Chapter: Medical Physiology: Propulsion and Mixing of Food in the Alimentary Tract
Movements of the Small Intestine
Movements of the Small Intestine
The movements of the small intestine, like those else-where in the gastrointestinal tract, can be divided into mixing contractions and propulsive contractions. To agreat extent, this separation is artificial because essen-tially all movements of the small intestine cause at least some degree of both mixing and propulsion. The usual classification of these processes is the following.
Mixing Contractions (Segmentation Contractions)
When a portion of the small intestine becomes dis-tended with chyme, stretching of the intestinal wall elicits localized concentric contractions spaced at intervals along the intestine and lasting a fraction of a minute.
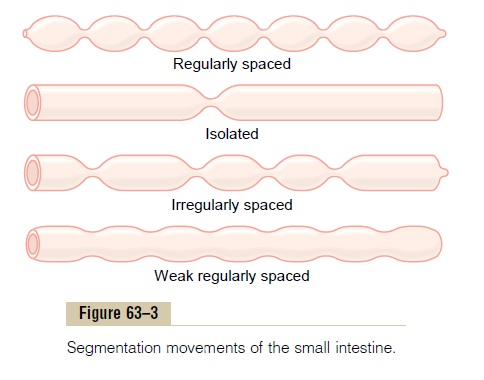
The contractions cause “segmentation” of the small intestine, as shown in Figure 63–3. That is, they divide the intestine into spaced segments that have the appearance of a chain of sausages. As one set of seg-mentation contractions relaxes, a new set often begins, but the contractions this time occur mainly at new points between the previous contractions. Therefore, the segmentation contractions “chop” the chyme two to three times per minute, in this way promoting pro-gressive mixing of the food with secretions of the small intestine.
The maximum frequency of the segmentation con-tractions in the small intestine is determined by the frequency of electrical slow waves in the intestinal wall, which is the basic electrical rhythm described. Because this frequency normally is not over 12 per minute in the duodenum and proximal jejunum, the maximum frequency of the segmentation contractions in these areas is also about 12 per minute, but this occurs only under extreme conditions of stim-ulation. In the terminal ileum, the maximum frequency is usually 8 to 9 contractions per minute.
The segmentation contractions become exceedingly weak when the excitatory activity of the enteric nervous system is blocked by the drug atropine. There-fore, even though it is the slow waves in the smooth muscle itself that cause the segmentation contractions, these contractions are not effective without back-ground excitation mainly from the myenteric nerve plexus.
Propulsive Movements
Peristalsis in the Small Intestine. Chyme is propelledthrough the small intestine by peristaltic waves. These can occur in any part of the small intestine, and they move toward the anus at a velocity of 0.5 to 2.0 cm/sec, faster in the proximal intestine and slower in the ter-minal intestine. They normally are very weak and usually die out after traveling only 3 to 5 centimeters, very rarely farther than 10 centimeters, so that forward movement of the chyme is very slow, so slow in fact that net movement along the small intestine normally averages only 1 cm/min. This means that 3 to 5 hours are required for passage of chyme from the pylorus to the ileocecal valve.
Control of Peristalsis by Nervous and Hormonal Signals. Peri-staltic activity of the small intestine is greatly increased after a meal. This is caused partly by the beginning entry of chyme into the duodenum causing stretch of the duodenal wall, but also by a so-called gastroentericreflex that is initiated by distention of the stomach andconducted principally through the myenteric plexus from the stomach down along the wall of the small intestine.
In addition to the nervous signals that may affect small intestinal peristalsis, several hormonal factors also affect peristalsis. They include gastrin, CCK, insulin, motilin, and serotonin, all of which enhanceintestinal motility and are secreted during various phases of food processing. Conversely, secretin and glucagon inhibit small intestinal motility. The physio-logic importance of each of these hormonal factors for controlling motility is still questionable.
The function of the peristaltic waves in the small intestine is not only to cause progression of chyme toward the ileocecal valve but also to spread out the chyme along the intestinal mucosa. As the chyme enters the intestines from the stomach and elicits peri-stalsis, this immediately spreads the chyme along the intestine; and this process intensifies as additional chyme enters the duodenum. On reaching the ileoce-cal valve, the chyme is sometimes blocked for several hours until the person eats another meal; at that time, a gastroileal reflex intensifies peristalsis in the ileum and forces the remaining chyme through the ileocecal valve into the cecum of the large intestine.
Propulsive Effect of the Segmentation Movements. The seg-mentation movements, although lasting for only a few seconds at a time, often also travel 1 centimeter or so in the anal direction and during that time help propel the food down the intestine. The difference between the segmentation and the peristaltic movements is not as great as might be implied by their separation into these two classifications.
Peristaltic Rush. Although peristalsis in the smallintestine is normally weak, intense irritation of the intestinal mucosa, as occurs in some severe cases of infectious diarrhea, can cause both powerful and rapid peristalsis, called the peristaltic rush. This is initiated partly by nervous reflexes that involve the autonomic nervous system and brain stem and partly by intrinsic enhancement of the myenteric plexus reflexes within the gut wall itself.The powerful peristaltic contractions travel long distances in the small intestine within minutes, sweeping the contents of the intestine into the colon and thereby relieving the small intestine of irri-tative chyme and excessive distention.
Movements Caused by the Muscularis Mucosae and Muscle Fibers of the Villi. Themuscularis mucosaecan cause shortfolds to appear in the intestinal mucosa. In addition, individual fibers from this muscle extend into the intes-tinal villi and cause them to contract intermittently. The mucosal folds increase the surface area exposed to the chyme, thereby increasing absorption. Also, contrac-tions of the villi—shortening, elongating, and shorten-ing again—“milk” the villi, so that lymph flows freely from the central lacteals of the villi into the lymphatic system. These mucosal and villous contractions are ini-tiated mainly by local nervous reflexes in the submu-cosal nerve plexus that occur in response to chyme in the small intestine.
Function of the Ileocecal Valve
A principal function of the ileocecal valve is to prevent backflow of fecal contents from the colon into the small intestine. As shown in Figure 63–4, the ileocecal valve itself protrudes into the lumen of the cecum and therefore is forcefully closed when excess pressure builds up in the cecum and tries to push cecal contents backward against the valve lips. The valve usually can resist reverse pressure of at least 50 to 60 centimeters of water.
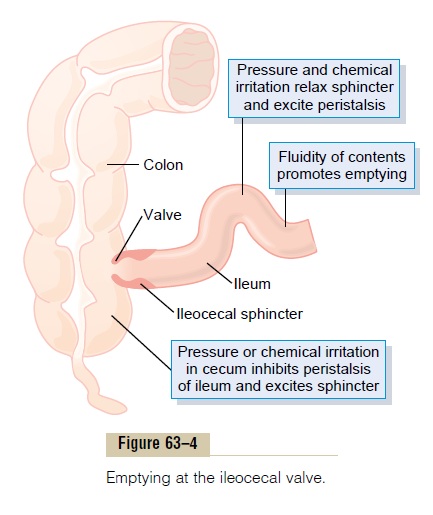
In addition, the wall of the ileum for several cen-timeters immediately upstream from the ileocecal valve has a thickened circular muscle called the ileo-cecal sphincter. This sphincter normally remains mildlyconstricted and slows emptying of ileal contents into the cecum. However, immediately after a meal, a gas-troileal reflex (described earlier) intensifies peristalsis in the ileum, and emptying of ileal contents into the cecum proceeds.
Resistance to emptying at the ileocecal valve pro-longs the stay of chyme in the ileum and thereby facilitates absorption. Normally, only 1500 to 2000 mil-liliters of chyme empty into the cecum each day.
Feedback Control of the Ileocecal Sphincter. The degree ofcontraction of the ileocecal sphincter and the intensity of peristalsis in the terminal ileum are controlled sig-nificantly by reflexes from the cecum. When the cecum is distended, contraction of the ileocecal sphincter becomes intensified and ileal peristalsis is inhibited, both of which greatly delay emptying of additional chyme into the cecum from the ileum. Also, any irri-tant in the cecum delays emptying. For instance, when a person has an inflamed appendix, the irritation of this vestigial remnant of the cecum can cause such intense spasm of the ileocecal sphincter and partial paralysis of the ileum that these effects together block emptying of the ileum into the cecum. The reflexes from the cecum to the ileocecal sphincter and ileum are mediated both by way of the myenteric plexus in the gut wall itself and of the extrinsic autonomic nerves, especially by way of the prevertebral sympa-thetic ganglia.
Related Topics