Chapter: Medical Physiology: Contraction and Excitation of Smooth Muscle
Nervous and Hormonal Control of Smooth Muscle Contraction
Nervous and Hormonal Control of Smooth Muscle Contraction
Although skeletal muscle fibers are stimulated exclu-sively by the nervous system, smooth muscle can be stimulated to contract by multiple types of signals: by nervous signals, by hormonal stimulation, by stretch of the muscle, and in several other ways. The principal reason for the difference is that the smooth muscle membrane contains many types of receptor proteins that can initiate the contractile process. Still other receptor proteins inhibit smooth muscle contraction, which is another difference from skeletal muscle. Therefore, in this section, we discuss nervous control of smooth muscle contraction, followed by hormonal control and other means of control.
Neuromuscular Junctions of Smooth Muscle
Physiologic Anatomy of Smooth Muscle Neuromuscular Junc-tions. Neuromuscular junctions of the highly struc-tured type found on skeletal muscle fibers do not occur in smooth muscle. Instead, the autonomic nerve fibers that innervate smooth muscle generally branch
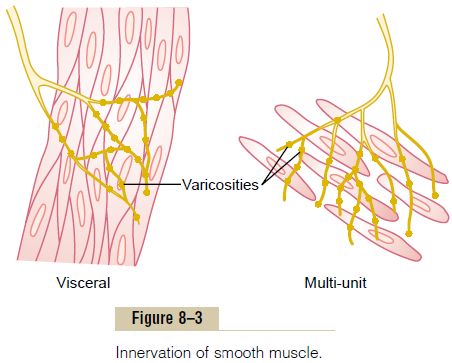
diffusely on top of a sheet of muscle fibers, as shown in Figure 8–3. In most instances, these fibers do not make direct contact with the smooth muscle fiber cell membranes but instead form so-called diffuse junc-tions that secrete their transmitter substance into thematrix coating of the smooth muscle often a few nanometers to a few micrometers away from the muscle cells; the transmitter substance then diffuses to the cells. Furthermore, where there are many layers of muscle cells, the nerve fibers often innervate only the outer layer, and muscle excitation travels from this outer layer to the inner layers by action potential con-duction in the muscle mass or by additional diffusion of the transmitter substance.
The axons that innervate smooth muscle fibers do not have typical branching end feet of the type in the motor end plate on skeletal muscle fibers. Instead, most of the fine terminal axons have multiple vari-cosities distributed along their axes. At these points the Schwann cells that envelop the axons are interruptedso that transmitter substance can be secreted through the walls of the varicosities. In the varicosities are vesi-cles similar to those in the skeletal muscle end plate that contain transmitter substance. But, in contrast to the vesicles of skeletal muscle junctions, which always contain acetylcholine, the vesicles of the autonomic nerve fiber endings contain acetylcholine in some fibers and norepinephrine in others—and occasionally other substances as well.
In a few instances, particularly in the multi-unit type of smooth muscle, the varicosities are separated from the muscle cell membrane by as little as 20 to 30 nanometers—the same width as the synaptic cleft that occurs in the skeletal muscle junction. These are called contact junctions, and they function in much the sameway as the skeletal muscle neuromuscular junction; the rapidity of contraction of these smooth muscle fibers is considerably faster than that of fibers stimulated by the diffuse junctions.
Excitatory and Inhibitory Transmitter Substances Secreted at the Smooth Muscle Neuromuscular Junction. The mostimportant transmitter substances secreted by the autonomic nerves innervating smooth muscle are acetylcholine and norepinephrine, but they are neversecreted by the same nerve fibers. Acetylcholine is an excitatory transmitter substance for smooth muscle fibers in some organs but an inhibitory transmitter for smooth muscle in other organs. When acetylcholine excites a muscle fiber, norepinephrine ordinarily inhibits it. Conversely, when acetylcholine inhibits a fiber, norepinephrine usually excites it.
But why these different responses? The answer is that both acetylcholine and norepinephrine excite or inhibit smooth muscle by first binding with areceptorprotein on the surface of the muscle cell membrane.Some of the receptor proteins are excitatory receptors, whereas others are inhibitory receptors. Thus, the type of receptor determines whether the smooth muscle is inhibited or excited and also determines which of the two transmitters, acetylcholine or norepinephrine, is effective in causing the excitation or inhibition.
Membrane Potentials and Action Potentials in Smooth Muscle
Membrane Potentials in Smooth Muscle. The quantitativevoltage of the membrane potential of smooth muscle depends on the momentary condition of the muscle. In the normal resting state, the intracellular potential is usually about -50 to -60 millivolts, which is about 30 millivolts less negative than in skeletal muscle.
Action Potentials in Unitary Smooth Muscle. Action poten-tials occur in unitary smooth muscle (such as visceral muscle) in the same way that they occur in skeletal muscle. They do not normally occur in many, if not most, multi-unit types of smooth muscle.
The action potentials of visceral smooth muscle occur in one of two forms: (1) spike potentials or (2) action potentials with plateaus.
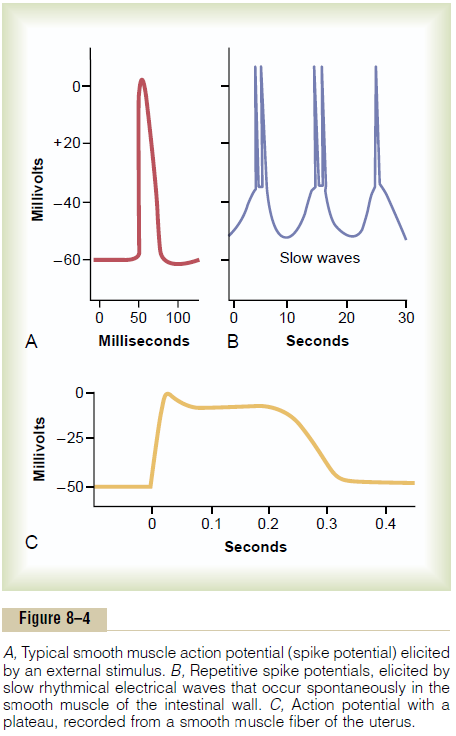
Spike Potentials. Typical spike action potentials, suchas those seen in skeletal muscle, occur in most types of unitary smooth muscle. The duration of this type of action potential is 10 to 50 milliseconds, as shown in Figure 8–4A. Such action potentials can be elicited in many ways, for example, by electrical stimulation, by the action of hormones on the smooth muscle, by the action of transmitter substances from nerve fibers, by stretch, or as a result of spontaneous generation in the muscle fiber itself, as discussed subsequently.
Action Potentials with Plateaus. Figure 8–4Cshows asmooth muscle action potential with a plateau. The onset of this action potential is similar to that of the typical spike potential. However, instead of rapid repolarization of the muscle fiber membrane, the repo-larization is delayed for several hundred to as much as 1000 milliseconds (1 second). The importance of the plateau is that it can account for the prolonged contraction that occurs in some types of smooth muscle, such as the ureter, the uterus under some con-ditions, and certain types of vascular smooth muscle. (Also, this is the type of action potential seen in cardiac muscle fibers that have a prolonged period of contraction.
Importance of Calcium Channels in Generating the Smooth Muscle Action Potential. The smooth musclecell membrane has far more voltage-gated calcium channels than does skeletal muscle but few voltage-gated sodium channels. Therefore, sodium participates little in the generation of the action potential in most smooth muscle. Instead, flow of calcium ions to the interior of the fiber is mainly responsible for the action potential. This occurs in the same self-regenerative way as occurs for the sodium channels in nerve fibers and in skeletal muscle fibers. However, the calcium channels open many times more slowly than do sodium channels, and they also remain open much longer. This accounts in large measure for the pro-longed plateau action potentials of some smooth muscle fibers.
Another important feature of calcium ion entry into the cells during the action potential is that the calcium ions act directly on the smooth muscle contractile mechanism to cause contraction. Thus, the calcium performs two tasks at once.
Slow Wave Potentials in Unitary Smooth Muscle, and Sponta-neous Generation of Action Potentials. Some smoothmuscle is self-excitatory. That is, action potentials arise within the smooth muscle cells themselves without an extrinsic stimulus. This often is associated with a basic slow wave rhythm of the membrane potential. A typical slow wave in a visceral smooth muscle of the gut is shown in Figure 8–4B. The slow wave itself is not the action potential. That is, it is not a self-regenerative process that spreads progressively over the membranes of the muscle fibers. Instead, it is a local property of the smooth muscle fibers that make up the muscle mass.
The cause of the slow wave rhythm is unknown. One suggestion is that the slow waves are caused by waxing and waning of the pumping of positive ions (presum-ably sodium ions) outward through the muscle fiber membrane; that is, the membrane potential becomes more negative when sodium is pumped rapidly and less negative when the sodium pump becomes less active. Another suggestion is that the conductances of the ion channels increase and decrease rhythmically.
The importance of the slow waves is that, when they are strong enough, they can initiate action potentials. The slow waves themselves cannot cause muscle con-traction, but when the peak of the negative slow wave potential inside the cell membrane rises in the positive direction from -60 to about -35 millivolts (the approximate threshold for eliciting action potentials in most visceral smooth muscle), an action potential develops and spreads over the muscle mass. Then con-traction does occur. Figure 8–4B demonstrates this effect, showing that at each peak of the slow wave, one or more action potentials occur. These repetitive sequences of action potentials elicit rhythmical con-traction of the smooth muscle mass. Therefore, the slow waves are called pacemaker waves.
Excitation of Visceral Smooth Muscle by Muscle Stretch.
When visceral (unitary) smooth muscle is stretched sufficiently, spontaneous action potentials usually are generated. They result from a combination of (1) the normal slow wave potentials and (2) decrease in overall negativity of the membrane potential caused by the stretch itself. This response to stretch allows the gut wall, when excessively stretched, to contract auto-matically and rhythmically. For instance, when the gut is overfilled by intestinal contents, local automatic con-tractions often set up peristaltic waves that move the contents away from the overfilled intestine, usually in the direction of the anus.
Depolarization of Multi-Unit Smooth Muscle Without Action Potentials
The smooth muscle fibers of multi-unit smooth muscle (such as the muscle of the iris of the eye or the piloerector muscle of each hair) normally contract mainly in response to nerve stimuli. The nerve endings secrete acetylcholine in the case of some multi-unit smooth muscles and norepinephrine in the case of others. In both instances, the transmitter substances cause depolarization of the smooth muscle membrane, and this in turn elicits contraction. Action potentials usually do not develop; the reason is that the fibers are too small to generate an action potential. (When action potentials are elicited in visceral unitary smoothmuscle, 30 to 40 smooth muscle fibers must depolarizesimultaneously before a self-propagating action poten-tial ensues.) Yet, in small smooth muscle cells, even without an action potential, the local depolarization (called the junctional potential) caused by the nerve transmitter substance itself spreads “electrotonically” over the entire fiber and is all that is needed to cause muscle contraction.
Effect of Local Tissue Factors and Hormones to Cause Smooth Muscle Contraction Without Action Potentials
Probably half of all smooth muscle contraction is initiated by stimulatory factors acting directly on the smooth muscle contractile machinery and without action potentials. Two types of non-nervous and non–action potential stimulating factors often involved are (1) local tissue chemical factors and (2) various hormones.
Smooth Muscle Contraction in Response to Local Tissue Chemical Factors. We discuss control of contraction of the arterioles, meta-arterioles, and pre-capillary sphincters. The smallest of these vessels have little or no nervous supply. Yet the smooth muscle is highly contractile, responding rapidly to changes in local chemical conditions in the surrounding intersti-tial fluid.
In the normal resting state, many of these small blood vessels remain contracted. But when extra blood flow to the tissue is needed, multiple factors can relax the vessel wall, thus allowing for increased flow. In this way, a powerful local feedback control system controls the blood flow to the local tissue area. Some of the specific control factors are as follows:
1. Lack of oxygen in the local tissues causes smooth muscle relaxation and, therefore, vasodilatation.
2. Excess carbon dioxide causes vasodilatation.
3. Increased hydrogen ion concentration causes vasodilatation.
Adenosine, lactic acid, increased potassium ions, diminished calcium ion concentration, and increased body temperature can all cause local vasodilatation.
Effects of Hormones on Smooth Muscle Contraction. Mostcirculating hormones in the blood affect smooth muscle contraction to some degree, and some have profound effects. Among the more important of these are norepinephrine, epinephrine, acetylcholine,angiotensin, endothelin, vasopressin, oxytocin, sero-tonin, and histamine.
A hormone causes contraction of a smooth muscle when the muscle cell membrane contains hormone-gated excitatory receptors for the respective hormone.Conversely, the hormone causes inhibition if the mem-brane contains inhibitory receptors for the hormone rather than excitatory receptors.
Mechanisms of Smooth Muscle Excitation or Inhibition by Hor-mones or Local Tissue Factors. Some hormone receptorsin the smooth muscle membrane open sodium or calcium ion channels and depolarize the membrane, the same as after nerve stimulation. Sometimes action potentials result, or action potentials that are already occurring may be enhanced. In other cases, depolari-zation occurs without action potentials, and this depo-larization allows calcium ion entry into the cell, which promotes the contraction.
Inhibition, in contrast, occurs when the hormone (or other tissue factor) closes the sodium and calciumchannels to prevent entry of these positive ions; inhi-bition also occurs if the normally closed potassiumchannels are opened, allowing positive potassium ionsto diffuse out of the cell. Both of these actions increase the degree of negativity inside the muscle cell, a state called hyperpolarization, which strongly inhibits muscle contraction.
Sometimes smooth muscle contraction or inhibition is initiated by hormones without directly causing any change in the membrane potential. In these instances, the hormone may activate a membrane receptor that does not open any ion channels but instead causes an internal change in the muscle fiber, such as release of calcium ions from the intracellular sarcoplasmic re-ticulum; the calcium then induces contraction. To inhibit contraction, other receptor mechanisms are known to activate the enzyme adenylate cyclase or guanylate cyclase in the cell membrane; the portions ofthe receptors that protrude to the interior of the cells are coupled to these enzymes, causing the formation of cyclic adenosine monophosphate (cAMP) or cyclicguanosine monophosphate (cGMP), so-called second messengers. The cAMP or cGMP has many effects, oneof which is to change the degree of phosphorylation of several enzymes that indirectly inhibit contraction. The pump that moves calcium ions from the sar-coplasm into the sarcoplasmic reticulum is activated, as well as the cell membrane pump that moves calcium ions out of the cell itself; these effects reduce the calcium ion concentration in the sarcoplasm, thereby inhibiting contraction.
Smooth muscles have considerable diversity in how they initiate contraction or relaxation in response to different hormones, neurotransmitters, and other substances. In some instances, the same substance may cause either relaxation or contraction of smooth muscles in different locations. For example, norepi-nephrine inhibits contraction of smooth muscle in the intestine but stimulates contraction of smooth muscle in blood vessels.
Source of Calcium Ions That Cause Contraction (1) Through the Cell Membrane and (2) from the Sarcoplasmic Reticulum
Although the contractile process in smooth muscle, as in skeletal muscle, is activated by calcium ions, the source of the calcium ions differs; the difference is that the sarcoplasmic reticulum, which provides virtually all the calcium ions for skeletal muscle con-traction, is only slightly developed in most smooth muscle. Instead, almost all the calcium ions that cause contraction enter the muscle cell from the extra-cellular fluid at the time of the action potential or other stimulus. That is, the concentration of calcium ions in the extracellular fluid is greater than 10-3 molar, in comparison with less than 10-7 molar inside the smooth muscle cell; this causes rapid diffusion of the calcium ions into the cell from the extracellular fluid when the calcium pores open. The time required for this diffusion to occur averages 200 to 300 mil-liseconds and is called the latent period before con-traction begins. This latent period is about 50 times as great for smooth muscle as for skeletal muscle contraction.
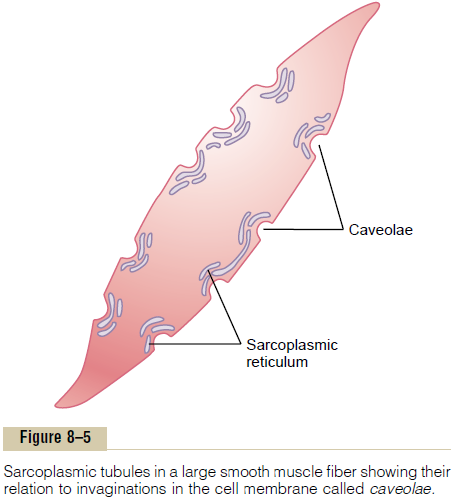
Role of the Smooth Muscle Sarcoplasmic Reticulum. Figure8–5 shows a few slightly developed sarcoplasmic tubules that lie near the cell membrane in some larger smooth muscle cells. Small invaginations of the cell membrane, called caveolae, abut the surfaces of these tubules. The caveolae suggest a rudimentary analog of the transverse tubule system of skeletal muscle. When an action potential is transmitted into the caveolae, this is believed to excite calcium ion release from the abutting sarcoplasmic tubules in the same way that action potentials in skeletal muscle transverse tubules cause release of calcium ions from the skeletal muscle longitudinal sarcoplasmic tubules. In general, the more extensive the sarcoplasmic re-ticulum in the smooth muscle fiber, the more rapidly it contracts.
Effect on Smooth Muscle Contraction Caused by Changing of Extracellular Calcium Ion Concentration. Although chang-ing the extracellular fluid calcium ion concentration from normal has little effect on the force of contrac-tion of skeletal muscle, this is not true for most smooth muscle. When the extracellular fluid calcium ion con-centration falls to about 1/3 to 1/10 normal, smooth muscle contraction usually ceases. Therefore, the force of contraction of smooth muscle usually is highly dependent on extracellular fluid calcium ion concentration.
A Calcium Pump Is Required to Cause Smooth Muscle Relaxation. To cause relaxation of smooth muscle afterit has contracted, the calcium ions must be removed from the intracellular fluids. This removal is achieved by a calcium pump that pumps calcium ions out of the smooth muscle fiber back into the extra-cellular fluid, or into a sarcoplasmic reticulum, if it is present.
This pump is slow-acting in comparison with the fast-acting sarcoplasmic reticulum pump in skeletal muscle. Therefore, a single smooth muscle contraction often lasts for seconds rather than hun-dredths to tenths of a second, as occurs for skeletal muscle.
Related Topics