Chapter: Plant Biochemistry: ATP is generated by photosynthesis
The electron chemical proton gradient can be dissipated by uncouplers to heat
The electron chemical proton gradient can be dissipated by uncouplers to heat
Photosynthetic electron transport from water to NADP+ is coupled with photophosphorylation. Electron transport occurs only if ADP and phos-phate are present as precursor substances for ATP synthesis. When an uncoupler is added, electron transport proceeds at a high rate in the absence of ADP; electron transport is then uncoupled from ATP synthesis. Therefore, in the presence of an uncoupler, ATP synthesis is abolished.
The chemiosmotic hypothesis explains the effect of uncouplers (Fig. 4.2). Uncouplers are amphiphilic compounds, soluble in both water and lipids. They are able to permeate the lipid phase of a membrane by diffusion and in this way to transfer a proton or an alkali ion across the membrane, thus eliminating a proton concentration gradient or a membrane potential, respectively. In the presence of an uncoupler a proton gradient is absent, but protons are transported by ATP synthase from the stroma to the thyla-koid lumen. This proton transport costs energy: ATP is hydrolyzed to ADP and phosphate. This is the reason why uncouplers cause an ATP hydrolysis (ATPase).
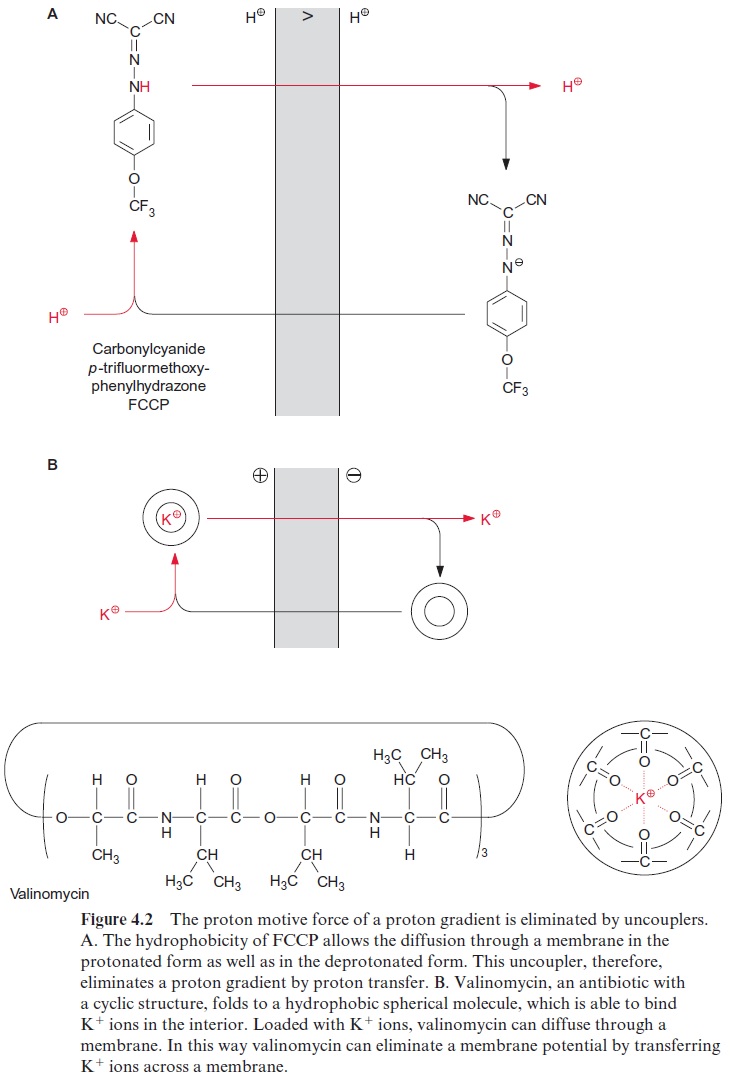
Figure 4.2A shows the effect of the uncoupler carbonylcyanide-p-trifluormethoxyphenylhydrazone (FCCP), which is a weak acid. FCCP dif-fuses in the undissociated (protonated) form from the compartment with a high proton concentration (on the left in Fig. 4.2A), through the membrane into the compartment with a low proton concentration, where it finally dis-sociates into a proton and the FCCP anion. The proton remains and the FCCP anion returns by diffusion to the other compartment, where it is pro-tonated again. In this way the presence of FCCP at a concentration of only 7 · 10–8 mol/L results in complete dissipation of the proton gradient. The substance SF 6847 (3.5-Di (tert-butyl)-4-hydroxybenzyldimalononitril) (Fig. 4.3) has an even higher uncoupling effect. Uncouplers such as FCCP or SF 6847, which transfer protons across a membrane, are called protonophores.

In addition to the protonophores, there is a second class of uncouplers, termed ionophores, which are able to transfer alkali cations across a membrane and thus dissipate a membrane potential. Valinomycin, an antibiotic from Streptomyces, is such an ionophore (Fig. 4.2B). Valinomycin is a cyclic mol-ecule containing the sequence (L-lactate)-(L-valine)-(D-hydroxyisovalerate)-(D-valine) three times. Due to its hydrophobic outer surface, valinomy-cin is able to diffuse through a membrane. Oxygen atoms directed towards the inside of the valinomycin molecule form the binding site for dehydrated Rb+ and K+ ions. Na+ ions because of their small size are only very loosely bound. When K+ ions are present, the addition of valinomycin results in the elimination of the membrane potential. The ionophore gramicidine, not discussed here in detail, is also a polypeptide antibiotic. Gramicidine incor-porates into membranes and forms a transmembrane ion channel by which both alkali cations and protons can diffuse through the membrane.
The chemiosmotic hypothesis was proved experimentally
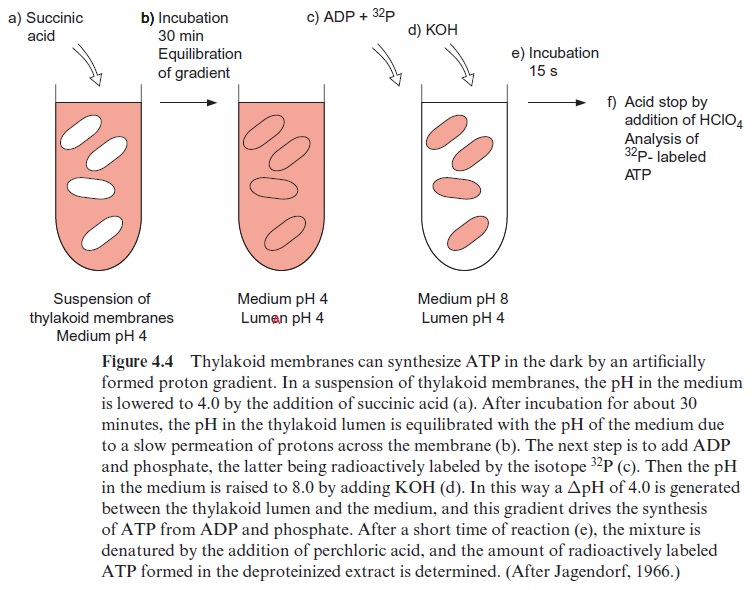
In 1966 the American scientist André Jagendorf presented conclusive evi-dence for the validity of the chemiosmotic hypothesis involved in chloro-plast photophosphorylation (Fig. 4.4). He incubated thylakoid membranes in an acidic medium of pH 4 in order to acidify the thylakoid lumen by unspecific uptake of protons. In a next step he added inorganic phosphate and ADP to the thylakoid suspension and then increased the pH of the medium to pH 8 by adding an alkaline buffer. This led to the sudden gen-eration of a proton gradient of ΔpH =4, and for a short time ATP was synthesized. Since this experiment was carried out in the dark, it presented evidence that synthesis of ATP in chloroplasts can be driven without illu-mination just by a pH gradient across a thylakoid membrane.
Related Topics