Chapter: Modern Medical Toxicology: Corrosive(Caustic) Poisons: Alkalis and Other Caustics
Iodine - Corrosive(Caustic) Poisons
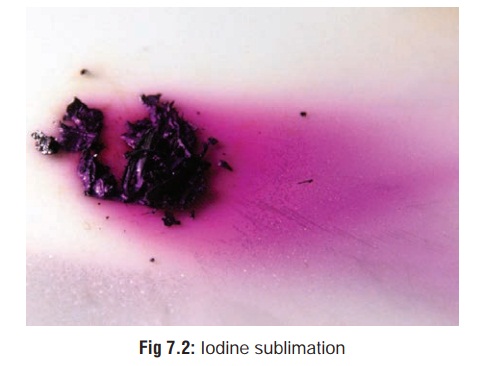
Iodine
Physical Appearance
Blue-black, glittering crystals
which constantly give off violet coloured vapour (a process called sublimation) (Fig 7.2) with a peculiar odour.
Uses
·
Antiseptic preparations:
Lugol’s iodine (5%
iodine with 10% potassium iodide in water).
Tincture of iodine (2.5%
iodine and 2.5% potassium iodide, or 2% iodine, 2.4% sodium iodide, 47%
alcohol, and water).
Povidone-iodine (5
to 10% iodophor, i.e. mixture of polyvinyl-pyrrolidone and iodine).*Iodex (iodine with methyl salicylate).
·
As a component of some expectorants
(e.g. potassium iodide, iodinated glycerol), anti-asthmatics (e.g. iophylline),
anti-arrhythmics (e.g. amiodarone), anti-amoebics (e.g. iodo-quinol),
antifungals (e.g. vioform), antithyroid drugs (e.g. potassium iodide), and
radiographic contrast agents (e.g. diatrizoate sodium, iopanoic acid, ipodate
sodium, etc.). As a solution for prepaing patients for thyroid surgery
(e.g.collosol liquid, which contains 8 mg iodine/5 ml).
·
Iodochlorhydroxyquinolone
(clioquinol), which is an iodi-nated hydroxyquinoline, is used sometimes in
amoebiasis. Iodine is used commonly to treat drinking water to make it potable.
Usual Fatal Dose
About
2 to 5 grams of free iodine, or 1 to 2 ounces of strong iodine tincture. The
presence of food in the stomach inactivates iodine by converting it to iodide
which is relatively innocuous.
Mode of Action
Locally,
strong iodine solution can be an intense irritant. Systemic toxicity is due to
combination of free iodine with serum sodium bicarbonate, leading to metabolic
acidosis.
Clinical Features
·
Initial manifestations of iodine poisoning include
rhinor-rhoea, conjunctivitis, and cough (especially if fumes have been
inhaled).
· There is burning pain extending from
the mouth to the abdomen, salivation, metallic taste, vomiting, and diarrhoea.
o
Vomitus and stools may appear yellowish (or sometimes
bluish) in colour. Blue coloured emesis indicates the presence of food (starch)
in the stomach and the conver-sion of iodine to iodide.
· Skin and mucous membranes are
stained yellowish brown.
·
Prolonged exposure to tincture of iodine can induce
super-ficial necrosis. Eye exposure can result in severe ocular burns.
· The following features have also
been reported: glottic oedema, pulmonary oedema, delirium, hallucinations,
convulsions, tachycardia, hypotension, metabolic acidosis, and renal failure.
· Hypersensitivity reactions including
angioedema and a serum sickness-like reaction may occur following oral,
topical, vaginal (douche), or IV administration of iodine.
· Iodides and iodophores (e.g.
povidone-iodine), are much less toxic and usually require only supportive
treament.
But
chronic poisoning can result from long-term thera-peutic intake of iodide salts
leading to iodism which is
characterised by metallic taste, anorexia, insomnia, lymphadenopathy, parotid
swelling (“iodide mumps”), stomatitis, pharyngitis, conjunctivitis,
rhinorrhoea, and skin manifestations (erythema, urticaria, acne, etc. together
referred to as “ioderma”).
· Hypothyroidism, hyperthyroidism, and
thyrotoxicosis have been reported secondary to iodine exposure.
· Iodine is a confirmed human
reproductive hazard.
a.Excess
iodine is harmful to the unborn, as shown in many cases of pregnant women
taking iodine-containing drugs. Several iodine-containing drugs have been
associated with foetal goitre, including ammonium iodide, potassium iodide, and
sodium iodide.
b.Iodine
deficiency is also harmful to the unborn. This has become evident in certain
areas of the world where either the low iodine content of the soil or the
practice of eating cassava as a major portion of the diet results in endemic
congenital cretinism. Iodine deficiency can cause congenital goitre, delayed
skeletal maturation, developmental delays, and perhaps a higher incidence of
birth defects.![]()
Diagnosis
1. Specific Tests :
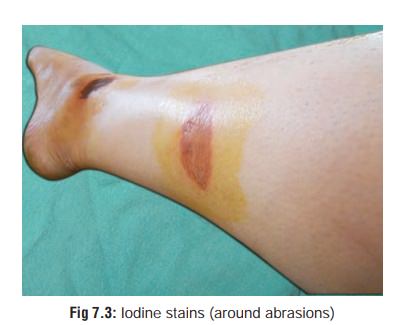
a. Iodine
Yellowish stains of clothing, skin (Fig 7.3), and mucosa.
Characteristic odour.
Vomitus or stomach contents when
heated in a beaker will cause the iodine to sublime on a cold surface (e.g. a
watch glass placed on top of the beaker with some ice).To 10 ml of vomitus or
gastric aspirate, add 1 ml of starch solution. A blue-black colour will develop.
Urine Test: Add 5 ml of chloroform
and a few drops of nitric acid to 10 ml of urine. Allow to stand for 3 minutes.
A pinkish-violet chloroform layer forms.
b. Iodides
To 10 ml of vomitus or gastric
aspirate, add 1 ml of 10% silver nitrate solution and 1 ml nitric acid.
Formation of a yellow precipitate
which is insoluble in ammonia is characteristic of iodides. Whitish precipitate
(soluble in ammonia) is characteristic of chlorides, while bromides produce
brown precipi-tate which is insoluble in ammonia.
2. Ancillary Tests:
Serum iodine levels are elevated, as
also aminotransferase. There is also hyperbilirubinaemia, neutropenia and
hypox-aemia.
Treatment
·
Decontamination:
o
Skin: Wash thoroughly with soap and water, or 20% alcohol.
o
Eyes: Irrigate with water for 15 minutes.
o
GIT: If oesophageal injury is not present or suspected,
gastric lavage can be attempted with starch solution, or 5% solution of sodium
thiosulfate, or even plain milk. It has been suggested that soluble starch be
administered, which forms a complex with iodine that is purple in colour and,
therefore, may aid in the removal of iodine from the stomach by gastric lavage
by making the iodine highly visible. Activated charcoal binds iodine, and can
be administered.
· Sodium bicarbonate IV for metabolic
acidosis.
· There is no specific antidote for
iodine or bound iodine.
·
Treatment is primarily supportive and includes monitoring
for the development of gastroenteritis, renal failure, tachy-cardia,
hypotension, and circulatory collapse. Anaphylactic type reactions may occur as
well.
· Osmotic diuresis, chloruretic
diuresis, and salt loading may enhance elimination.
·
Iodism is treated by ceasing iodide intake while enhancing
the intake of sodium chloride which promotes excretion of iodides. Chloride competes
with iodide at the level of the renal tubules.
Autopsy Features
·
Brownish or yellowish stains of skin
and mucosa. The mucosal staining in the GI tract will appear bluish if starch
solution had been administered while attempting to treat the patient.
·
Characteristic odour (rarely
perceptible).
·
Congestion of viscera, especially
the kidneys.
Forensic Issues
· Most cases result from accidental
therapeutic exposure where iodine or iodides had been administered in excess.
Deaths have been reported even with excessive local appli-cations of
povidone-iodine. The commonest abnormalities reported with such iodophore
applications (repeatedly) are acid-base disturbances and metabolic acidosis.
· Chronic poisoning can occur from
prolonged therapy with iodides, or may be a result of occupational exposure.
Iodinated radiologic contrast agents are well
known to produce anaphylactic reactions.
Related Topics