Chapter: Essentials of Psychiatry: Psychiatric Pathophysiology: Anxiety Disorders
Norepinephrine
Norepinephrine
Like serotonergic neurons, noradrenergic neurons
are anatom-ically restricted, with the majority (.70%) found in the nu-cleus locus
coeruleus. Noradrenergic neurons also possess an extensively ramified system of
efferent projections, diffusely innervating brain regions (e.g., cortex,
amygdala, hippocam-pus, hypothalamus) associated with anxiety disorders. From a
historical perspective, the evidence linking norepinephrine (and by
implication, the recognition sites subserving this trans-mitter) to anxiety
disorders clearly predates (Blaschko, 1972). The ability of stress to impact
brain norepinephrine levels has been known for almost 40 years (e.g., Barchas
and Freedman, 1963; Maynert and Levi, 1964). A wide range of stressors have
been reported to increase norepinephrine turnover in the CNS (Stone, 1975;
Tilson et al., 1975). This activation
of noradrenergic neurons by stress can be attenuated by benzodiazepines (Taylor
and Laverty, 1973; Ida et al., 1985).
The locus coeruleus sends projections to many
fore-brain areas associated with fear and anxiety via the dorsal noradrenergic
bundle. Redmond and his colleagues (Red-mond and Huang, 1979) provided perhaps
the most com-pelling preclinical data linking noradrenergic pathways to anxiety
disorders. Thus, it was demonstrated that electrical stimulation of the locus
coeruleus in monkeys produced fear-like behaviors, and that “symptom’’ severity
appeared depen-dent on the strength of the applied stimulus. Bilateral ablation
of the locus coeruleus blunted the behavioral responses of animals to
threatening environmental stimuli (Redmond and Huang, 1979). Pharmacological
studies in humans have also implicated noradrenergic pathways in some of the
anxiety disorders.
A variety of clinical and preclinical findings
indicate that physiological or pharmacological activation of noradrenergic
pathways mimics or exacerbates anxiety. At face value, these findings are at
odds with the reported efficacy of TCAs and MAOIs in anxiety disorders
(Hoehn-Saric, 2000; Buller and Jorga, 2000). In a broad sense, this body of
evidence parallels the apparent contradictory findings that increasing
serotonergic transmission is associated with anxiety, yet drugs whose primary
neurochemical action (e.g., SSRIs) results in increased synaptic concentrations
of serotonin are useful in the treatment of anxiety disorders.
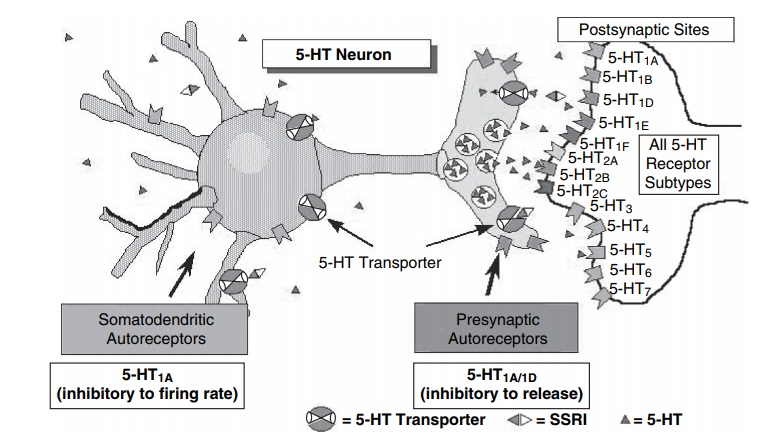
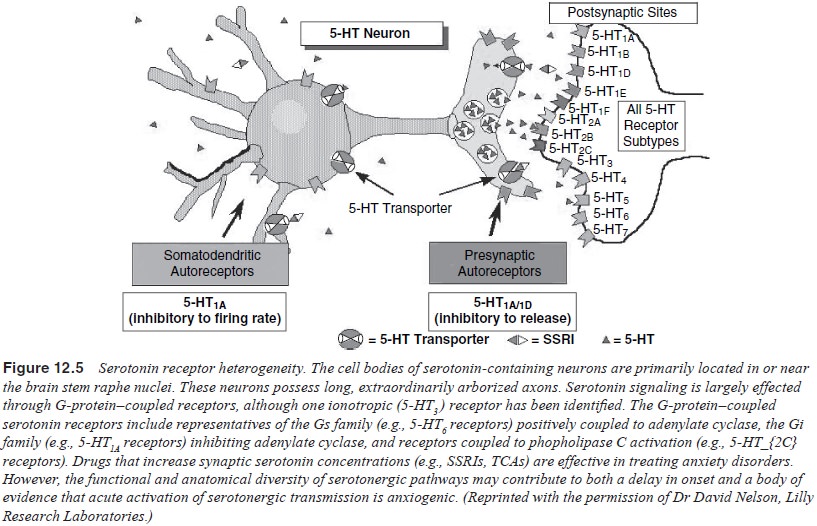
Future studies on potential targets downstream of
the monoaminergic synapse (e.g., Rossby and Sulser, 1997) (Figure 12.5) may
result in a better understanding of the antianxi-ety actions of these compounds
and the neurochemical substrates of anxiety.
Related Topics