Chapter: Modern Pharmacology with Clinical Applications: Hypocholesterolemic Drugs and Coronary Heart Disease
Management of Hyperlipidemias With Drugs
MANAGEMENT OF
HYPERLIPIDEMIAS WITH DRUGS
Drug Treatment of Polygenic and Familial Hypercholesterolemia
Statins
Mechanism of Action
The statin family of six
closely related hypocholes-terolemic drugs are all potent competitive
inhibitors of the enzyme 3-hydroxy-3-methylglutaryl coenzyme A re-ductase (HMG
CoA reductase), the rate-limiting en-zyme in cholesterol biosynthesis. The
liver is their target organ, and decreased hepatic cholesterol synthesis
ulti-mately leads to increased removal of LDL particles from the circulation.
As a consequence, all other hypocholes-terolemic drugs have been relegated to
secondary status.
Clinical trials with lovastatin (Mevacor), simvastatin (Zocor) and pravastatin (Pravachol) provided much of the evidence supporting the observation that lowering of blood cholesterol lowers the risk of CHD.
Reductions in CHD risk appear to be due to multiple
consequences of inhibiting the cholesterol synthesis pathway. Drug-induced
inhibition of hepatic cholesterol synthesis leads to lowering of liver
cholesterol concen-trations and feedback up-regulation at the gene level of
both HMG CoA reductase and the LDL receptor (mechanisms IV and VII in Fig.
23.2). As long as the statin is present at adequate concentration in the liver,
the extra HMG CoA reductase activity is not ex-pressed. However, the increased
hepatic LDL receptor protein results in increased rates of removal of LDL
particles from the circulation by the liver, lowering of blood LDL-cholesterol
levels, slowing of athero-sclerosis, and decreased risk of heart attack. An
overview of lipoprotein metabolism and the sites where drugs can influence
plasma lipoprotein levels is pro-vided in Figure 23.2.

The reduced risk of CHD achieved with the statins may also be due to drug actions independent of lower-ing blood cholesterol. Many important molecules be-sides cholesterol are generated by intermediates in the complex cholesterol synthesis pathway. These include the isoprenes geranylgeranyl and farnesyl, which are co-valently attached to some proteins (isoprenylation) and target them to membranes where they function.
The reported capacities of statins
to inhibit proliferation of ar- terial wall smooth muscle cells and to improve
en- dothelial cell functions may be due to inhibited protein isoprenylation in
these cells secondary to HMG CoA reductase inhibition
Clinical Uses
With the possible exception
of atorvastatin, the statins are used to lower LDL cholesterol in familial or
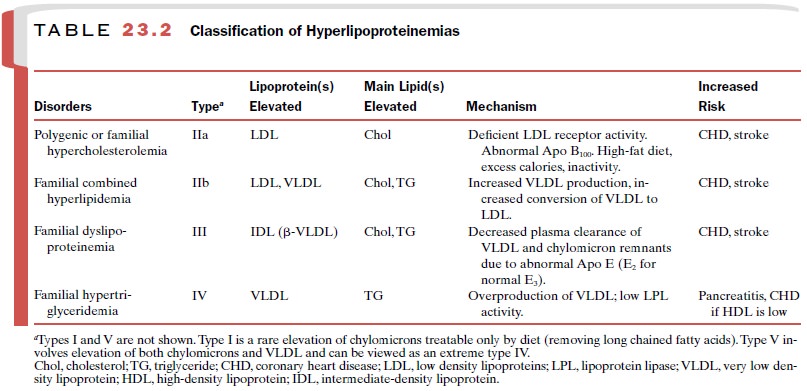
polygenic ( multifactorial) hypercholesterolemia (type IIa) and in combination
with triglyceride-lowering drugs to treat combined hyperlipidemia (type IIb)
when both LDL and VLDL (very low density lipoproteins) are elevated (Table
23.2). However, the statins probably should not be given with the fibrates
(triglyceride-lowering drugs, discussed later), since this combination may
greatly increase statin toxicity. Atorvastatin, the most potent of the
available statins (Fig. 23.3), has also been shown to lower blood triglycerides
significantly.
This effect may be due to
decreasing hepatic choles-terol and cholesterol ester levels to such an extent
that hepatic formation of VLDL is impaired. The statins also have been claimed
to reduce blood cholesterol levels modestly in some patients with homozygous
familial hy-percholesterolemia, a condition often fatal in childhood or in
early adulthood.
The statins may lower the
risk of CHD by decreasing inflammation, an important component of
atherogene-sis. Lovastatin decreased elevated plasma levels of C-reactive
protein, a marker for cellular inflammation, and acute coronary events in
patients with relatively low plasma cholesterol levels. Recent studies also
suggest that use of statins may decrease the risk of stroke, de-mentia, and
Alzheimer’s disease and may improve bonedensity in postmenopausal women. These
broad actions may be related to the hypocholesterolemic, antiprolifer-ative,
antiinflammatory, or antioxidant properties of the statins or some combination
of these properties.
Adverse Effects
The statins generally appear
to be well tolerated, with muscle pain and liver dysfunction seen in 1 to 2% of
patients. However, the consequences of 20 to 30 years of continuous use are
unknown. This fact has been dramatically reinforced by the recent recognition of
a potentially fatal consequence of statin use. A relatively common side effect
of the statins (perhaps 1% of pa-tients) is myositis, that is, inflammation of
skeletal mus-cle accompanied by pain, weakness, and high levels of serum
creatine kinase. Rhabdomyolysis, i.e., disintegra-tion of muscle with urinary
excretion of myoglobin and kidney damage, was considered to be a rare and
extreme toxic outcome. However, cerivastatin (Baycol) has now been withdrawn from the market by its manufacturer
(Bayer) because of 31 deaths linked to fatal rhabdomy-olysis. The risk of
muscle damage is said to increase with simultaneous use of the
triglyceride-lowering fibrates. Pravastatin may be less toxic than other
statins because it does not readily penetrate extrahepatic cells and may be
more confined to the liver after oral dosage.
Drug Interactions
Most of the statins
(lovastatin, simvastatin, atorvas-tatin, and cerivastatin) are metabolized by
the cytochro-mal P450 3A4 system of intestines and liver to more wa-ter-soluble
metabolites that are excreted in both the bile and urine. Drugs that inhibit
P450 3A4, such as itra-conazole, cyclosporine, and erythromycin, can vastly
(10-fold) increase plasma statin levels and thus increase the risk of toxicity.
Unexpectedly, grapefruit juice can inhibit intestinal metabolism of the statins
and can result in an 8- to 10-fold increase in simvastatin serum levels. Since
fluvastatin is metabolized by cytochrome P450 2C9, which is also responsible
for metabolism of warfarin, warfarin toxicity may be increased if these drugs
are si-multaneously given. Grapefruit juice should obviously not be consumed
within several hours of statin adminis-tration. Drugs that induce the P450 3A4
system, such as barbiturates, can accelerate statin metabolism and sup-press
statin blood levels.
Related Topics