Chapter: Modern Pharmacology with Clinical Applications: General Organization and Functions of the Nervous System
Receptors on the Autonomic Effector Cells
RECEPTORS ON THE
AUTONOMIC EFFECTOR CELLS
The receptors for
acetylcholine and related drugs (cholinoreceptors)
and for norepinephrine and related drugs (adrenoceptors)
are different. Acetylcholine will not interact with receptors for
norepinephrine, and nor-epinephrine will not interact with cholinoreceptors.
These receptors are selective not only for their respec-tive agonists but also
for their respective antagonist drugs; that is, drugs that antagonize or block
acetyl-choline at cholinoreceptors will not antagonize norepi-nephrine at
adrenoceptors and vice versa.
Cholinoceptors
The action of administered
acetylcholine on effector systems innervated by parasympathetic postganglionic
neurons (smooth muscle cells, cardiac muscle cells, and exocrine gland cells)
resembled the actions produced by the naturally occurring plant alkaloid muscarine. The actions of both
acetylcholine and muscarine on the vis-ceral effectors are similar to those
produced by parasympathetic nerve stimulation. Furthermore, the effects of
acetylcholine, muscarine, and parasympa-thetic nerve stimulation on visceral
effectors are antag-onized by atropine, another plant alkaloid.
The administration of
acetylcholine mimics the stimulatory effect of nicotine, the alkaloid from the to-bacco plant, on autonomic
ganglia and the adrenal medulla. It has become common practice to refer to the
effects of acetylcholine on visceral effectors as the mus-carinic action of acetylcholine and to its effects on
theautonomic ganglia and adrenal medulla as the nicotinic action of acetylcholine. The respective receptors are
called the muscarinic and nicotinic cholinoreceptors or the muscarinic and
nicotinic receptors of acetylcholine.
The action of acetylcholine
at the skeletal muscle motor end plate resembles that produced by nicotine.
Thus, the cholinoreceptor on skeletal muscle is a nico-tinic receptor. Based on
antagonist selectivity, however, the autonomic and somatic nicotinic receptors
are not pharmacologically identical .
Acetylcholine can stimulate a
whole family of re-ceptors. However, these receptors are sufficiently chem-ically
diverse that different exogenous agonists and antagonists can distinguish among
them. Great thera-peutic benefit has been obtained from this diversity be-cause
it allows the development of therapeutic agents that can selectively mimic or
antagonize actions of acetylcholine. Such a diversity of receptor subtypes
ex-ists for other neurotransmitters in addition to acetyl-choline.
Adrenoceptors
Adrenoceptors interact not
only with norepinephrine but also with the adrenal medullary hormone
epineph-rine and a number of chemically related drugs. However, the responses
produced by the drugs in dif-ferent autonomic structures differ quantitatively
or qualitatively from one another.
On the basis of the observed
selectivity of action among agonists and antagonists, it was proposed that two
types of adrenoceptors exist. These were designated as α- and β-adrenoceptors. Subsequently,
it has become necessary to classify the adrenoceptors further into α 1-, α 2-, β 1-, and β 2-receptor subtypes. Table 9.1
indicates present knowledge of the distribution of the subtypes of
adrenoceptors in various tissues.
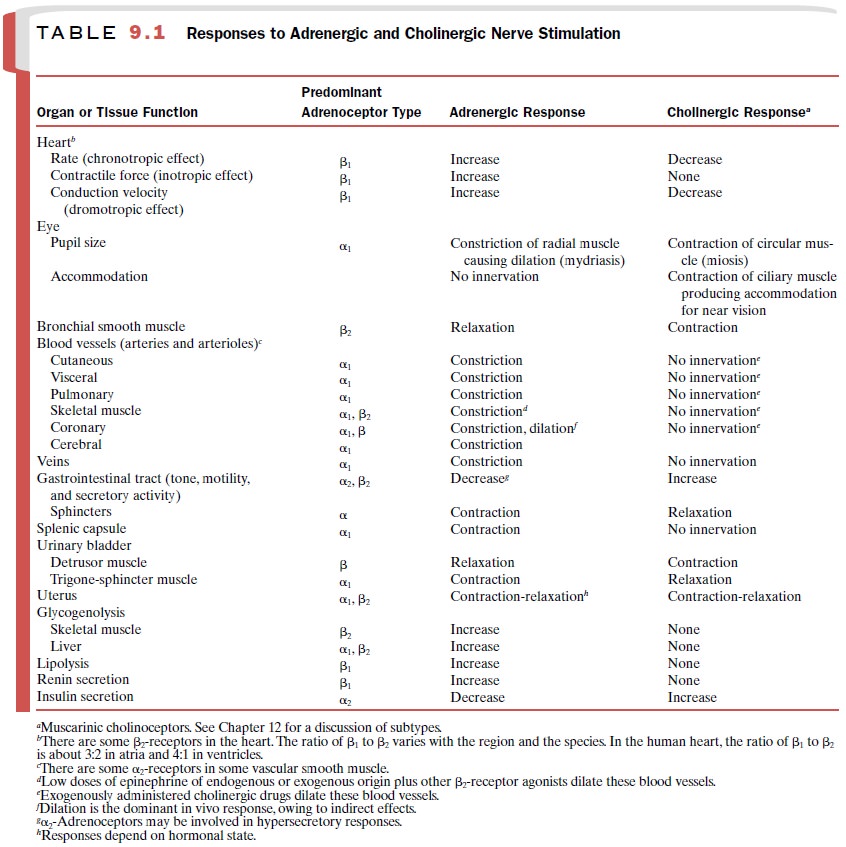
The α 1-adrenoceptors are located at
postjunctional (postsynaptic) sites on tissues innervated by adrenergic
neurons. α 2-Adrenoceptors having a presynaptic (i.e., neuronal) location are
involved in the feedback inhibi-tion of norepinephrine release from nerve
terminals (discussed later). α 2-Receptors also can occur postjunc-tionally. The β 1-adrenoceptors are found
chiefly in the heart and adipose tissue, while β 2-adrenoceptors are lo-cated in a number of
sites, including bronchial smooth muscle and skeletal muscle blood vessels, and
are asso-ciated with smooth muscle relaxation.
Activation of α1-adrenoceptors in smooth muscle of blood vessels leads to vasoconstriction, while activation of β 2-adrenoceptors in blood vessels of skeletal muscle produces vasodilation. Activation of β 1-adrenoceptors on cardiac tissue produces an increase in the heart rate and contractile force.
Norepinephrine and epinephrine are potent α- adrenoceptor agonists, while isoproterenol, a synthetic adrenomimetic, is selective
for β 1- and β 2-adrenocep-tors.
Norepinephrine and epinephrine are thus potent vasoconstrictors of vascular
beds that contain predomi-nantly α-adrenoceptors, while isoproterenol has little
effect in these vessels.
Isoproterenol and epinephrine
are potent β2-adrenoceptor agonists; norepinephrine is a relatively weak β2-adrenoceptor agonist.
Isoproterenol and epi-nephrine produce vasodilation in skeletal muscle, but
norepinephrine does not; rather it produces vasocon-striction through the α1-adrenoceptors.
Isoproterenol, epinephrine, and norepinephrine are potent β1-adreno-ceptor agonists;
thus, all three can stimulate the heart (Table 9.1).
The existence of a β 3-adrenoceptor has recently
been demonstrated in human adipose tissue along with the β 1-adrenoceptor. This
observation raises the possi-bility that eventually therapeutic drugs may
selectively alter lipid metabolism and therefore provide therapeu-tic
management of obesity. The β 3-receptor and the re-cently identified subtypes within the α 1- and α 2-receptor groups ( α1A, α 1B, etc.) also have not been
included in thetable, since as yet few therapeutic drugs distinguish among
these further subtypes. One exception is tam-sulosin, an antagonist with some
selectivity for α 1A-receptors in the urinary tract.
Presynaptic Receptors
Presynaptic or prejunctional receptors are
located on the presynaptic nerve
endings and function to control the amount of transmitter released per nerve
impulse and in some instances to affect the rate of transmitter synthesis
through some as yet undetermined feedback mechanism. For instance, during
repetitive nerve stimu-lation, when the concentration of transmitter released
into the synaptic or junctional cleft is relatively high, the released
transmitter may activate presynaptic receptors and thereby reduce the further
release of transmitter. Such an action may prevent excessive and prolonged
stimulation of the postsynaptic cell. In this case, the ac-tivation of the
presynaptic receptor would be part of a negative
feedback mechanism.
The presynaptic receptors may
have pharmacologi-cal significance, since several drugs may act in part ei-ther
by preventing the transmitter from reaching the presynaptic receptor, thus
causing excessive transmitter release, or by directly stimulating presynaptic
receptors and thereby diminishing the amount of transmitter re-leased per
impulse.
The inhibitory presynaptic α-adrenoceptors found on
noradrenergic neurons are of the α 2-subtype. Adrenoceptors of the β2 subclass also occur
presynap-tically, and activation of these receptors leads to en-hanced
norepinephrine release. The physiological and pharmacological importance of
these presynaptic β2-receptors is less certain than it is for presynaptic α2-receptors.
Presynaptic receptors for
nonadrenomimetic sub-stances (e.g., acetylcholine, adenosine) also have been
found on the sympathetic presynaptic nerve ending. Their importance and role in
the modulation of neuro-transmission have not been definitively established.
Related Topics