Chapter: Plant Anatomy:An Applied Approach: Practical microtechnique
Preparation of surfaces - Materials and methods in Practical microtechnique
Preparation of surfaces
Many of the the staining methods applied to sections can equally well be used for surface preparations.
Leaf surfaces
The epidermis of most leaves can be readily removed by the scraping method. Only those leaves with very prominent veins, or large, numerous hairs pose problems, and these will demand a great deal of patience.
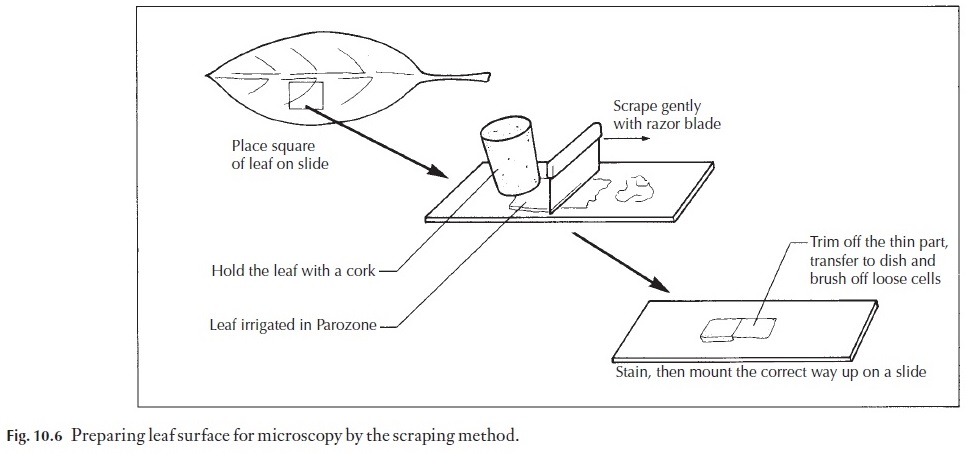
Material may be fresh, or washed after fixing. A suitable piece is cut from the leaf (Fig. 10.6). The surface which you wish to study is placed face down on a glazed tile or on a glass plate. It is irrigated with a few drops of sodium hypochlorite. One end may be held securely with a cork, and the other end scraped lightly with a safety razor blade. With practice a double-edged blade can be used, but it is better to start off by using a single-edged blade. The blade is held at 90 degrees to the leaf (Fig. 10.6) and the gentle scraping is continued, adding extra hypochlorite as necessary, keeping the leaf well irrigated. If the leaf is not severed by a forceful scrape, you will end up with a thin, clear area, which can be cut off, placed in a cavity block for a few minutes with sodium hypochlorite, and then washed in a petri dish con-taining tap water. Loosely adhering cells can then be brushed off with a fine camel hair paintbrush.
The preparation can then be viewed in water under the microscope. It is easy to see if enough has been scraped away. Experience will soon enable you to judge when the end point is reached in scraping. Make sure the sur-face is placed the right way up before the final mounting.
With some material it is unnecessary to scrape the leaf, since the epider-mis can be removed by peeling it off the fresh leaf. This is done by folding the leaf to break the surface, and either stripping directly, pulling one part of the leaf downwards relative to the other, or by holding as thin a layer of the surface as possible between forceps and peeling it back.
Stem surfaces
A thin strip of stem surface can be obtained by making the first longitudinal section cut on the microtome just pass through the surface layers. This re-quires careful microtome adjustment, but is quite satisfactory.
Surface replicas
Sometimes it is not possible or desirable to remove the epidermis itself – a plant may be rare, or there may be little material readily available. A good imprint of the surface can be obtained with a film of cellulose acetate (nail varnish). It may be necessary to wipe the leaf surface with acetone to clean it. Then clear nail varnish is brushed on. More than one layer may be re-quired, and it is advisable to make the individual coats quite thin. Once dry, the nail varnish can be removed as a film and then mounted under a cover-slip, preferably in a medium with a different refractive index (immersion oil will work well) or little will be seen.
Although replicas can be made easily using a variety of materials (latex, for example), the epidermis itself is most useful in determining the shape of the epidermis, and its associated subsidiary and guard cells. Replicas can tell us little about cell contents.
Cuticular preparations
Various chemical treatments can be used to cause the cuticle to separate from the leaf. The cuticle is in many respects better than the replica, but can be very delicate.
![]()
![]() One method is to digest away the leaf tissues using nitric acid (care!). The cuticles will frequently float to the surface, or, when the leaf has been fully washed, can be teased off the dissolving tissues.
One method is to digest away the leaf tissues using nitric acid (care!). The cuticles will frequently float to the surface, or, when the leaf has been fully washed, can be teased off the dissolving tissues.
Clearing material
Whole thin leaves or stems or flowers can be made transparent by soaking them in chloral hydrate, washing and soaking in sodium hydroxide solu-tion, alternately, for several changes, with several hours at each stage
After the final washing, the organ can be carefully stained in safranin (1% in 50% alcohol) dehydrated through a very gradual alcohol series and mounted. Veins and sclereids show up well.
Alternatively, the preparation may be left unsigned for examination by various optical methods.
Related Topics