Chapter: Medical Physiology: Metabolism of Carbohydrates, and Formation of Adenosine Triphosphate
Release of Energy from Glucose by the Pentose Phosphate Pathway
Release of Energy from Glucose by the Pentose Phosphate Pathway
In almost all the body’s muscles, essentially all the carbohydrates utilized for energy are degraded to pyruvic acid by glycolysis and then oxidized. However, this gly-colytic scheme is not the only means by which glucose can be degraded and used to provide energy. A second important mechanism for the breakdown and oxidation of glucose is called the pentose phosphate pathway (or phosphogluconate pathway), which is responsible for asmuch as 30 per cent of the glucose breakdown in theliver and even more than this in fat cells.
This pathway is especially important because it can provide energy independently of all the enzymes of the citric acid cycle and therefore is an alternative pathway for energy metabolism when certain enzymatic abnor-malities occur in cells. It has a special capacity for pro-viding energy to multiple cellular synthetic processes.
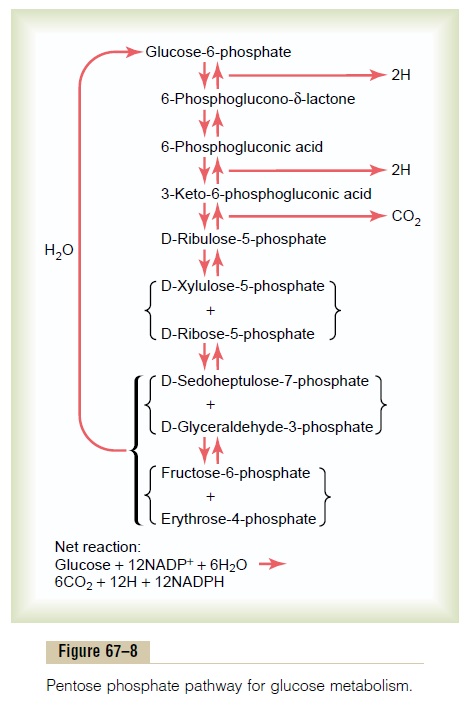
Release of Carbon Dioxide and Hydrogen by Means of the Pentose Phosphate Pathway. Figure 67–8 shows most of the basicchemical reactions in the pentose phosphate pathway. It demonstrates that glucose, during several stages of con-version, can release one molecule of carbon dioxide and four atoms of hydrogen, with the resultant formation of a five-carbon sugar, D-ribulose. This substance can change progressively into several other five-, four-, seven-, and three-carbon sugars. Finally, various combi-nations of these sugars can resynthesize glucose. However, only five molecules of glucose are resynthe-sized for every six molecules of glucose that initially enter into the reactions. That is, the pentose phosphatepathway is a cyclical process in which one molecule of glucose is metabolized for each revolution of the cycle. Thus, by repeating the cycle again and again, all the glucose can eventually be converted into carbon dioxide and hydrogen, and the hydrogen can enter the oxidative phosphorylation pathway to form ATP; more often, however, it is used for the synthesis of fat or other sub-stances, as follows.
Use of Hydrogen to Synthesize Fat; the Function of Nicotinamide Adenine Dinucleotide Phosphate. The hydrogen releasedduring the pentose phosphate cycle does not combine with NAD+ as in the glycolytic pathway but combines with nicotinamide adenine dinucleotide phosphate (NADP+), which is almost identical to NAD+ except for an extra phosphate radical, P. This difference is extremely significant, because only hydrogen bound with NADP+ in the form of NADPH can be used for the synthesis of fats from carbohydrates and for the synthesis of some other substances.
When the glycolytic pathway for using glucose becomes slowed because of cellular inactivity, the pentose phosphate pathway remains operative (mainly in the liver) to break down any excess glucose that con-tinues to be transported into the cells, and NADPH becomes abundant to help convert acetyl-CoA, also derived from glucose, into long fatty acid chains. This is another way in which energy in the glucose molecule is used other than for the formation of ATP—in this instance, for the formation and storage of fat in the body.
Glucose Conversion to Glycogen or Fat
When glucose is not immediately required for energy, the extra glucose that continually enters the cells is either stored as glycogen or converted into fat. Glucose is preferentially stored as glycogen until the cells have stored as much glycogen as they can—an amount suffi-cient to supply the energy needs of the body for only 12 to 24 hours.
When the glycogen-storing cells (primarily liver and muscle cells) approach saturation with glycogen, the additional glucose is converted into fat in liver and fat cells and is stored as fat in the fat cells.
Related Topics