Chapter: Plant Anatomy:An Applied Approach: The structure of xylem and phloem
Structure-function relationships in primary and secondary vascular tissues
Structure-function relationships in primary and secondary vascular tissues
The structure of the xylem and phloem in higher plants has been reviewed in a number of excellent texts. Here we propose to highlight some aspects that we consider relevant to the general student. For a study in more depth, readers are advised to refer to texts mentioned under the further reading.
The evolution of the conducting system, together with the development of the lignin synthesis pathway, must be among the important factors which contributed to the evolution of vascular plants.
![]()
![]() Physiologically, the elements within the xylem and phloem can act inde-pendently of one another, yet the phloem relies on water provided by the xylem in order to support the driving force required for long-distance translocation of assimilated material, hence the two are almost invariably found together. Structurally, many of the elements within the xylem (with the exception of parenchymatous elements and, sometimes, fibres) are dead at maturity and have highly modified wall structure. By contrast, the phlo-em (with the possible exception of sclerenchymatous elements) consists of cells which contain protoplasts at maturity. Sieve elements, including the more primitive sieve cells which occur in gymnosperms, are unique, in that they either lack nuclei or contain only remnants of nuclei, which are of un-known functional or regulatory capacity.
Physiologically, the elements within the xylem and phloem can act inde-pendently of one another, yet the phloem relies on water provided by the xylem in order to support the driving force required for long-distance translocation of assimilated material, hence the two are almost invariably found together. Structurally, many of the elements within the xylem (with the exception of parenchymatous elements and, sometimes, fibres) are dead at maturity and have highly modified wall structure. By contrast, the phlo-em (with the possible exception of sclerenchymatous elements) consists of cells which contain protoplasts at maturity. Sieve elements, including the more primitive sieve cells which occur in gymnosperms, are unique, in that they either lack nuclei or contain only remnants of nuclei, which are of un-known functional or regulatory capacity.
Transport through the xylem is driven in part by root pressure and by the evapotranspirative processes which take place mostly through stomata, lenticels and possibly through cracks in cuticular layers. Transport in the phloem, on the other hand, relies on a build-up of solutes (loaded into the sieve tubes at the sources) and the subsequent attraction of solvent to this area. Increasing pressure and the resultant enhancing of flow within the sieve tubes, but away from the source, moves them to some local or distant regions of the plant (termed sinks) where the solutes are unloaded and uti-lized. This is termed translocation.
In the primary plant body in leaves and stems, xylem and phloem gener-ally occur either in vascular bundles; or in roots, they are found as strands, with xylem and phloem on alternating radii. In plants that have undergone secondary thickening, the xylem and phloem in roots and stems become penetrated, by a series of secondary rays, which are usually parenchyma-tous. Whilst the interrelationships are easier to follow in primary vascular tissues, clearly the development of radially arranged ray tissues are of para-mount importance in the regulation of solute and solvent transport, and storage of metabolized materials.
One could ask ‘why do these disparate systems occur in close proximity?’ Clearly, the xylem does not require any direct inputs from the phloem, but does the phloem require or obtain any input from the xylem? Examination of the leaf blade bundles in gymnosperms and angiosperms demonstrates close spatial relationships between the tissues. Functional sieve tubes may occur adjacent to tracheary elements in many monocotyledonous plants, particularly among the grasses and sedges. If not in direct contact, they are separated by a few layers only of narrow parenchyma cells. In grasses and sedges these sieve tubes are the last to differentiate and mature, and curi-ously, they have thick walls, which in some instances (barley and wheat) have been reported to undergo lignification. Even more curious is the lack![]()
![]() of the identifiable companion cell–sieve tube complex, found in the early metaphloem in these plants, and commonly in all other vascular bundles in angiosperms.
of the identifiable companion cell–sieve tube complex, found in the early metaphloem in these plants, and commonly in all other vascular bundles in angiosperms.
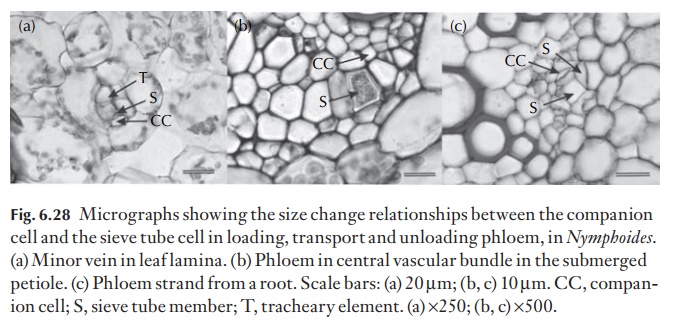
Perhaps the answers to the spatial proximity of the xylem to the phloem lies in the physiological requirements for successful phloem loading at the source, the maintenance of long-distance transport and the unloading process in local and distant sinks elsewhere in the plant. Figure 6.28 illus-trates the difference in size between loading, transport and unloading phloem sieve tubes and companion cells in Nymphoides. In the root, the ma-ture metaphloem is about 5–10 µm in cross-section, and the companion cells are 15–40 µm in cross-section; the increase in the size of the com-panion cells reflects its role in the phloem unloading process.
Related Topics