Chemistry - Metallurgy: Summary | 12th Chemistry : UNIT 1 : Metallurgy
Chapter: 12th Chemistry : UNIT 1 : Metallurgy
Metallurgy: Summary
Summary
· Metallurgy relates to the science and technology of metals.
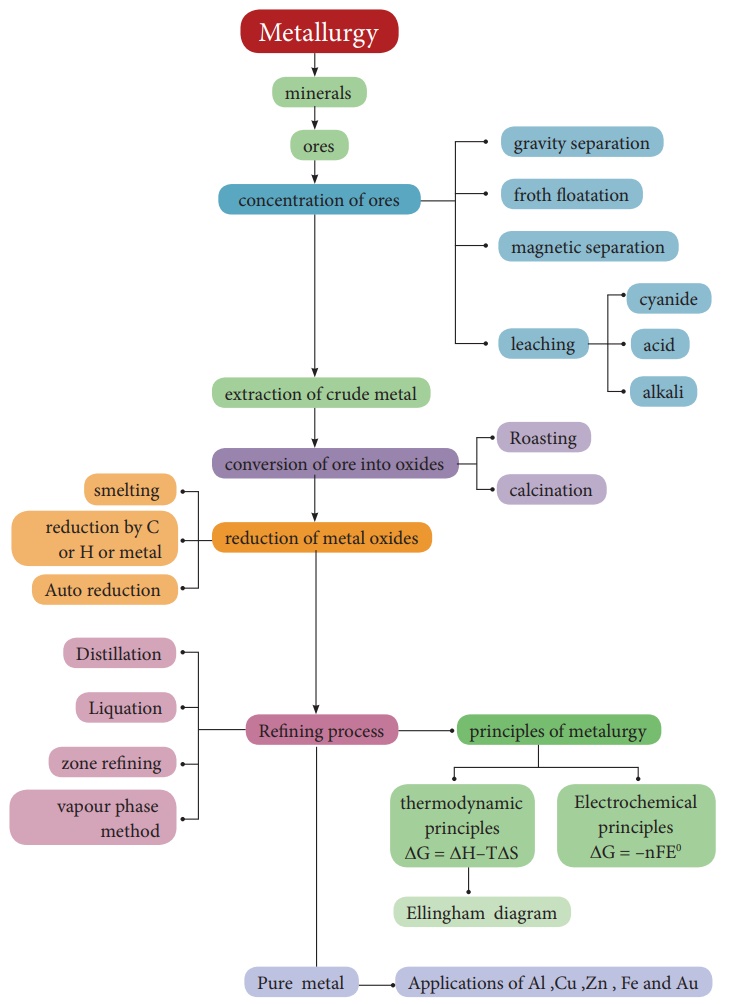
· A naturally occurring substance obtained by mining which contains the metal in free state or in the form of compounds like oxides, sulphides etc... is called a mineral.
· minerals that contains a high percentage of metal, from which it can be extracted conveniently and economically are called ores.
· The extraction of a metal of interest from its ore consists of the following metallurgical processes.
(i) concentration of the ore
(ii) extraction of crude metal
(ii) refining of crude metal
· The extraction of crude metals from the concentrated ores is carried out in two steps namely, (i) conversion of the ore into oxides of the metal of interest and (ii) reduction of the metal oxides to elemental metals.
· The graphical representation of variation of the standard Gibbs free energy of reaction for the formation of various metal oxides with temperature is called Ellingham diagram
· Ellingham diagram helps us to select a suitable reducing agent and appropriate temperature range for reduction.
· Similar to thermodynamic principles, electrochemical principles also find applications in metallurgical process.
· If E0 is positive then the ΔG is negative and the reduction is spontaneous and hence a redox reaction is planned in such a way that the e.m.f of the net redox reaction is positive. When a more reactive metal is added to the solution containing the relatively less reactive metal ions, the more reactive metal will go into the solution.
· Generally the metal extracted from its ore contains some impurities such as unreacted oxide ore, other metals, nonmetals etc...Removal of such impurities associated with the isolated crude metal is called refining process.
Related Topics