Chapter: Medical Physiology: Metabolism of Carbohydrates, and Formation of Adenosine Triphosphate
Role of Adenosine Triphosphate in Metabolism
Role of Adenosine Triphosphate in Metabolism
Adenosine triphosphate (ATP) is an essential link between energy-utilizing and energy-producing functions of the body (Figure 67–1). For this reason, ATP has been called the energy currency of the body, and it can be gained and spent repeatedly.
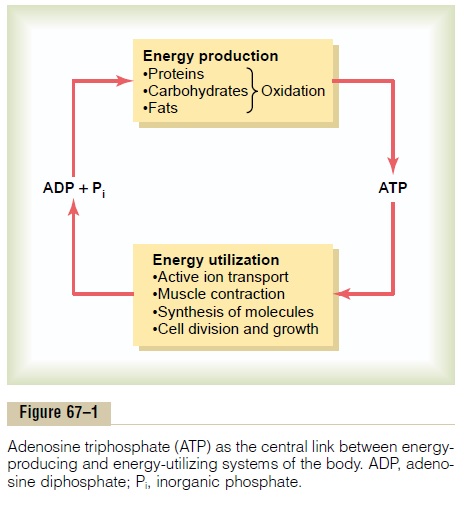
Energy derived from the oxidation of carbohydrates, proteins, and fats is used to convert adenosine diphosphate (ADP) to ATP, which is then consumed by the various reactions of the body that are necessary for (1) active transport of mole-cules across cell membranes; (2) contraction of muscles and performance of mechanical work; (3) various synthetic reactions that create hormones, cell mem-branes, and many other essential molecules of the body; (4) conduction of nerve impulses; (5) cell division and growth; and (6) many other physiologic functions that are necessary to maintain and propagate life.
ATP is a labile chemical compound that is present in all cells. It has the chemical structure shown in Figure 67–2. From this formula, it can be seen that ATP is a combination of adenine, ribose, and three phosphate radicals. The last two phosphate radicals are connected with the remainder of the molecule by high-energy bonds, which are indicated by the symbol ~.
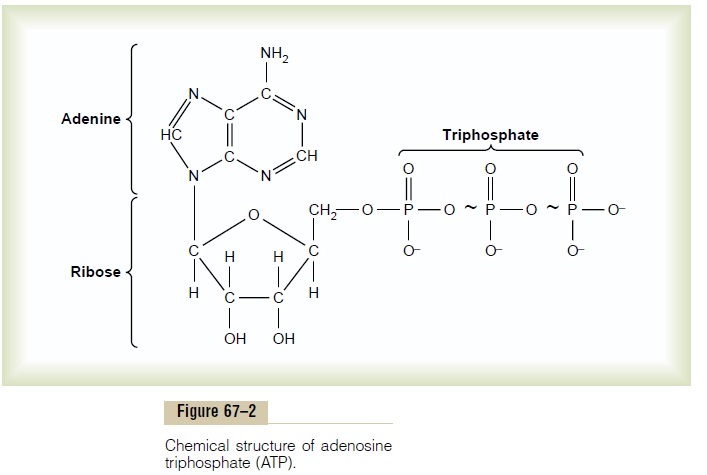
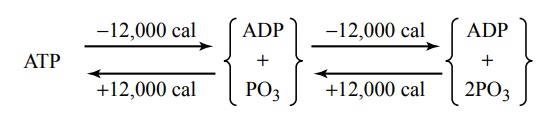
The amount of free energy in each of these high-energy bonds per mole of ATP is about 7300 calories under standard conditions and about 12,000 calories under the usual conditions of temperature and concen-trations of the reactants in the body. Therefore, in the body, removal of each of the last two phosphate radicals liberates about 12,000 calories of energy. After loss of one phosphate radical from ATP, the compound becomes ADP, and after loss of the second phosphate radical, it becomes adenosine monophosphate (AMP).
The interconversions among ATP, ADP, and AMP are the following:
ATP is present everywhere in the cytoplasm and nucleoplasm of all cells, and essentially all the physio-logic mechanisms that require energy for operation obtain it directly from ATP (or another similar high-energy compound—guanosine triphosphate [GTP]). In turn, the food in the cells is gradually oxidized, and the released energy is used to form new ATP, thus always maintaining a supply of this substance. All these energy transfers take place by means of coupled reactions.
Related Topics