Chapter: 11th Botany : Chapter 8 : Biomolecules
Carbohydrates - Biomolecules
Carbohydrates
Carbohydrates are organic compounds made of carbon
and water. Thus one molecule of water combines with a carbon atom to form CH 2O
and is repeated several (n) times to form (CH2O)n
where n is an integer ranging from 3–7. These are also called as saccharides. The common term sugar
refers to a simple carbohydrate such as a monosaccharide or disaccharide that
tastes sweet are soluble in water (Figure 8.7).
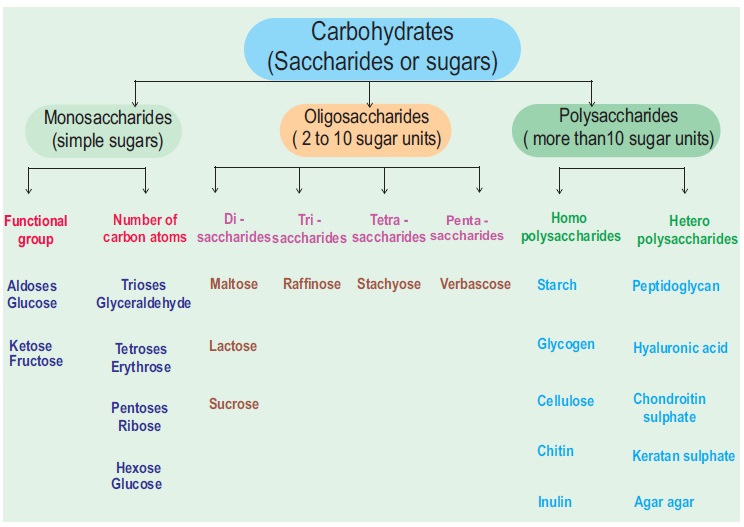
1. Monosaccharides – The Simple Sugars
Monosaccharides
are
relatively small molecules
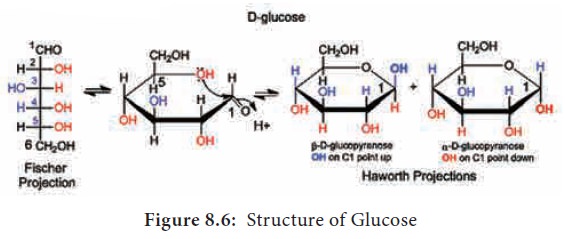
constituting single sugar unit. Glucose has a chemical formula of C6H12O6
. It is a six carbon molecule and hence is called as hexose (Figure 8.6).
All monosaccharides contain one of two functional
groups. Some are aldehydes, like glucose and are referred as aldoses ; other are ketones, like
fructose and are referred as ketoses.
2. Disaccharides
Disaccharides are formed when two monosaccharides join
together. An example is sucrose. Sucrose is formed from a
molecule of α-glucose
and a molecule of fructose.
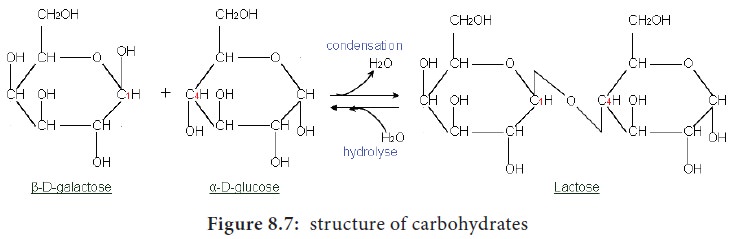
This is a condensation reaction releasing water.
The bond formed between the glucose and fructose molecule by removal of water
is called glycosidic bond. This is
another example of strong, covalent bond.
In the reverse process, a disaccharide is digested
to the component monosaccharide in a hydrolysis reaction. This reaction
involves addition of a water (hydro) molecule and splitting (lysis) of the
glycosidic bond.
3. Polysaccharides
These are made of hundreds of monosaccharide units. Polysaccharides also called "Glycans". Long chain of branched or unbranched monosaccharides are held together by glycosidic bonds. Polysaccharide is an example of giant molecule, a macromolecule and consists of only one type of monomer. Polysaccharides are insoluble in water and are sweetless. Cellulose is an example built from repeated units of glucose monomer.
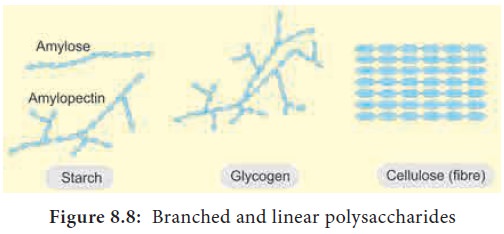
Depending on the function, polysaccharides are of
two types - storage polysaccharide and
structural polysaccharide (Figure 8.8).
4. Starch
Starch is a storage polysaccharides made up of
repeated units of amylose and amylopectin. Starch grains are made up of successive layers of amylose and
amylopectin, which can be seen as growth rings. Amylose is a linear, unbranched
polymer which makes up 80% of
starch. Amylopectin is a polymer with some 1, 6 linkages that gives it a
branched structure.
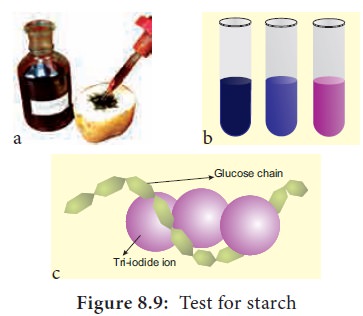
5. Test for Starch
We test the presence of starch by adding a solution
of iodine in potassium iodide. Iodine molecules fit nearly into the starch
helix, creating a blue-black colour
(Figure 8.9).
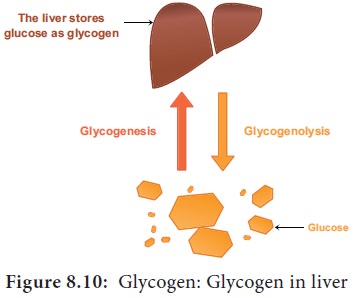
6. Glycogen
Glycogen is also a storage polysaccharide otherwise
called as animal starch. It is the
only carbohydrate stored in animals and fungi. Like amylopectin glycogen is a
polymer of glucose with (α1-6) linked branches. Glycogen is seen in liver
cells, skeletal muscle fibre and throughout the human body except brain (Figure
8.10).
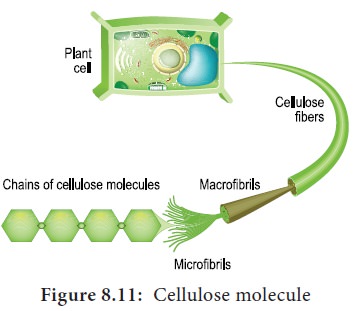
7. Celluloses
![]()
![]()
![]()
Cellulose is a structural polysaccharide made up of
thousands of glucose units. In this case, β-glucose
units are held together by 1,4 glycosidic linkage, forming long unbranched
chains. Cellulose fibres are straight and uncoiled. It has many industrial uses
which include cellulose fibres as cotton, nitrocellulose for explosives,
cellulose acetate for fibres of multiple uses and cellophane for packing
(Figure 8.11).
Most herbivores have a problem:
➢ Cellulose is one of the most abundant organic compound in the
biosphere.
➢ eat grass: principle component is cellulose
➢ cannot produce cellulase
Solution: Mutualistic bacteria in digestive system produce cellulases.
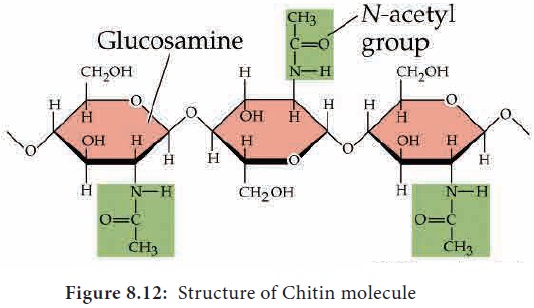
8. Chitin
Chitin is a homo polysaccharide with amino acids
added to form mucopolysaccharide.
The basic unit is a nitrogen containing glucose derivative known as N-acetyl glucosamine. It forms the
exoskeleton of insects and other arthropods. It is also present in the cell
walls of fungi (Figure 8.12).
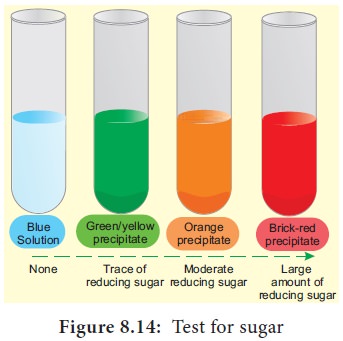
9. Test for Reducing Sugars
Aldoses and ketoses are reducing sugars. This means
that, when heated with an alkaline solution of copper (II) sulphate (a blue
solution called benedict’s solution),
the aldehyde or ketone group reduces Cu2+ ions to Cu+ ions forming brick red
precipitate of copper(I) oxide. In the process, the aldehyde or ketone group is
oxidised to a carboxyl group (–COOH). This reaction is used as test for
reducing sugar and is known as Benedict’s
test. The results of benedict’s test depends on concentration of the sugar.
If there is no reducing sugar it remains blue (Figure 8.14).
•
Sucrose is not a reducing sugar
•
The greater the concentration of reducing sugar,
the more is the precipitate formed and greater is the colour change.
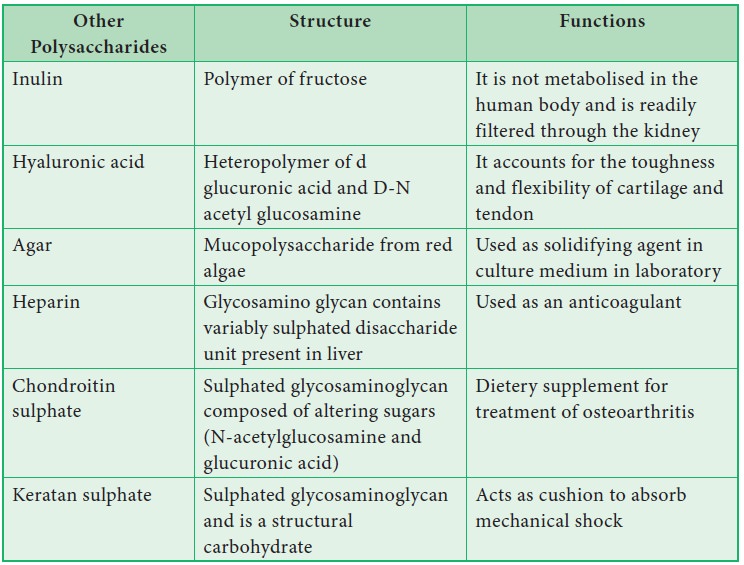
Other Sugar Compounds
Related Topics