Chapter: Essential Clinical Immunology: Immunological Aspects of Allergy and Anaphylaxis
Systemic Anaphylaxis and Anaphylactoid Reactions
SYSTEMIC ANAPHYLAXIS AND ANAPHYLACTOID REACTIONS
Overview
Systemic anaphylaxis represents the clinical manifestation of type I hypersensitivity that occurs when a specific antigen and a homocytotrophic antibody interact. The reaction can be sudden and progress rap-idly, often without a clear cause. Death can occur because of vascular collapse or airway obstruction. The term anaphylaxis is derived from the Greek ana, meaning back-ward, and phylaxis, meaning protection. Poiter and Richet coined the term in 1902 after sea anemone antigen injected into a previously tolerant dog caused a fatal reac-tion instead of the expected immunological protection, or prophylaxis.
Generally, antigen-specific IgE anti-bodies on mast cells, or basophils, inter-act with previously encountered antigens mediating anaphylaxis. Mast cells are found in large numbers beneath cutaneous and mucosal surfaces and are closely asso-ciated with blood vessels and peripheral nerves. Basophils, most closely related to eosinophils, function similarly to mast cells. Basophils are present in the circula-tion, while mast cells are present only in tissue but in much greater numbers. When either of these cell types is triggered, a biphasic release of mediators occurs. The pathophysiology of anaphylaxis is clini-cally defined by the physiological effects of the immediate-phase and late-phase medi-ators on the target organs. By definition, anaphylaxis involves the cardiovascular, respiratory, gastrointestinal, or epidermal system; in most cases, multiple organs are involved.
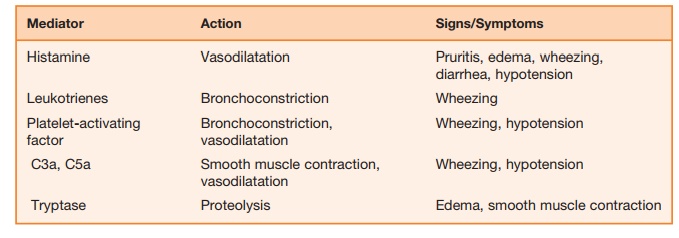
In the immediate phase, preformed enzymes and toxic mediators such as his-tamine, tryptase, and heparin are the pre-dominant bioactive substances released. Acting on H1 and H2 receptors, histamine promotes multiple effects such as increased vascular permeability, vasodilatation, con-striction of bronchial smooth muscle, and increased mucous secretion. Clinically, these reactions lead to
1. Airway and laryngeal edema and bron-chospasm with potential for complete asphyxiation;
2. Gastrointestinal tract smooth muscle contraction, causing pain, vomiting, and diarrhea;
3. Blood vessel dilatation with potential for progression to circulatory collapse;
4. Cutaneous vascular permeability, resulting in flushing, urticaria, and angioedema.
Late-phase reactions are characterized by induced production of mediators such as leukotrienes, chemokines, and cytokines, which are not preformed. Therefore, the effects of these mediators are seen later into the course, usually hours after the onset of an anaphylactic reaction. The late-phase effects include activation and recruitment of TH2 inflammatory cells, including neu-trophils and eosinophils, as well as prom-ulgation of the smooth muscle contraction and vasodilatation initiated by the imme-diate-phase reactants.
Treatment of anaphylaxis is aimed at restoring blood pressure, decreasing tis-sue edema, and reversing bronchospasm. Subcutaneous or intravenous epinephrine, oral or intravenous antihistamines, and systemic steroids are the mainstay of treat-ment. Patients should be monitored for at least six to twelve hours because late-phase reactions are possible.
Experimental Models
Animal models have been use to under-stand various manifestations of anaphy-laxis. Circulatory collapse typically occurs in dogs; rabbits may suffer acute pulmo-nary hypertension; and guinea pigs may experience acute respiratory obstruction. These reactions are classically mediated by the interaction of IgE; the high-affinity IgE receptor found on mast cells, FcεRI; and histamine. However, findings from several rodent studies suggest an alternative path-way involving the IgG receptor (Fc γ RIII), macrophages, platelets, and platelet-activating factor (PAF) may be more important in anaphylaxis than previously realized. Strait and colleagues immunized wild-type, IgE-deficient, FcεRI-deficient, and mast cell–deficient mice using goat antimouse IgD antibody. This technique induced mastocytosis and a large response to goat antigen (IgG) with increased IgE and IgG production. After fourteen days, they challenged the mice with antigen (goat IgG) or rat antimouse IgE monoclonal anti-body (mAb). The severity of anaphylaxis was gauged by changes in body tempera-ture, physical activity, and mortality. Find-ings in this experimental model included similar anaphylactic responses regardless of anti-IgE, mAb-induced, or goat IgG antigen challenge. Anti-IgE, mAb-induced anaphylaxis was FcεRI dependent and mediated predominantly by histamine. In contrast, antigen-induced anaphylaxis was Fc γ RIII and macrophase dependent with PAF as the predominant mediator. This mouse model provides evidence that ana-phylaxis may have alternative pathways and mediators contributing to its pathogen-esis. Table 9.2 summarizes key substances and their role in anaphylactic reactions.
Table 9.2 Mediators of Anaphylaxis
Representative Agents Causing Anaphylaxis
Multiple substances have been implicated as possible causes of anaphylaxis. The most common substances include drugs, specifically low-molecular-weight com-pounds. In most cases, the parenteral mode of administration is implicated, although oral, inhaled, and even cutaneous exposure can induce anaphylaxis as well.
Penicillin, the prototypic beta-lactam antibiotic, is the most frequent cause of anaphylaxis in humans. The reaction occurs when the compound is covalently bound to tissue carrier proteins to form drug-protein conjugates known as haptens. Ninety-five percent of tissue-bound peni-cillin is haptenized as benzylpenicilloyl, termed the major antigenic determinant. IgE antibodies directed at this hapten appear to be implicated in type I hypersensitivity reactions.
Skin testing using a commercially avail-able benzylpenicilloyl polylysine conjugate (Pre-Pen) may be used to predict the likeli-hood of an immediate-type reaction. Skin testing cannot predict non-IgE-mediated reactions; therefore, a thorough history to determine the necessity for skin testing is important.
Insect venom, another common cause of anaphylaxis, accounts for approximately forty to fifty deaths each year in the United States. The Hymenoptera order, including hornets, wasps, honeybees, and yellow jack-ets, contains the most common allergenic species. Immunotherapy is 90 to 95 percent effective after five years of therapy.
Foods, including nuts and crustaceans (i.e., shrimp), are frequently the cause of anaphylactic reactions. With exercise-induced anaphylaxis, certain foods that are normally tolerated such as celery, apples, and shrimp can induce anaphylaxis when ingestion is followed by exercise.
In recent years, natural rubber latex has been recognized as a source of ana-phylaxis. Exposure can occur through con-tact or inhalation, and various scenarios of exposure include surgical procedures, dental exams, or sexual intercourse with latex condoms. Health care workers and spina bifida patients are particularly at risk. IgE antibodies to latex antigen Hev b1 have been demonstrated by immunoassay and are felt to play a pathogenic role in this clinical entity.
Anaphylactic reactions involve an immunological mechanism. However, there are subsets of reactions clinically indistinct from anaphylaxis, which occur in a non-IgE-mediated fashion. In these anaphylactoid reactions, certain substances such as morphine and other agents such as radiocontrast media are common triggers. In contrast to IgE-mediated anaphylaxis, prior exposure is generally not required. In non-IgE anaphylaxis, some patients with selective absence of IgA demonstrate ana-phylactic symptoms following transfusion of IgA-containing plasma. Physiological changes such as exercise, emotions, and overheating can provoke symptoms in patients with cholinergic anaphlyactoid reaction. In this rare condition, an increase in core body temperature causes a rise in plasma histamine from mast cells. This disease is an exaggerated form of urticaria, described later. In all cases, no single pathogenic mechanism has been defined, but it is likely that direct mast cell activation accounts for most of these disorders; complement activation has also been reported.
Related Topics