Chapter: Medical Physiology: Pulmonary Circulation, Pulmonary Edema, Pleural Fluid
Pulmonary Capillary Dynamics
Pulmonary Capillary Dynamics
Exchange of gases between the alveolar air and the pulmonary capillary blood is discussed in the next. However, it is important for us to note here that the alveolar walls are lined with so many capil-laries that, in most places, the capillaries almost touch one another side by side. Therefore, it is often said that the capillary blood flows in the alveolar walls as a “sheet of flow,” rather than in individual capillaries.
Pulmonary Capillary Pressure. No direct measurementsof pulmonary capillary pressure have ever been made. However,“isogravimetric” measurement of pul-monary capillary pressure, using a technique described, has given a value of 7 mm Hg. This is probably nearly correct, because the mean left atrial pressure is about 2 mm Hg and the mean pulmonary arterial pressure is only 15 mm Hg, so the mean pulmonary capillary pressure must lie somewhere between these two values.
Length of Time Blood Stays in the Pulmonary Capillaries. From histological study of the total cross-sectional area of all the pulmonary capillaries, it can be calcu-lated that when the cardiac output is normal, blood passes through the pulmonary capillaries in about 0.8 second. When the cardiac output increases, this can shorten to as little as 0.3 second. The shortening would be much greater were it not for the fact that additional capillaries, which normally are collapsed, open up to accommodate the increased blood flow. Thus, in only a fraction of a second, blood passing through the alveolar capillaries becomes oxygenated and loses its excess carbon dioxide.
Capillary Exchange of Fluid in the Lungs, and Pulmonary Interstitial Fluid Dynamics
The dynamics of fluid exchange across the lung capil-lary membranes are qualitatively the same as for peripheral tissues. However,quantitatively, there are important differences, as follows:
1. The pulmonary capillary pressure is low, about 7 mm Hg, in comparison with a considerably higher functional capillary pressure in the peripheral tissues of about 17 mm Hg.
2. The interstitial fluid pressure in the lung is slightly more negative than that in the peripheral subcutaneous tissue. (This has been measured in two ways: by a micropipette inserted into the pulmonary interstitium, giving a value of about –5 mm Hg, and by measuring the absorption pressure of fluid from the alveoli, giving a value of about –8 mm Hg.)
3. The pulmonary capillaries are relatively leaky to protein molecules, so that the colloid osmotic pressure of the pulmonary interstitial fluid is about 14 mm Hg, in comparison with less than half this value in the peripheral tissues.
4. The alveolar walls are extremely thin, and the alveolar epithelium covering the alveolar surfaces is so weak that it can be ruptured by any positive pressure in the interstitial spaces greater than alveolar air pressure (greater than 0 mm Hg), which allows dumping of fluid from the interstitial spaces into the alveoli.
Now let us see how these quantitative differences affect pulmonary fluid dynamics.
Interrelations Between Interstitial Fluid Pressure and Other Pressures in the Lung. Figure 38–6 shows a pulmonarycapillary, a pulmonary alveolus, and a lymphatic capil-lary draining the interstitial space between the blood capillary and the alveolus. Note the balance of forces at the blood capillary membrane, as follows:

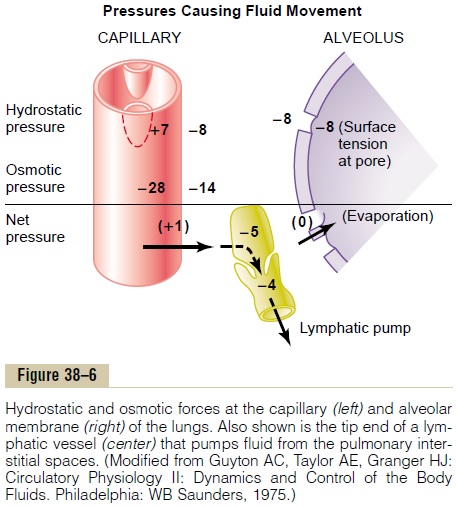
Thus, the normal outward forces are slightly greater than the inward forces, providing a mean filtration pressure at the pulmonary capillary membrane; thiscan be calculated as follows:

This filtration pressure causes a slight continual flow of fluid from the pulmonary capillaries into the inter-stitial spaces, and except for a small amount that evap-orates in the alveoli, this fluid is pumped back to the circulation through the pulmonary lymphatic system.
Negative Pulmonary Interstitial Pressure and the Mechanism for Keeping the Alveoli “Dry.” One of the most importantproblems in lung function is to understand why the alveoli do not normally fill with fluid. One’s first incli-nation is to think that the alveolar epithelium is strong enough and continuous enough to keep fluid from leaking out of the interstitial spaces into the alveoli. This is not true, because experiments have shown that there are always openings between the alveolar epithelial cells through which even large protein mol-ecules, as well as water and electrolytes, can pass.
However, if one remembers that the pulmonary cap-illaries and the pulmonary lymphatic system normally maintain a slight negative pressure in the interstitial spaces, it is clear that whenever extra fluid appears in the alveoli, it will simply be sucked mechanically into the lung interstitium through the small openings between the alveolar epithelial cells. Then the excess fluid is either carried away through the pulmonary lymphatics or absorbed into the pulmonary capillaries. Thus, under normal conditions, the alveoli are kept “dry,” except for a small amount of fluid that seeps from the epithelium onto the lining surfaces of the alveoli to keep them moist.
Pulmonary Edema
Pulmonary edema occurs in the same way that edema occurs elsewhere in the body. Any factor that causes the pulmonary interstitial fluid pressure to rise from the negative range into the positive range will cause rapid filling of the pulmonary interstitial spaces and alveoli with large amounts of free fluid.
The most common causes of pulmonary edema are as follows:
1. Left-sided heart failure or mitral valve disease, with consequent great increases in pulmonary venous pressure and pulmonary capillary pressure and flooding of the interstitial spaces and alveoli.
2. Damage to the pulmonary blood capillary membranes caused by infections such as pneumonia or by breathing noxious substances such as chlorine gas or sulfur dioxide gas. Each of these causes rapid leakage of both plasma proteins and fluid out of the capillaries and into both the lung interstitial spaces and the alveoli.
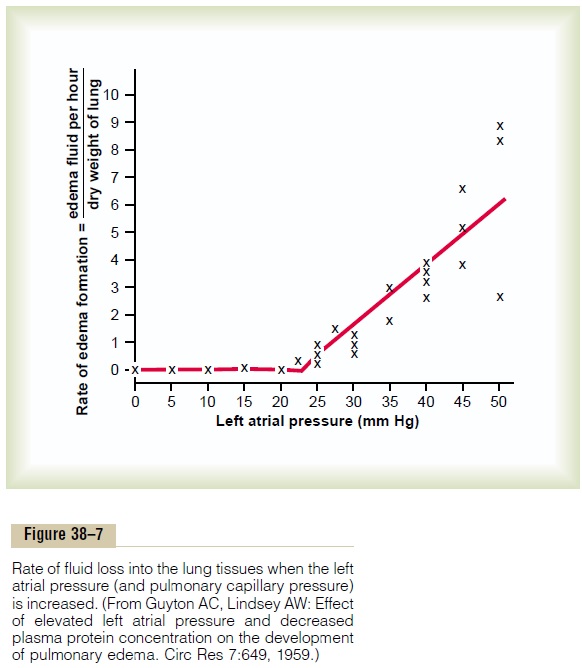
“Pulmonary Edema Safety Factor.” Experiments in animalshave shown that the pulmonary capillary pressure nor- mally must rise to a value at least equal to the colloid osmotic pressure of the plasma inside the capillaries before significant pulmonary edema will occur. To give an example, Figure 38–7 shows how different levels of left atrial pressure increase the rate of pulmonary edema formation in dogs. Remember that every time the left atrial pressure rises to high values, the pul-monary capillary pressure rises to a level 1 to 2 mm Hg greater than the left atrial pressure. In these experi-ments, as soon as the left atrial pressure rose above 23 mm Hg (causing the pulmonary capillary pressure to rise above 25 mm Hg), fluid began to accumulate in the lungs. This fluid accumulation increased even more rapidly with further increases in capillary pressure. The plasma colloid osmotic pressure during these experi-ments was equal to this 25 mm Hg critical pressure level. Therefore, in the human being, whose normal plasma colloid osmotic pressure is 28 mm Hg, one can predict that the pulmonary capillary pressure must rise from the normal level of 7 mm Hg to more than 28 mm Hg to cause pulmonary edema, giving an acutesafety factor against pulmonary edema of 21 mm Hg.
Safety Factor in Chronic Conditions. When the pul-monary capillary pressure remains elevated chronically (for at least 2 weeks), the lungs become even more resistant to pulmonary edema because the lymph vessels expand greatly, increasing their capability of car-rying fluid away from the interstitial spaces perhaps as much as 10-fold. Therefore, in patients with chronic mitral stenosis, pulmonary capillary pressures of 40 to 45 mm Hg have been measured without the develop-ment of lethal pulmonary edema.
Rapidity of Death in Acute Pulmonary Edema. When the pul-monary capillary pressure rises even slightly above the safety factor level, lethal pulmonary edema can occur within hours, or even within 20 to 30 minutes if the cap-illary pressure rises 25 to 30 mm Hg above the safety factor level. Thus, in acute left-sided heart failure, in which the pulmonary capillary pressure occasionally does rise to 50 mm Hg, death frequently ensues in less than 30 minutes from acute pulmonary edema.
Related Topics