Chapter: Obstetrics and Gynecology: Ovarian and Adnexal Disease
Malignant Ovarian Neoplasms
MALIGNANT OVARIAN NEOPLASMS
Ovarian
cancer is the fifth most common of all cancers in women in the United States
and the most common cause of gynecologic cancer. The mortality rate of this
disease is the highest of all the gynecologic malignancies, primarily
because of the difficultyin detecting the disease before widespread
dissemination. Of the estimated 25,000 new cases of ovarian cancer yearly,
approximately 60% will die within 5 years. Sixty-five to seventy percent are
diagnosed at an advanced state when the 5-year survival rate is 20–30%.
Risk Factors and Early Symptoms
Ovarian
cancer presents most commonly in the fifth and sixth decades of life. The
incidence of ovarian cancer in WesternEuropean countries and in the United
States is higher, with a five- to sevenfold greater incidence than age-matched
populations in East Asia. Whites are 50% more likely to develop ovarian cancer
than blacks living in the United States.
Symptoms of ovarian cancer are often
confused with benign conditions or interpreted as part of the aging process,
with the final diagnosis often delayed.
The most
common symptoms in order from highest percent-age to lowest are abdominal
fullness or distension, abdomi-nal or back pain, decreased energy or lethargy,
and urinary frequency.
Because no clinically applicable
screening test is avail-able, approximately two-thirds of patients with ovarian
cancer have advanced disease at the time of diagnosis.
The risk
of a woman developing ovarian cancer during her lifetime is approximately 1 in
70. The risk increaseswith age until approximately age
70. In addition to age, the epidemiologic factors associated with development
of ovarian cancer include nulliparity, primary infertility, and endometriosis.
Approximately 8% to 13% of cases of ovarian cancer are caused by inherited
mutations in the cancer-susceptibility genes BRCA-1 and BRCA-2.
Addition-ally, women affected with hereditary nonpolyposis colo-rectal cancer
(HNPCC; formerly called Lynch syndrome) have approximately a 13-fold greater
risk of developing ovarian cancer than the general population.
Long-term
suppression of ovulation may protect against the development of ovarian cancer,
at least for epithelial celltumors. It has been
suggested that so-called incessant ovu-lation may predispose to neoplastic
transformation of the epithelial cell surfaces of the ovary. Oral
contraceptives that prevent ovulation appear to provide significant protec-tion
against the occurrence of ovarian cancer. Five years’ cumulative use of oral
contraceptives decreases the lifetime risk by one-half. No evidence exists to
implicate the use of postmenopausal hormone replacement therapy in the
development of ovarian cancer.
Pathogenesis and Diagnosis
Malignant
ovarian epithelial cell tumors spread primarily by direct extension within the
peritoneal cavity as a result of direct cell sloughing from the ovarian
surface. This processexplains widespread peritoneal
dissemination at the time of diagnosis, even with relatively small primary
ovarian lesions. Although epithelial cell ovarian cancers also spread by
lymphatic and blood-borne routes, their direct extension into the virtually
unlimited space of the peri-toneal cavity is the primary basis for their late
clinical presentation.
Currently, it appears that the
best way to detect early ovarian cancer is for both the patient and her
clinician to be aware of early warning signs (Box 46.3). These signs should not
be ignored in postmenopausal women (median age, approximately 60 years).

Early Warning Signs of Ovarian Cancer
Increase in abdominal size
Abdominal bloating
Fatigue
Abdominal pain
Indigestion
Inability to eat normally
Urinary frequency
Constipation
Back pain
Urinary incontinence of recent onset
Unexplained weight loss
The early diagnosis of ovarian cancer is made even more difficult by the lack of effective screening tests. CA-125 shouldnot be routinely used to screen for ovarian cancer, but, instead, should be used to follow response to therapy and evaluate for recurrent disease. CA-125 can also be used to evaluate the presence of ovarian cancer in selected cases:
·
In premenopausal women with
symptoms, a CA-125 measurement has not been shown to be useful in most
circumstances, because elevated levels of CA-125 are associated with a variety
of common benign conditions, including uterine leiomyomata, pelvic inflammatory
disease, endometriosis, adenomyosis, pregnancy, and even menstruation.
·
In postmenopausal women with a
pelvic mass, a CA-125 measurement may be helpful in predicting a higher
likelihood of a malignant tumor than a benign tumor. However, a normal CA-125
measurement alone does not rule out ovarian cancer, because up to 50% of
early-stage cancers and 20% to 25% of advanced cancers are associated with
normal values.
HISTOLOGIC CLASSIFICATION
The cell
type of origin, similar to their benign counterparts, usu-ally categorizes
malignant ovarian neoplasms: malignantepithelial
cell tumors, which are the most common type;malignant germ cell tumors; and malignant stromal celltumors (see Box 46.1). Most
malignant ovarian tumorshave histologically similar but benign counterparts.
The relationship between a benign ovarian neoplasm and its malignant
counterpart is clinically important. If the benign counterpart is found in a
patient, removal of both ovaries is considered, because there is a possibility
of future malig-nant transformation in the remaining ovary. The decision
regarding removal of one or both ovaries, however, must be individualized based
on age, type of tumor, desire for future fertility, genetic predisposition for
malignancy, and the patient’s concern regarding future risks.

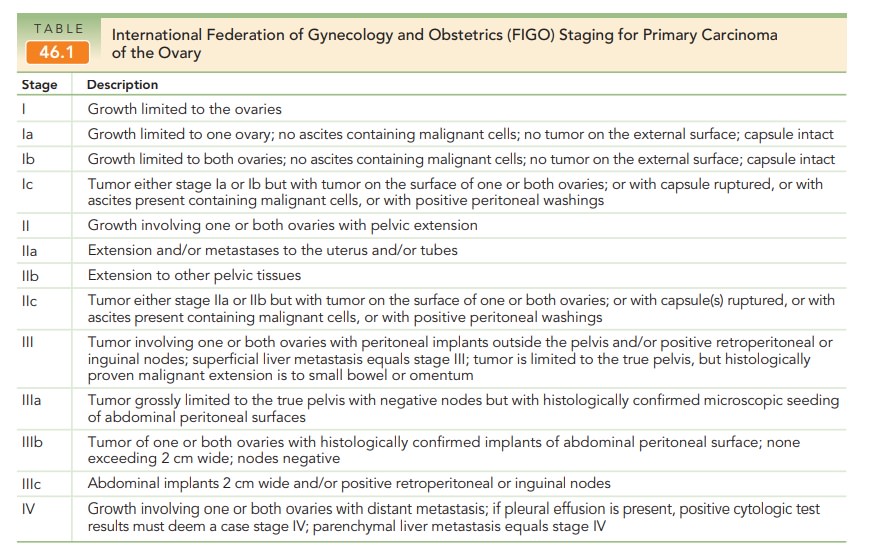
STAGING
The staging of ovarian carcinoma
is based on extent of spread of tumor and histologic evaluation of the tumor.
The International Federation of Gynecology and Obste-trics (FIGO)
classification of ovarian cancer is presented in Table 46.1.
Borderline Ovarian Tumors
Approximately
10% of seemingly benign epithelial cell tumors may contain histologic evidence
of intraepithelial neoplasia, com-monly referred to as borderline malignancies,
or “tumors of low malignant potential.” These
tumors generally remain con-fined to the ovary, are more common in
premenopausal women (30 to 50 years of age), and have good prognoses. About 20%
of such tumors show spread beyond the ovary. They require carefully
individualized therapy following the initial surgical resection of the primary
tumor. If frozen section pathology demonstrates borderline histology,
uni-lateral oophorectomy with a staging procedure and follow-up is appropriate,
assuming the woman wishes to retain ovarian function and/or fertility and
understands the risks of such conservative management.
Epithelial Cell Ovarian Carcinoma
Approximately
90% of all ovarian malignancies are of the epithelial cell type, derived from
mesothelial cells. The ovarycontains these cells as
part of an ovarian capsule just over-lying the actual stroma of the ovary. When
these meso-thelial cell elements are situated over developing follicles, they
go through metaplastic transformation whenever ovulation occurs. Repeated
ovulation is, therefore, associ-ated with the histologic change in these cells
derived from coelomic epithelium.
Malignant
epithelial serous tumors (serous cyst-adenocarcinoma) are
the most common malignant epithe-lial cell tumors. Approximately 50% of these
cancers are thought to be derived from their benign precursors (serous
cystadenoma), and as many as 30% of these tumors are bilateral at the time of
clinical presentation. They are typi-cally multiloculated and often have
external excrescences on an otherwise smooth capsular surface. Calcified,
laminated structures, psammoma bodies,
are found in more than one-half of serous carcinomas.
Another epithelial cell variant
that contains cells rem-iniscent of endocervical glandular mucous-secreting
cells is the malignant mucinous
epithelial tumor (mucinouscystadenocarcinoma). They make up
approximatelyone-third of all epithelial tumors, the majority of which are
benign or of low malignant potential; only 5% are can-cerous. These tumors have
a lower rate of bilaterality and can be among the largest of ovarian tumors,
often measur-ing more than 20 cm. They may be associated with wide-spread
peritoneal extension with thick, mucinous ascites, termed pseudomyxomatous peritonei.
Although most epithelial
carcinomas occur sporadi-cally, a small percentage (5% to 10%) occurs in
familial or hereditary patterns involving first- or second-degree
Having a first-degree relative (i.e., mother,
sister, daughter) with an epithelial carcinoma gives a 5% lifetime risk for
ovarian cancer, whereas having two first-degree relatives increases this risk
to 20% to 30%. Such hereditary ovarian cancers generally occur earlier than
nonhereditary tumors.
Women with breast/ovarian familial cancer syn-drome, a combination of
epithelial ovarian and breast can-cers in first- and second-degree family members,
are at two to three times the risk of these cancers as the general pop-ulation.
Women with this syndrome have an increased risk of bilaterality of breast
cancer and developing ovarian tumors at a younger age. This syndrome has been
associ-ated with the BRCA-1 gene.
Women with this gene muta-tion have a cumulative lifetime risk of 85% to 90%
for breast cancer and 50% for ovarian cancer. Women of Ashkenazi Jewish
ancestry have a 1% chance of carrying this gene, a 10-fold risk over the
general population.
HNPCC occurs in
families with first- and second-degree members with combinations of colon,
ovarian, endometrial, and breast cancers. Women in families with this syndrome
may have a threefold increased risk of cancer over the general population.
Women in families with these syndromes should have more frequent screening
tests and may benefit from risk-reducing salpingo-oophorectomy.
ENDOMETRIOID TUMORS
Most endometrioid tumors are malignant. These tumors contain histologic
features similar to those of endometrial carcinoma, and are commonly found in
association with endometriosis or are coincident with endometrial cancer of the
uterus.
OTHER EPITHELIAL CELL OVARIAN CARCINOMAS
Of the remaining epithelial cell
carcinomas of the ovary, clear cell carcinomas are thought to arise from
mesonephric elements, and Brenner tumors are thought to arise rarely from their
benign counterpart. Brenner tumors may occur in the same ovary that contains
mucinous cystadenoma; the reason for this is unclear.
Germ Cell Tumors
Germ cell tumors are the most common ovarian
cancers inwomen younger than 20 years of age. Germ cell
tumors may befunctional, producing hCG or α-fetoprotein (AFP), both of which
can be used as tumor markers. The most common germ cell malignancies are dysgerminoma
and immatureteratoma. Other tumors are recognized as mixed germ cell tumors,
endodermal sinus tumors, and embryonal tumors. Improved chemotherapeutic and
radiation protocols have resulted in greatly improved 5-year survival rates.
Dysgerminomas are usually unilateral and are
the mostcommon type of germ cell tumor seen in patients with gonadal dysgenesis
(Fig. 46.5). These tumors often arise in
benigncounterparts, called the gonadoblastoma. The tumors are particularly
radiosensitive and chemosensitive.
Because of the young age of
patients with dysgermino-mas, removing only the involved ovary while preserving
the uterus and contralateral tube and ovary may be considered if the tumor is
less than approximately 10 cm and if no evi-dence of extraovarian spread is
found. Unlike the epithelial cell tumors, these malignancies are more likely to
spread by lymphatic channels, and therefore the pelvic and periaortic lymph
nodes must be sampled at the time of surgery. If dis-ease has spread outside
the ovaries, conventional hysterec-tomy and bilateral salpingo-oophorectomy are
necessary, usually followed by cisplatin-based chemotherapy that is used in
combination with bleomycin and etoposide. The prognosis of these tumors is
generally excellent. The over-all 5-year survival rate for patients with
dysgerminoma is 90% to 95% when the disease is limited to one ovary.
Immature teratomas are the malignant counterpart ofbenign cystic teratomas (dermoids). These tumors are the sec-ond most common germ cell cancer and are most often found in women younger than 25 years of age. They are usually uni-lateral, although on occasion a benign counterpart may be found in the contralateral ovary. Because these tumors are rapidly growing, they may produce painful symptomatology relatively early, due to hemorrhage and necrosis. The dis-ease is therefore diagnosed when it is limited to one ovary in two-thirds of patients. As with dysgerminoma, if an imma-ture teratoma is limited to one ovary, unilateral oophorec-tomy is sufficient. Treatment with chemotherapeutic agents provides a good prognosis.
Rare Germ Cell Tumors
Endodermal
sinus tumors and
embryonal cell carcino-mas are uncommon malignant ovarian tumors that havehad
a remarkable improvement in cure rate. Until about 10 years ago, these tumors
were almost uniformly fatal. New chemotherapeutic protocols have resulted in an
over-all 5-year survival rate of more than 60%. These tumors typically occur in
childhood and adolescence, with the pri-mary treatment being surgical resection
of the involved ovary followed by combination chemotherapy. The endo-dermal
sinus tumor produces AFP, whereas the embryonal cell carcinoma produces both
AFP and β-hCG.
Gonadal Stromal Cell Tumors
The gonadal stromal cell tumors
make up an unusual group of tumors characterized by hormone production; hence,
these tumors are called functioning
tumors. The hormonal output from these tumors is usually in the form of
female or male sex steroids, or on occasion, adrenal steroid hormones.
The granulosa cell tumor is the most common in this group. These tumors
occur in all ages, although in older patients they are more likely to be
benign. Granulosa celltumors may secrete
large amounts of estrogen, which in some older women may cause endometrial
hyperplasia or endometrial carci-noma. Thus, endometrial sampling is
especially importantwhen ovarian tumors such as the granulosa tumor are
estrogen-producing. Surgical treatment should include removal of the uterus and
both ovaries in postmenopausal women as well as in women of reproductive age
who no longer wish to remain fertile. In a young woman with the lesion limited
to one ovary with an intact capsule, unilat-eral oophorectomy with careful
surgical staging may be adequate. This tumor may demonstrate recurrences up to
10 years later. This is especially true with large tumors, which have a 20% to
30% chance of late recurrence.
Sertoli–Leydig
cell tumors (arrhenoblastoma) are the rare, testosterone-secreting counterparts
to granulosa cell tumors. They usually occur in older
patients and should be sus-pected in the differential diagnosis of
perimenopausal or postmenopausal patients with hirsutism or virilization and an
adnexal mass. Treatment of these tumors is similar to that for other ovarian
malignancies in this age group, and is based on extirpation of uterus and
ovaries.
Other stromal cell tumors include
fibromas and thecomas, which rarely demonstrate malignant potential,and their
malignant counterparts, the fibrosarcoma
and malignant thecoma.
Other Ovarian Cancers
Rarely, the ovary may be the site
of initial manifestation of lymphoma. These tumors are usually found in
associ-ation with lymphoma elsewhere, although cases have been reported of
primary ovarian lymphoma. Once diag-nosed, their management is similar to that
for lymphoma of other origin.
Malignant
mesodermal sarcomas (carcinosarcomas)are another rare
type of ovarian tumor that usually show aggressive behavior and are diagnosed
at late stages. The survival rate is poor, and clinical experience with these
tumors is limited.
Cancer Metastatic to the Ovary
Classically, the term Krukenberg tumor describes an ovarian
tumor that is metastatic from other sites such as the gastrointestinal tract and
breast. Between 5% and 10% of women thought to have a primary ovarian
malignancy ultimately will receive the diagnosis of a nongenital tract
malignancy. Most of these tumors are characterized as infiltrative, mucinous
carcinoma of predominantly signet-ring cell type and as bilateral and
associated with wide-spread metastatic disease. On occasion, these tumors are
associated with abnormal uterine bleeding or virilization, leading to the
supposition that some may produce estro-gens or androgens. Breast cancer
metastatic to the ovary is common, with autopsy data suggesting ovarian
metastasis in one quarter of cases.
In approximately 10% of patients
with cancer metasta-tic to the ovary, an extraovarian primary site cannot be
demonstrated. In this regard, it is important to consider ovarian preservation
versus prophylactic oophorectomy at the time of hysterectomy in patients who
have a strong family history (first-degree relatives) of epithelial ovarian
cancer, primary gastrointestinal tract cancer, or breast can-cer. In patients
previously treated for breast or gastrointesti-nal cancer, consideration should
be given to the incidental removal of the ovaries at the time of hysterectomy,
because these patients have a high predilection for development of ovarian cancer.
The prognosis for most patients with carci-noma metastatic to the ovary is
generally poor.
Related Topics