Chapter: Plant Anatomy:An Applied Approach: The structure of xylem and phloem
Secondary xylem construction - Structure of xylem
Secondary xylem construction
Whilst primary xylem consists of the axial cell system only, that is, xylem cells that are elongated parallel with the long axis of the organ or vascular trace in which they occur, secondary xylem, one of the products of the vas-cular cambium, is more complex. As we have seen, the cam-bium is composed of two sorts of cells, the axially elongated fusiform initials, which give rise to the axial system of cells, and the short, more or less isodiametric ray initials giving rise to the radial system or rays. Figure 3.1 shows the axial and radial systems in Alnus glutinosa wood.
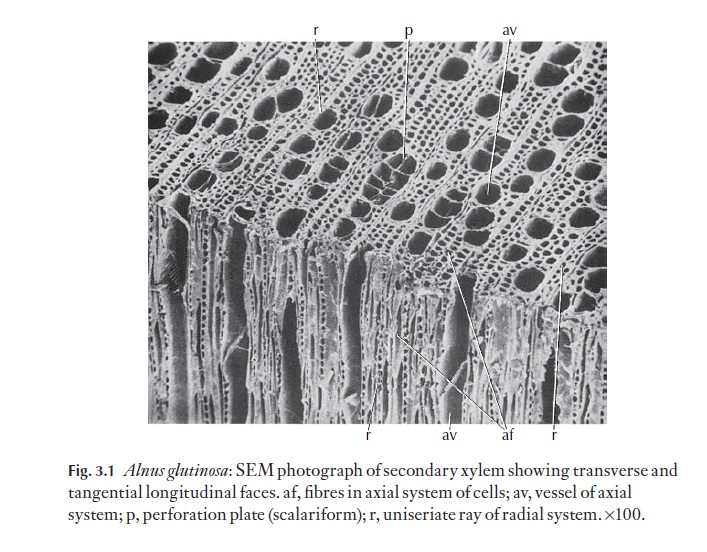
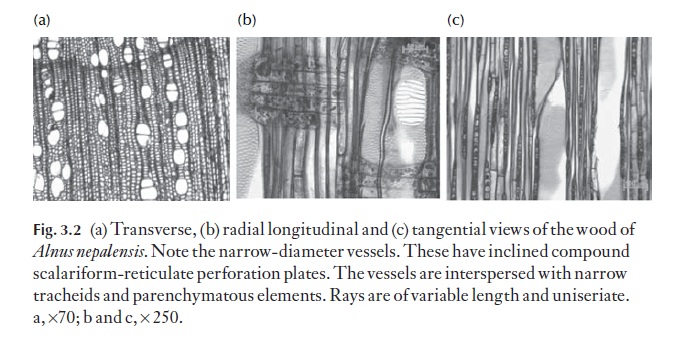
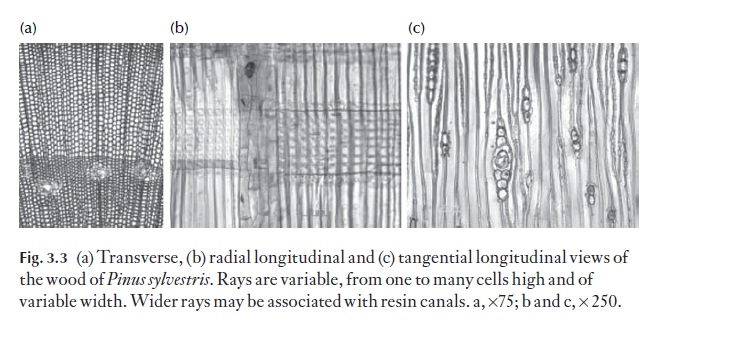
Because both axial and radial systems are present, the study of secondary xylem can only be carried out properly by examination of three specific planes of section from a block of wood. These are the transverse section (TS) the radial longitudinal section (RLS) and the tangential longitudinal section (TLS). These expose details of both systems of cells. Figures 3.2 and 3.3 show these planes of section.
The complex nature of secondary xylem will be addressed by considering first the less complex wood of gymnosperms, and next the much more varied wood found in the angiosperms.
Gymnosperm wood (conifer and Ginkgo wood)
In conifer woods (and gymnosperms generally), the axial water conduct-ing system is composed largely of tracheids (imperforate cell involved in water transport, i.e. with intact pit membrane between it and adjacent cell). Tracheids are rather like elongated boxes, with a rectangular cross-section and tapering upper and lower ends. They have thickened walls. The prima-ry wall is thin, and overlain on the side adjacent to the lumen by the second-ary wall. The secondary wall is usually constructed of a series of layers, in which the microfibrils, submicroscopic, thread-like and usually cellulosic components are laid down. Microfibrils are often arranged in a spiral, wound along the long axis of the cell. The orientation of the microfibrils differs in the successive layers, each layer often reversing the direction of spiral winding of that in adjacent layers. The matrix of the wall is lignified. Tracheids communicate with one another mainly through bordered pits in the lateral (radial) walls. The size, number of rows and details of pit struc-ture are often characteristic for given species or genera. Figure 3.3 shows sections of Pinuswood, and the glossary (see Pit, bordered) gives a recon-struction of the bordered pit pair between adjacent tracheids. Tracheids formed during the flush of spring growth are usually wider radially than those formed later in the growing season. It is normally easy to see the ex-tent of thickness of a growth increment for this reason. Note the torus, a central thickening on the pit membrane, which is characteristic of tracheid pits in confers. It acts like a plug, closing against the pit aperture if there are potentially damaging changes in pressure between adjacent tracheids. In ![]()
![]() this way, the spread of air embolisms may be controlled and reduced. Thick-ened areas between the pits termed bars of Sanio. These are characteristic of conifer wood.
this way, the spread of air embolisms may be controlled and reduced. Thick-ened areas between the pits termed bars of Sanio. These are characteristic of conifer wood.
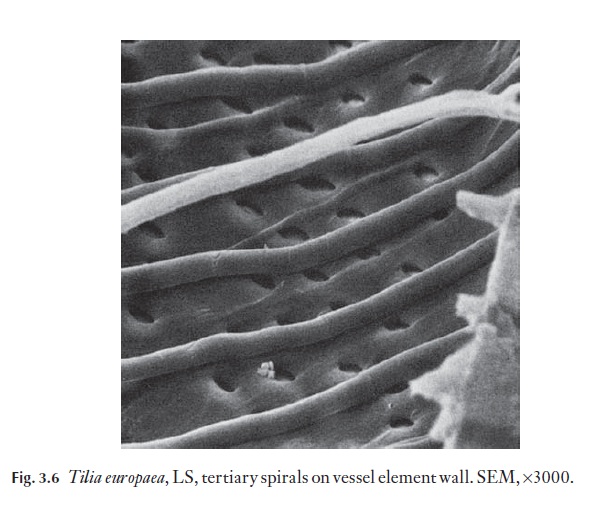
Sometimes a band of helical (spiral) thickening occurs inside the second-ary wall of the tracheid. This is characteristic of Pseudotsuga and Taxus and some Picea spp., Cephalotaxus and Torreya in the mature trunk wood. How-ever, many conifers have such a tertiary helical thickening on tracheid walls in twig wood, so if narrow diameter specimens are to be identified this must be borne in mind. In badly decomposed wood, helical splits may appear in tracheid walls, following the alignment of cellulose microfibrils in one of the wall layers; these can be confused with true helical thickenings.
There are no vessel elements.
Fibres are not normally found in conifer woods; if present, they run alongside the tracheids, axially. Axial parenchyma cells are rare; the cells are narrow, elongated axially, are situated alongside the tracheids, and have square end walls. Members of the Pinaceae (except Pseudolarix) and Sequoia spp. of Cupressaceae have axial resin ducts. These ducts are lined by a secre-tory epithelial layer of cells which remains thin-walled in Pinus species but becomes lignified in the other genera. The presence of resin ducts is, then, of taxonomic significance in the conifers. Many genera, like Cedrus, may have traumatic ducts (ducts arising from damage) which must not be con-fused with the regular resin ducts of Pinaceae.
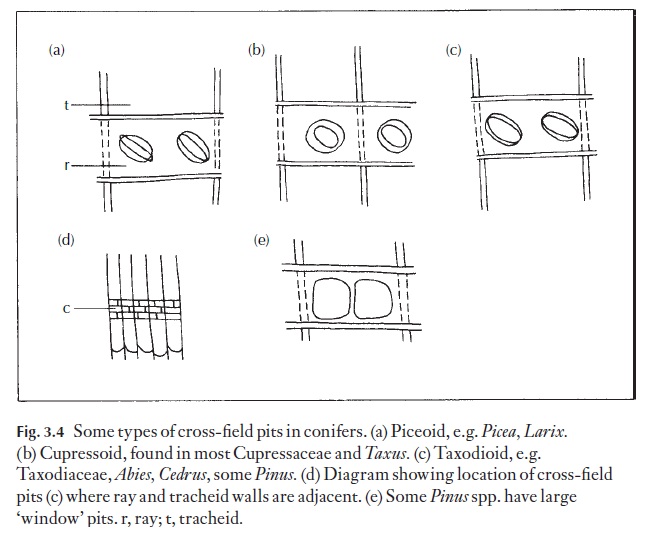
The radial (ray) system in gymnosperms consists of parenchymatous cells, which are, on the whole, procumbent. Some genera have radial trac-heids (ray tracheids) as well, and some Pinaceae have radial resin ducts. (Fig. 3.3c). The part of the ray cell wall bordering onto the tracheid is usually pit-ted. This is termed the cross-field area and the pits here are commonly characteristic for the genus or for a group of genera and can be used diag-nostically. Figure 3.4 shows some such pit types. The wall pitting and thick-enings of ray tracheids may also be of a characteristic form, for example, the ‘dentate’ ray tracheids ofPinus species. Rays are normally one, sometimes two cells wide, and several to many cells high.
Detailed variations between genera are too numerous for mention in this text, but useful references are given. There is a well-illustrated glossary of gymnosperm wood published by the International Association of Wood Anatomists.
Angiosperm wood
In dicotyledonous hardwoods there are more sorts of cell to be considered, and their appearance and distribution in the wood give rise to a great range of wood types. Although details of wall pitting still remain important in the identification of specimens, as for gymnosperms, there are many more characters that are readily observable in dicotyledonous woods than there are in coniferous woods. Families or groups of families often exhibit quite characteristic features for study and comparison.
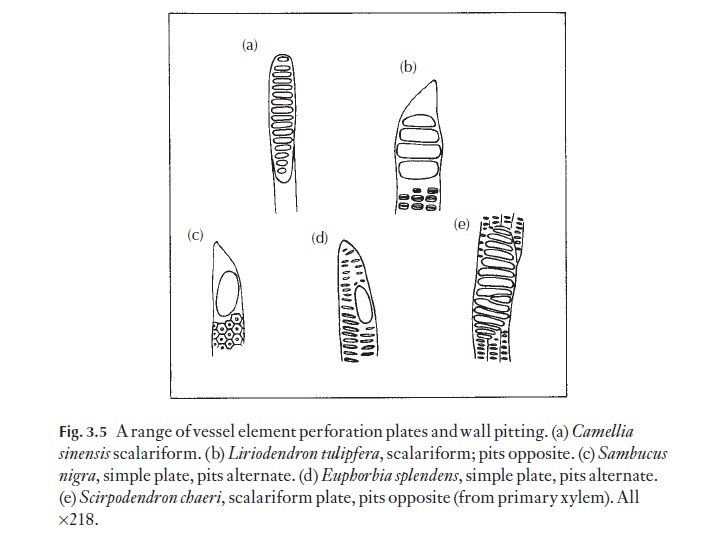
In the axial system of dicotyledons, tracheids are usually relatively sparse, most of the cells being vessel elements (the component cells of vessels; vessel elements are arranged in an axial file, with the terminal ele-ments imperforate at their outer ends, but with all element to element junc-tions with perforations (perforation plates) in their end walls) and fibres or fibre tracheids (cells intermediate in appearance between fibres and trac-heids; the pits usually intermediate in size) with varying amounts of axial parenchyma. These are all derivatives of the fusiform cambial initials. Fi-bres can be longer than the initial from which they were derived. Many fi-bres are capable of elongation by apical intrusive growth. Axial parenchyma cells are usually shorter than the initials that gave rise to them because the derivative cells often divide twice or three times, to form a chain of four or eight cells without significant enlargement. Xylem parenchyma cells nor-mally have somewhat thickened, lignified walls. Vessel elements are ex-tremely variable in their mature form, but particular forms are fairly consistently present in a given species. Details of wall pitting and per-foration plates are illustrated in Fig. 3.5. Each of these features may be used
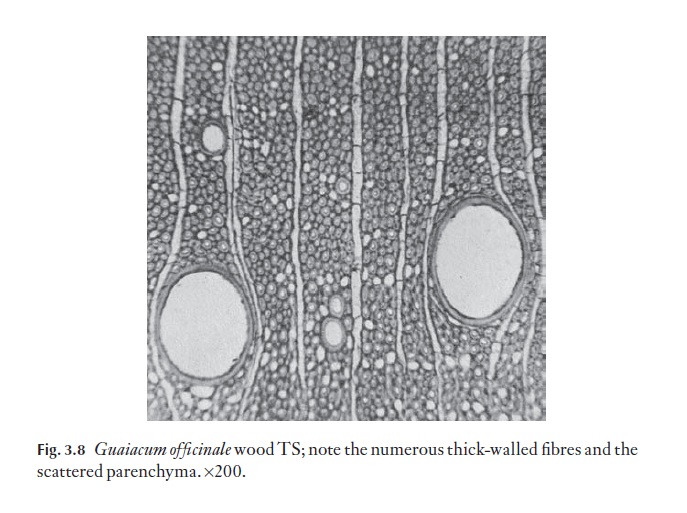
The main cell types in the axial system of most angiosperms are, then, vessels, fibres, tracheids and parenchyma. Some species have secretory ca-nals, lined by epithelial cells. Vessels, fibres and axial parenchyma may be arranged so that clear growth rings can be distinguished in TS, as in north and south temperate and montane species and those from other strongly seasonal regions. Lowland tropical species generally do not show growth ring boundaries. The overall density of a wood is governed not only by the sizes and proportions of the cell types present, but also by the thickness of their cell walls. Wall thickness also has a bearing on the hardness of a wood. For example balsa wood, Ochroma lagopus, is lighter than cork (Fig. 3.7 shows Ochroma pyramidalis wood in TS; note the thin-walled fibres); lignum vitae, Guaiacum offi cinale, is extremely dense and heavy (Fig. 3.8; note thick-walled fibres) and is used for making bowls, mallets and pulleys, among other things, and black ironwood,Krugiodendron ferreum is even more dense.

Where growth rings are apparent, vessels may grade in size, from wider in early growth, to narrower in late season growth. This produces a diffuse porous wood. In ring porous wood, vessels formed at the start of a growth
ring are much wider than those formed soon afterwards. Vessels as seen in TS may be solitary, in pairs or in small groups. The arrangement of these groups or multiples varies greatly. For example, there may be radial chains, oblique chains, tangential groups and so on. It is variations in vessel distri-bution, as seen in TS, that give the characteristic appearance of the wood. This is often further enhanced by the distribution of the axial parenchyma, when present.
If the strands are solitary and scattered, they might not make much impact on the appearance of the TS. If there is abundant parenchyma in strands, or groups which are either closely associated with the vessels, or clearly separated from them, a whole new range is established.
Rays are far more complex and show a wider range of variation in dicoty-ledons than in gymnosperms. They do not normally contain tracheids and may only sometimes contain vessels or perforated parenchyma cells. The parenchyma cells of which they are composed may exhibit a range of shapes and sizes.
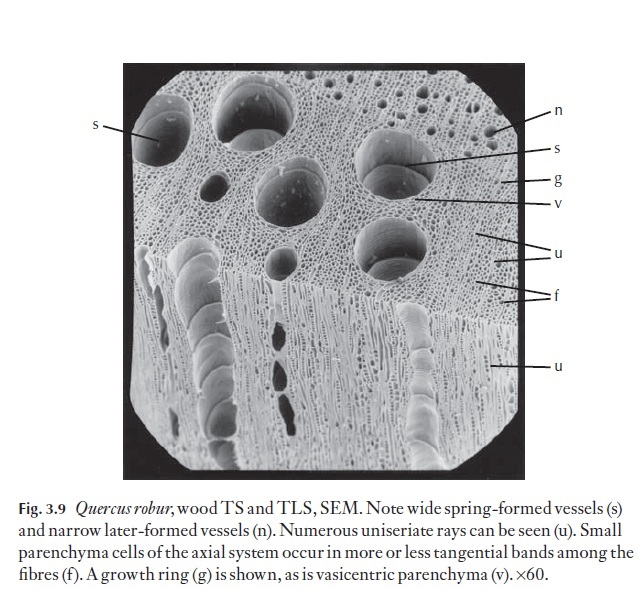
The cells making up the rays are evident in all three planes of section. In TS both their frequency and width (uniseriate – one cell wide to multiseri-ate, many cells wide) contribute strongly to the appearance of the wood. Where there are growth rings, rays in some species may show a widening of their cells at the rings. Rays may be all uniseriate, or they may be a mixture of uniseriate and wide multiseriate, or there can be a gradation from unise-riate to wide multiseriate, without a wide gap in width. In Castanea and Lithocarpus, for example, the rays are all uniseriate. Ulmus and Fagus are ex-amples of genera in which rays may be from one to several cells wide. Quer-cus has rays of two distinct sizes, some uniseriate and others wide andmultiseriate, with no intermediates (Fig. 3.9) shows uniseriate rays.
Rays may appear to have a ‘random’ distribution as seen in TLS, or, in some species, they may be storied, and arranged in clear horizontal bands (relative to the long axis of the wood). This difference can be used diagnostically because storied rays are characteristic of some families, for example Leguminosae (= Fabaceae), or of some genera, for example Hip-pophae (Elaeagnaceae)
In radial view (RLS) ray cells appear to look like courses of bricks in a wall. In some species all the cells are of similar size and proportions (homo-cellular); in others distinctly recognizable differences in cell size may occur (heterocellular). Cells of any particular size or shape are usually arranged in regular ‘courses’ or may be in particular positions, for example at the top and bottom of a ray. A range of different ray types is shown in Fig. 3.10, and includes procumbent and upright types.
With the possible combinations of cells and rays it is easy to see how dif-ferent particular woods may be from one another.

Related Topics