Chapter: 9th Science : Periodic classification of Elements
Metals, Non-Metals and Metalloids
METALS, NON-METALS AND METALLOIDS
1. Metals
Metals are typically
hard, shiny, malleable (can be made as sheet), fusible and ductile (can be
drawn into wire) with good electrical and thermal conductivity. Except mercury,
most of the metals are solids at room temperature. Metals occupy larger area in
the periodic table and are categorized as:
(i) Alkali metals e.g.
Sodium and Potassium
(ii) Alkaline earth
metals e.g: Calcium and Magnesium
(iii) Transition Metals e.g: Iron and Nickel
(iv) Other Metals e.g: Aluminium and Tin
2. Non-metals
A non-metal is an element that does not have the characters of
hard, shiny, malleable, suitable and ductile. In other words, a non-metal is an
element that does not have the properties of metal. E.g. Oxygen, Nitrogen
3. Metalloids
Elements which have the
properties of both metals and non-metals are called as metalloids. (eg) Boron,
Arsenic.
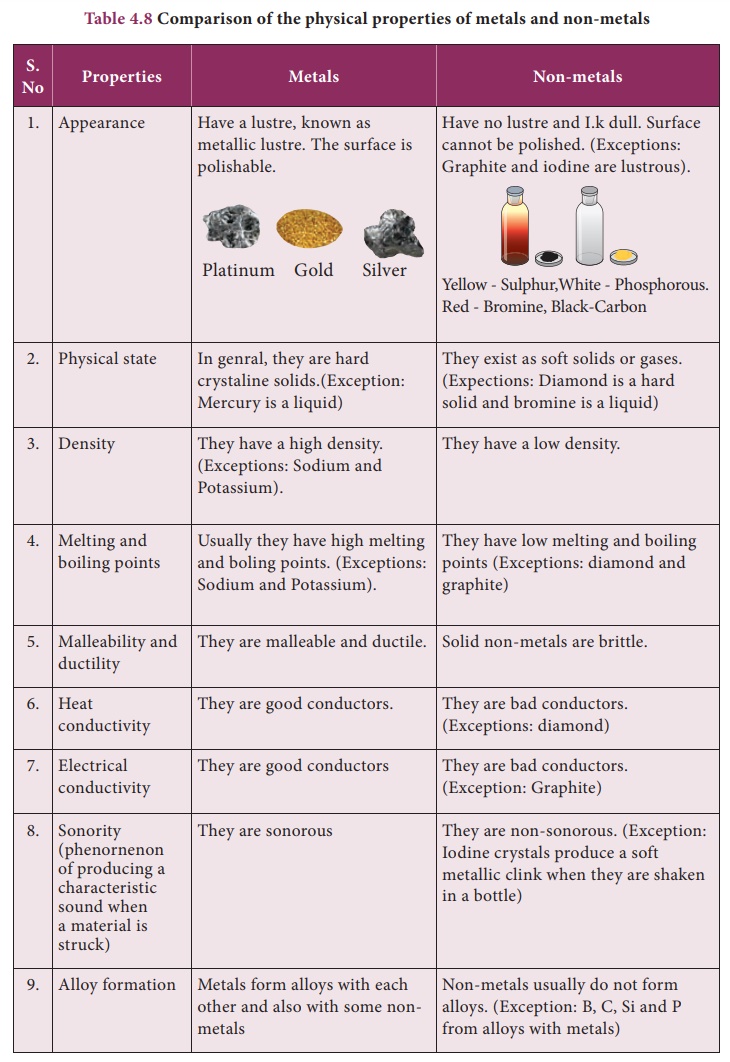
Comparison of the physical properties of metals and non-metals
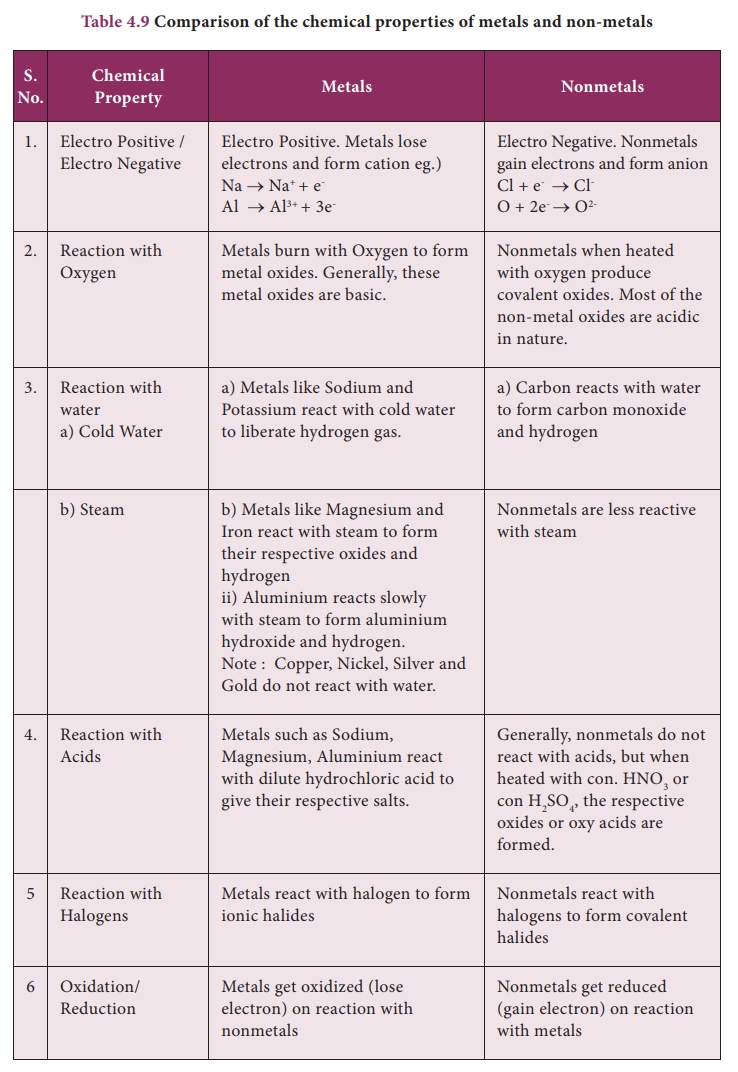
Comparison of the chemical properties of metals and non-metals
Related Topics