Chapter: Modern Medical Toxicology: Neurotoxic Poisons: Anticonvulsants and Antiparkinsonian Drugs
Valproic Acid - Anticonvulsant (Anti-Epileptic) Drug
Valproic Acid
Uses
■■ Valproic acid
(n-dipropylacetic acid) is a branched-chain carboxylic acid that is used in the
treatment of tonic-clonic, myoclonic and absence seizures.
■■ It is also useful in
the management of the manic phase of bipolar affective disorder.
Toxicokinetics
·
Valproic acid (related compounds:
sodium valproate, dival- proex, valpromide) is rapidly and completely absorbed
on oral administration, and peak concentrations usually occur in 1 to 4 hours.
·
Peak levels occur 4 to 5 hours after
ingestion of divalproex tablets.
·
Valproic acid is 88 to 91% protein
bound in healthy patients but decreased in uraemia. Following a massive
overdose however, saturated protein binding results in increased fraction of
unbound valproic acid.
·
The half-life in patients receiving
chronic doses of valproic acid usually ranges from 10 to 14 hours. The serum
half-life appears to be prolonged in the overdose situation and may be up to 30
hours.
·
Most of the drug is metabolised to
the conjugated ester of glucuronic acid, while mitochondrial metabolism
accounts for the remainder.
Clinical (Toxic) Features
· Acute
poisoning is characterised by lethargy, somnolence, confusion, gastrointestinal
upset (mainly vomiting), tachy-cardia, hypotension, encephalopathy, respiratory
depres-sion, coma, liver damage (with elevation of liver enzymes and hyperammonaemia), metabolic
acidosis, thrombocy-topenia, leukopenia, pancytopenia, aplastic anaemia, and
acute pancreatitis.
o
Hypotension is often refractory to fluid resuscitation and
vasopressor therapy.
o
Significant QT interval prolongation appears to be a common
effect of acute valproic acid poisoning.
o
The manufacturer of valproic acid has issued a warning
concerning the development of life-threatening pancreatitis in children and
adults following therapeutic dosing. Some cases of haemorrhagic pancreatitis
with rapid progression from initial symptoms to death have been described.
Onset of pancreatitis may be shortly after initiation of valproic acid therapy
or following several years of use.
o
Miosis and nystagmus have been reported.
o
Cerebral oedema and coma have been reported in fatal cases.
o
Onset of toxicity and peak levels may be delayed more than 8
hours after ingestion of divalproex, enteric coated formulations, or
coingestion with drugs that slow gastrointestinal absorption.
·
Chronic poisoning is strongly associated with hepatotox-
icity which may terminate fatally. Jaundice, drug-induced hepatitis,
hepatocellular necrosis, transient elevated liver enzymes (SGPT/ALT, SGOT/AST),
and fatal cholestatic hepatitis have been associated with chronic valproate
administration. Fatal hepatotoxicity is reported in 1 out of every 800 children
under the age of 2 years following antiepileptic therapy with valproic acid. It
is suggested that valproic acid may induce a carnitine deficiency in young
children and result in non-specific symptoms of deficiency, hepatotoxicity, and
hyperammonaemia. Carnitine supplementation (100 mg/kg/day or 2 gm/day, divided
into 3 or 4 doses) may help prevent the onset of hepatotoxicity.
o
Apnoea, pulmonary haemorrhage, and bronchopneu- monia have
been reported with chronic valproate administration.
o
Seizures, behavioural changes (irritability, longer and
deeper sleep, superficial sleep, hyperactivity, increased alertness, lassitude,
drowsiness, increased sociability, calmness, increased sadness, happiness,
aggression), multiple system atrophy, extrapyramidal disorder, and cerebellar
syndrome have also been seen as adverse effects of chronic therapeutic use.
o
Severe hyperammonaemic encephalopathy has been reported
following chronic therapy, and as a syner- gistic effect of other
anticonvulsants (phenobarbitone, phenytoin, and topiramate) administered with
valproic acid.
o
Renal failure has also occurred in some cases.
o
Hypothermia has been reported in children following
short-term therapeutic dosing.
o
Parkinson’s syndrome has been associated with chronic
valproate therapy.
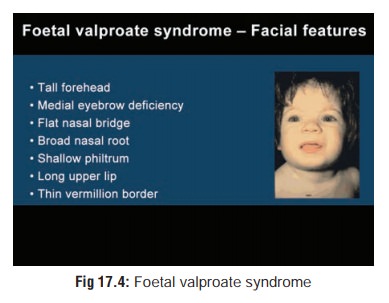
o Valproic acid may be teratogenic and is suspected to cause spina bifida and facial malformations (foetalvalproate syndrome)(Fig 17.4). The risk of congenital anomalies is greater in infants whose mothers were treated with multiple anti-epileptic drugs at the same time. Valproic acid in combination with other primary anti-epileptic drugs may be more teratogenic than other combinations of anti-epileptic drugs.
Usual Fatal Dose
■■ Therapeutic levels
of valproate vary between 50 and 100 mcg/ml. Serum concentrations are not reliable
for predicting severity of CNS depression in valproic acid poisoning cases;
however, more serious effects (coma, acidosis, aspiration, respiratory
depression) are generally seen at concentrations > 850 mcg/ml.
■■ Patients
ingesting less than 400 mg/kg of valproate are not likely to develop severe
toxicity. Severe toxicity has been associated with ingestions of 19 to 45 grams
in adults.
Treatment
·
Treatment of acute valproate
poisoning involves the usual measures of stabilisation, administration of activated
char-coal, and supportive measures.
·
Haemodialysis or charcoal
haemoperfusion appear to be beneficial.
·
Several cases have been reported
concerning the use of combinations of multiple-dose activated charcoal, forced
diuresis and continuous arteriovenous haemofiltration (CAVH) following severe
valproic acid intoxications.
·
CAVH is recommended when imminent
haemodynamic instability is present.
·
Orthoptic liver transplantation
could help patients with fulminant hepatic failure.
·
While carnitine supplementation has
been suggested to counter hyperammonaemia, its actual utility is doubtful.
Carnitine has also been used in cases of valproic acid overdose, though
conclusive beneficial effects have not been demonstrated.
Related Topics