Chapter: Pharmaceutical Biotechnology: Fundamentals and Applications : Follicle-Stimulating Hormone
Structural Characteristics of Isohormones - Follicle Stimulating Hormone
ISOHORMONES
Structural Characteristics
As explained above, FSH exists in many distinct molecular forms
(isohormones), with identical poly-peptide backbones but diffences in
oligosaccharide structure, in particular in the degree of terminal sialylation.
These isohormones can be separated by chromatofocusing or isoelectric focusing
on the basis of their different isoelectric points [pI, as has been
demonstrated for follotropin b (de Leeuw et al., 1996)] (Fig.
2). The typical pattern for FSH indicates an
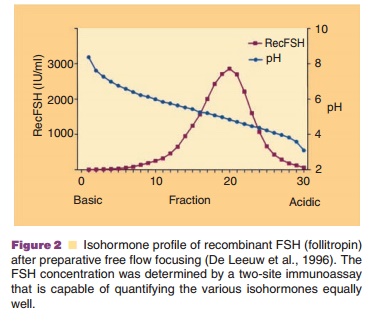
isohormone distribution between pI values of 6 and 4. To obtain
structural information at the subunit level, the two subunits were separated by
RP-HPLC and treated to release the N-linked carbohydrate side-chains. Fractions
with low pI values (acidic fractions) displayed a high content of tri- and
tetrasialo oligosaccharides and a low content of neutral and monosialo
oligosaccharides. For fractions with a high pI (basic fractions) value the
reverse was found. The β-subunit carbohydrate side chains appeared to bemore
heavily sialylated and branched than the α-subunit carbohydrate side chains.
The low pI value isohormones of follitropin b have a
high sialic acid/ galactose ratio and are rich in tri- and tetra-antennary
N-linked carbohydrate side chains, as compared with the side chains of the high
pI value isohormones.
Related Topics