Chapter: Plant Anatomy:An Applied Approach: Practical microtechnique
Staining - Materials and methods in Practical microtechnique
Staining
Whilst observation in the natural state is important, it may be difficult for the student to differentiate, for example, between unlignified and lignified tissues and it is for this reason that we recommend staining sections. There are several staining combinations that may be used to enhance details with-in sections. Sections of freshly cut material should be washed gently, to re-move cell debris which will obscure details once the section has been stained.
Two main types of stain can be used: (i) those which are temporary, whose colour fades, or which gradually damage the section; and (ii) those which are regarded as permanent. Even permanent stains may lose their colour if exposed to sunlight, so be careful and store your collection in the dark.
With care, stains can be selected to give the maximum contrast between the various cell and tissue types in the plant. They might be selected be-cause they colour particular parts of the cell wall structure and indicate its chemical composition. The stains and the protocol described below are in daily use in many laboratories throughout the world, and not just those as-sociated with the authors. Those who want comprehensive lists of stains, procedures and protocols, should see the books by Gurr (1965), Foster (1950) or Peacock revised by Bradbury (1973) to mention but three of the many guides to micro technique.
![]()
![]()
We have included other techniques which will be useful to the student on the CD-ROM. These can be accessed quite simply by following the ‘Tech-niques’ links associated with each of the exercises.
Temporary stains
1 1% aqueous methylene blue. All cell walls turn blue, except cutin orcutinized walls which remain unstained; cell walls take up a degree of in-tensity of blue depending upon their chemical composition and physical structure; various wall layers frequently stain differently. The stain may be mixed with 50% glycerine, about 10 ml of a 1% aqueous stain, to 90 ml of 50% glycerine, and sections mounted directly into this medium. This mixture is also useful for staining macerated tissues which are difficult to handle. A drop of washed macerate in water is mixed with a drop of the mixture on a slide, and the coverslip put on.
2 Chlorzinciodine solution (CZI, Schulte’s solution). This solutionconsists of: 30 g zinc chloride, 5 g potassium iodide, 1 g iodine and 140 ml distilled water. Cellulose walls turn blue, starch turns blue-black, lignin and suberin turn yellow and moderately lignified walls turn green-blue. Sections are placed on the slide, and a drop or two of CZI added. This can be drawn off and replaced by 50% glycerine after 2–4 minutes, but satisfac-tory results can be obtained by adding 50% glycerine directly, and mount-ing in the mixture. This stain swells the walls and eventually dissolves them. Consequently, care must be taken when describing wall thickness.
4 Chlorazol black saturated solution in 70% alcohol. Stains walls blackor grey and is particularly good for showing pitting.
5 Saturated carbolic acid solution. Sections are mounted directly inthe solution (which should be kept off the hands). Silica bodies usually turn pink; this helps to distinguish them from crystals which remain colourless.
6 Phloroglucin/HCl. The phloroglucin is added to the section, and thenthe dilute HCl. Lignin turns red. Caution: HCl is highly corrosive of skin, clothes and the microscope!
7 Sudan IV. Sections can be mounted directly in the stain. Stains fats,cuticles turn orange.
![]()
![]() 8 Ruthenium red.Mucilage and some gums turn pink. Sections can bemounted directly in the stain.
8 Ruthenium red.Mucilage and some gums turn pink. Sections can bemounted directly in the stain.
A simple double stain
Fuchsin, aniline blue and iodine in lactophenol (FABIL) is a most useful stain and mountant for all types of plant material. Although not available commercially it is easily prepared from the following stock solutions: lac-tophenol : phenol (crystals), glycerol, lactic acid and distilled water in equal parts by weight.
Aniline blue, 0.5% in lactophenol. A
Basic Fuchsin, 0.5% in lactophenol. B
Iodine, KI, 0.6 g : lactophenol, 1 litre. C
The stain is made up by mixing the stock solutions in the proportions of A,4 : B,6 : C,5. This is allowed to stand overnight and after filtering it is ready for use and will keep indefinitely.
FABIL is superior to other commonly used temporary reagents such as aniline double stain, phloroglucinol or zinc chloride, the particular advantage being that it incorporates a differential stain, a clearing agent and a semi-permanent mountant. Sections cut from fresh or alcohol-stored material are transferred directly to the stain, enclosed by a coverslip and examined immediately. If desired, the stain may be replaced after about 10 minutes by plain lactophenol or 25% aqueous glycerol, but this is by no means essential. The solution is only slightly volatile and mounts can be kept for several months without drying up, although additions from time to time at the edge of the coverslip will prevent air bubbles from being drawn in. However, if it is necessary to store the mounts for a very long time the coverslip can be sealed with melted beeswax or nail varnish. The cytoplasmic cell contents, including nuclei and callose plugs of sieve tubes, are stained blue, Cellulose walls stain a paler blue and lignified tissue becomes a bright yellow, orange or pink, depending on the nature of the specimen. Staining is rapid but improves with time and overstaining is not possible, even after immersion for several weeks.
Much of the success of this reagent is due not simply to staining but also to differential clearing, so that in practice the tissues are rendered more dis-tinct than the above-mentioned colour reactions would suggest. Moreover, because of the extremely good light-transmitting properties of the solu-tion, cellular detail can be studied even in thick sections. FABIL may also be used for mounting fungal or algal material, including seaweeds, and causes very little distortion. Alternatively the aniline blue solution A, or fuchsin solution B, may be used alone. With fungi, gentle heating of the mount improves the absorption of the stain.
Safranin (1% in 50% alcohol) and Delafield’s haematoxylin is a very useful combination. In cell walls cellulose turns dark blue; lignin turns red and cellulose walls with some lignins turn purple.
Freshly mix the prepared safranin with matured Delafield’s haematoxy-lin in the proportions of 1 : 4; filter. The stock mixture can be used for up to about 1 week, but should be filtered before use each day.
Sections should be transferred from water (after washing all bleach away) into a suitable dish containing the stain, and covered. Most sections take up the stain in 24 hours, others will need less time. The sections should then transferred to a Petri dish in which there is 50% acidified alcohol (use a few drops of conc. HCI). This solution removes the stain, acting on the safranin first. Whilst the object is to obtain a satisfactory colour balance, only expe-rience will tell when this has been reached.
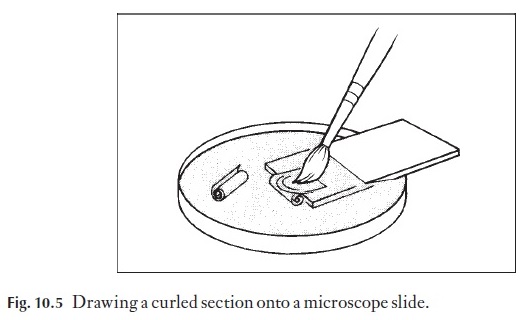
Sections should be removed when they still appear to be slightly dark or overstained for best results – the colours look less intense under the micro-scope. The decolorizing action is halted by placing the sections into a Petri dish containing 95% alcohol. After about 5 minutes they can be transferred to absolute alcohol in a covered Petri dish. Five minutes later they can be transferred either to a 50–50 mixture of absolute alcohol xylene in a covered dish, or this step may be eliminated and they may be transferred directly into xylene. Xylene fumes should not be inhaled; use a fume hood. After 10 minutes in the xylene, the sections can be mounted in Canada balsam on the microscope slide. Any milkiness in the section at this stage means that water is still present, and the section should be taken back through xylene, then fresh absolute alcohol, fresh absolute alcohol xylene and fresh xylene before remounting. Sections which curl up or roll up should be straightened out in 50% alcohol. As they progressively dehydrate in purer alcohol they become more brittle and cannot be un-rolled without breaking. Curled wood sections can be flattened by drawing them over the edge of a slide partly immersed in 50% alcohol (Fig. 10.5), a process needing three hands! Alternatively a section lifter can be used. Once on the slide they can be ‘set’ using a few drops of 95% alcohol.
If it is more convenient to stain overnight, the safranin-haematoxylin mixture can be used in the proportions 94 : 6. Although fast green can be used as a counter stain for safranin, we have found that haematoxylin pro-duces a colour which photographs better on normal panchromatic film. Al-cian blue may be used as an alternative to the haematoxylin; a 1% aqueous solution is satisfactory. It is easier to use and gives blue colours where the haematoxylin would have stained purple.
Fast Green can be used on its own as a stain for macerated material. The macerate is dehydrated by decanting alcohols of 50, 70, 90, 95% and abso
Related Topics