Advantages, Composition and uses - Alloys | 9th Science : Periodic classification of Elements
Chapter: 9th Science : Periodic classification of Elements
Alloys
Alloys
During 3500 BC(BCE),
people used an alloy named ‘bronze’. The idea of making an alloy was quite old.
The majority of the metallic substances used today are alloys. Alloys are
mixtures of two or more metals and are formed by mixing molten metals
thoroughly. Rarely nonmetals are also mixed with metals to produce alloys.
It is generally found that
alloying produces a metallic substance that has more useful properties than the original pure metals from which it is
made. For example, the alloy brass is made from copper and zinc.
Advantages of alloys
·
Alloys do not get corroded or get corroded to very less extent.
·
They are harder and stronger than pure metals (example: gold is
mixed with copper and it is harder than pure gold)
·
They have less conductance than pure metals (example: copper is good
conductor of heat and electricity whereas brass and bronze are not good
conductors)
·
Some alloys have lower melting point than pure metals (example:
solder is an alloy of lead and tin which has lower melting point than each of
the metals)
·
When metal is alloyed with mercury, it is called amalgam
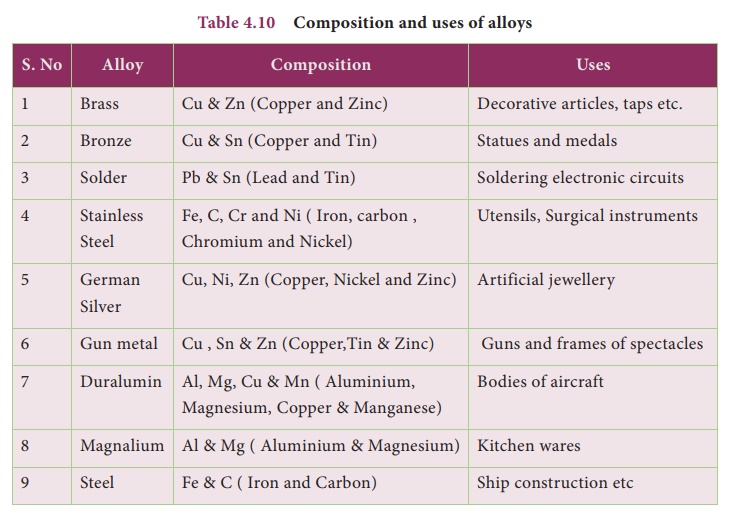
Composition and uses of alloys

Related Topics