Introduction - Periodic classification of Elements | 9th Science : Periodic classification of Elements
Chapter: 9th Science : Periodic classification of Elements
Periodic classification of Elements
Periodic
classification of Elements
Introduction
Think of a morning
prayer in your school. Students stand in rows which are horizontal as well as
vertical. Each class stands in a single line, height wise. Generally, height of
students of class I is the shortest and that of class 12 is the tallest.

The vendor in a medical
store could locate the medicines we seek in a flash of time with the use of a
pattern they are arranged. We can easily identify the books in the library as
quickly as possible.
There is a pattern in
all these cases and this pattern makes the selection easy. (Pattern: regular
arrangement)
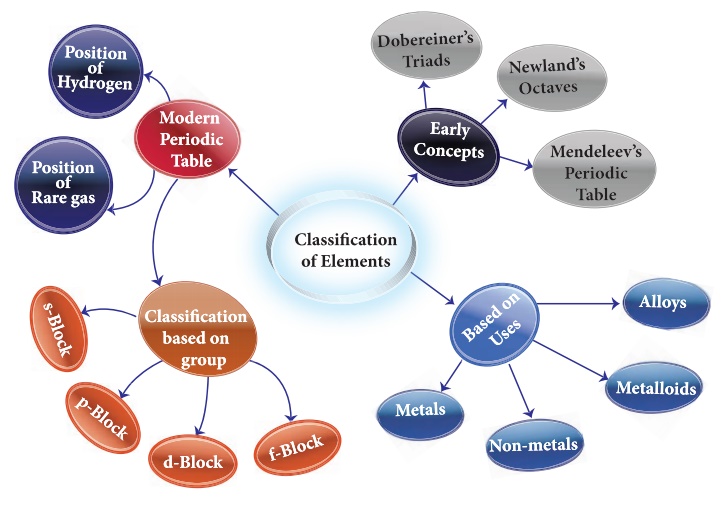
We live in the world of
substances with great diversity. The substances are formed by the combination
of various elements. All the elements are unique in their nature and property.
To categorize these elements according to their properties, scientists started
to look for a way. In 1800, there were only 31 known elements. By 1865, their
number became 63. Now 118 elements have been discovered. As different elements
were being discovered, scientists gathered more and more information about the
properties of these elements. They found it difficult to organize all that was
known about the elements. They started looking for some pattern in their
properties, on the basis of which they could study such a large number of
elements with ease. Let us discuss the concepts of classification of elements
proposed by various scientists from early to modern period.


Related Topics