Chapter: Mechanical : Engineering Thermodynamics : Gas Mixtures and Psychrometry
Psychrometry
PSYCHROMETRY
psychrometry is
a term used to describe the field of engineering concerned with the
determination of physical and thermodynamic properties of gas-vapor mixtures.
The term derives from the Greek psuchron meaning "cold" and metron
meaning "means of measurement"
Common applications
The principles of
psychrometry apply to any physical system consisting of gasvapor mixtures. The
most common system of interest, however, are mixtures of water vapor and air because
of its application in heating, ventilating, and air-conditioning and
meteorology.
Psychrometric ratio
The
psychrometric ratio is the ratio of the heat transfer coefficient to the
product of mass transfer coefficient and humid heat at a wetted surface. It may
be evaluated with the following equation
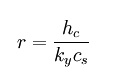
where:
•r = Psychrometric ratio,
dimensionless
•hc = convective heat
transfer coefficient, W m-2 K-1
•ky = convective mass
transfer coefficient, kg m-2 s-1
•cs = humid heat, J kg-1
K-1
Humid heat is
the constant-pressure specific heat of moist air, per unit mass of dry air.
The psychrometric ratio
is an important property in the area of psychrometrics as it relates the
absolute humidity and saturation humidity to the difference between the dry
bulb temperature and the adiabatic saturation temperature. Mixtures of air and
water vapor are the most common systems encountered in psychrometry. The
psychrometric ratio of air-water vapor mixtures is approximately unity which
implies that the difference between the adiabatic saturation temperature and
wet bulb temperature of air-water vapor mixtures is small. This property of
air-water vapor systems simplifies drying and cooling calculations often
performed using psychrometic relationships.
PSYCHROMETRIC
CHART
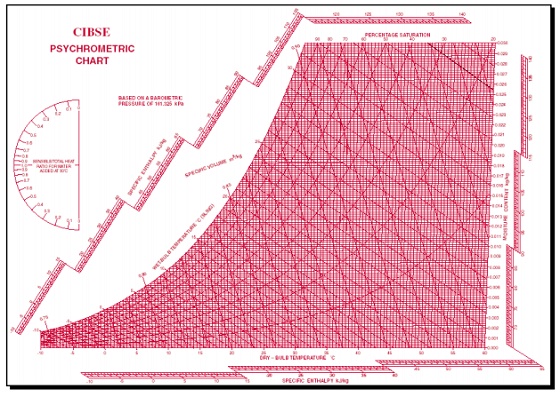
A psychrometric chart
is a graph of the physical properties of moist air at a constant pressure
(often equated to an elevation relative to sea level). The chart graphically
expresses how various properties relate to each other, and is thus a graphical
equation of state. The thermophysical properties found on most psychrometric
charts are:
•Dry-bulb
temperature (DBT) is that of an air sample, as determined by an
ordinary thermometer, the thermometer's bulb being dry. It is typically the
x-axis, the horizontal axis, of the graph. The SI units for temperature are
Kelvin; other units are Fahrenheit.
•Wet-bulb
temperature (WBT) is that of an air sample after it has passed
through a constant-pressure, ideal, adiabatic saturation process, that is,
after the air has passed over a large surface of liquid water in an insulated
channel. In practice, this is the reading of a thermometer whose sensing bulb
is covered with a wet sock evaporating into a rapid stream of the sample air.
The WBT is the same as the DBT when the air sample is saturated with water. The
slope of the line of constant WBT reflects the heat of vaporization of the water
required to saturate the air of a given relative humidity.
•Dew point
temperature (DPT) is that temperature at which a moist air sample at
the same pressure would reach water vapor saturation. At this saturation point,
water vapor would begin to condense into liquid water fog or (if below
freezing) solid hoarfrost, as heat is removed. The dewpoint temperature is
measured easily and provides useful information, but is normally not considered
an independent property. It duplicates information available via other humidity
properties and the saturation curve.
•Relative humidity (RH)
is the ratio of the mole fraction of water vapor to the mole fraction of
saturated moist air at the same temperature and pressure. RH is dimensionless,
and is usually expressed as a percentage. Lines of constant RH reflect the
physics of air and water: they are determined via experimental measurement.
Note: the notion that air "holds" moisture, or that moisture
dissolves in dry air and saturates the solution at some proportion, is an
erroneous (albeit widespread) concept (see relative humidity for further
details).
•Humidity ratio
(also known as moisture content, mixing ratio, or specific humidity) is the
proportion of mass of water vapor per unit mass of dry air at the given
conditions (DBT, WBT, DPT, RH, etc.). It is typically the y-axis, the vertical
axis, of the graph. For a given DBT there will be a particular humidity ratio
for which the air sample is at 100% relative humidity: the relationship
reflects the physics of water and air and must be measured. Humidity ratio is
dimensionless, but is sometimes expressed as grams of water per kilogram of dry
air or grains of water per pound of air.
•Specific enthalpy
symbolized by h, also called heat content per unit mass, is the sum of
the internal (heat) energy of the moist air in question, including the heat of
the air and water vapor within. In the approximation of ideal gases, lines of
constant enthalpy are parallel to lines of constant WBT. Enthalpy is given in
(SI) joules per kilogram of air or BTU per pound of dry air.
•Specific volume,
also called inverse density, is the volume per unit mass of the air
sample. The SI units are cubic meters per kilogram of dry air; other units are
cubic feet per pound of dry air.
Specific humidity
Specific
humidity is the ratio of water vapor to air (including water
vapor and dry air) in a particular mass. Specific humidity ratio is
expressed as a ratio of kilograms of water vapor, mw, per kilogram of
air (including water vapor), mt .
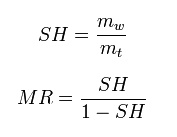
Relative
Humidity
The
relative humidity of an air-water mixture is defined as the ratio of the partial
pressure of water vapor in the mixture to the saturated vapor pressure of water
at a prescribed temperature. Relative humidity is normally expressed as a
percentage and is defined in the following manner
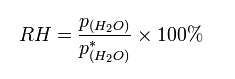
RH
BH is the relative humidity of the mixture being considered;
P(H2O) is the partial pressure of
water vapor in the mixture; and
P(H2O) is the saturated vapor
pressure of water at the temperature of the mixture. The international symbols U
and Uw, expressed in per cent, are gaining recognition.
Related Topics