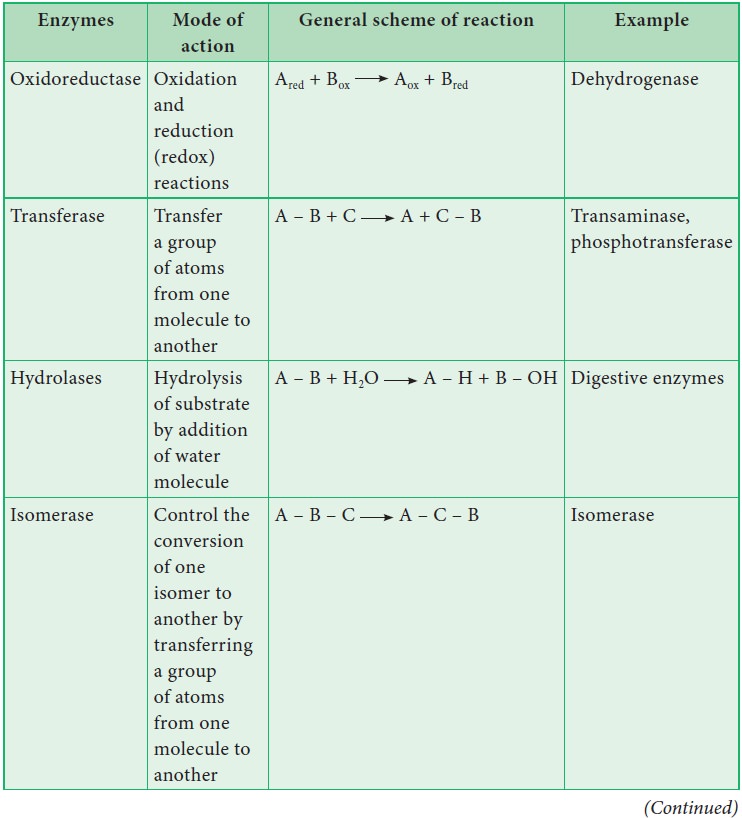
16. Classification of Enzymes
Enzymes are classified into six groups based on
their mode of action.

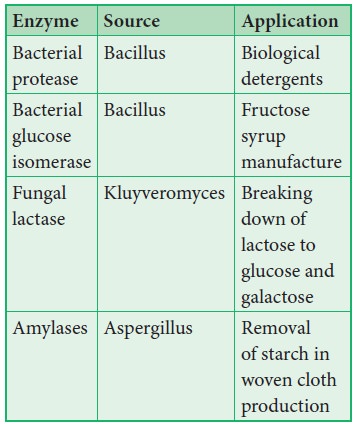
17. Uses of Enzymes
Related Topics