Chapter: Essential Anesthesia From Science to Practice : Clinical management : Monitoring
Invasive monitors - Anesthesia Clinical management
Invasive monitors
Arterial catheter
The ease
with which a small catheter can be inserted into an artery, usually the radial,
has caused many patients to be monitored with arterial catheters (often
Before inserting a catheter into a
radial (rarely the ulnar) artery, many clinicians like to check the patency of
the volar arterial arch that connects radial and ulnar arteries. In the
so-called Allen’s test, the hand is blanched, both arteries occluded by
external pressure, then one occluded artery is freed. If now the entire hand,
rather than the vascular bed of just one artery, turns pink, we accept the idea
that the volar arch is patent and should one artery become obstructed by a clot
or through damage to the intima, the other artery will prevent necrosis of
fingers.
For this
and all other invasive pressure measurements, we use saline or heparin-filled
non-compressible (pressure) tubing connected to a transducer, which con-verts
the pressure waveform into an electrical signal. We need to make sure that the
instrument is properly calibrated and that the zero level (open to air) is at
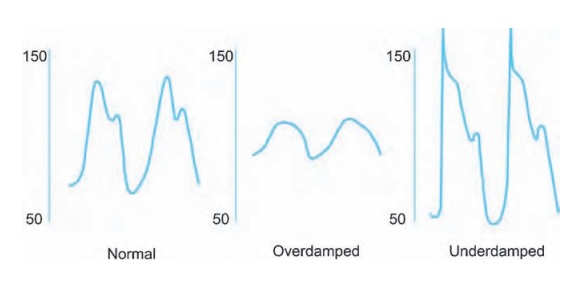
the level of the heart. Two problems can cause the system to report faulty
systolic and diastolic – but usually correct mean – pressures. When the signal
is damped, for example owing to an air bubble somewhere in the tubing, the
systolic pressure will read falsely low, and the diastolic pressure falsely
high. When the system is not damped enough, it might ring (like a bouncing
spring), now reporting falsely high systolic (and low diastolic) pressures (see
Fig. 7.7).
Arterial
catheters give ready access to arterial blood and thus to an analysis of blood
gases. When drawing arterial blood for analysis, be sure you are not diluting
the blood and that you have the analysis performed without delay so that the
normal metabolism of the cells does not affect the results.
Central venous catheter
Placement
of a central venous catheter offers not only the ability to determine the
central venous pressure but also an avenue for rapid infusions (see Vascular
access). Because no valve separates the vena cavae from the atrium, central
venous pressure (CVP) reflects right atrial pressure. Similarly, when the
tricus-pid valve is open, and pressure has equalized between the atrium and
ventricle
If
we assume a normal ventricular compliance (pressure–volume rela-tionship), we
now have an indication of the end-diastolic volume or preload. However, because
of its intrathoracic location, the central venous catheter also records
pressures in the thorax as a whole, and thus, CVP fluctuates with venti-lation.
In a spontaneously breathing patient, normal pressures might range from −2 to +6 cm H2O. If we then mechanically
ventilate that patient’s lungs, pressuresof +4 to +12 (or more with high peak inspiratory
pressures) can be expected – this without changing his intravascular volume
and, in fact, likely lowering his
preload as venous return is hampered by high intrathoracic pressure. The shape
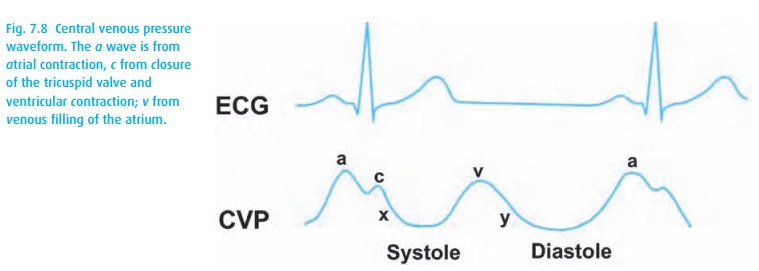
of the CVP waveform reflects the cardiac cycle (Fig. 7.8)
and may suggest conditions that limit the extrapolation of preload from CVP,
such as tricuspid valve disease or a poorly compliant ventricle. As with all
monitors, when interpreting CVP data we must consider the clinical scenario and
look more at trends in a given patient than the actual values.
Pulmonary artery catheter
Once the
catheter is properly positioned, best in an area where the balance between
blood flow and ventilation favors flow (below the level of the left atrium or
zone III according to West),3 the
cuff can be inflated, blocking the vessel so that the tip of the catheter no
longer senses PA pressure. Instead, it now looks down-stream and registers
pressures submitted retrograde from the left atrium. This pulmonary artery
occlusion or wedge pressure helps to identify situations affect-ing left
ventricular preload. However, as with the CVP, many factors can influence the
readings, e.g., mitral valve disease, pulmonary hypertension. Normal data
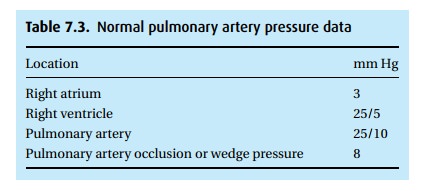
appear in Table 7.3.
A number
of refinements add utility to the PA catheter. For one, a thermistor at the tip
of the catheter can record the temperature of the blood flowing past. After the
injection of cold saline through a port situated in the vena cava, the observed
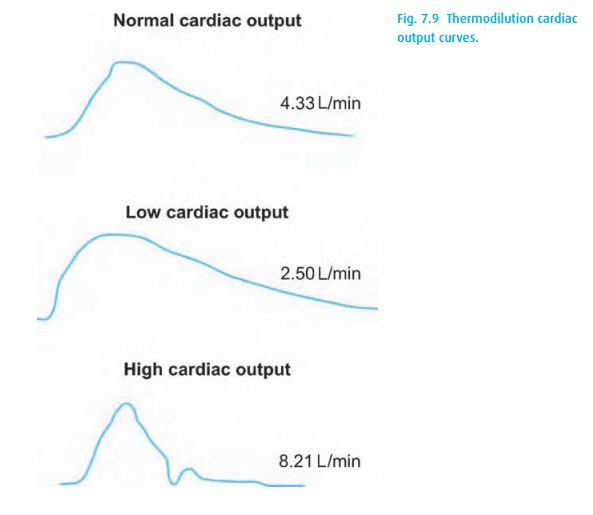
When the output is low, blood will flow slowly past the thermis-tor,
and a large thermodilution curve will result. Conversely, with a large cardiac
output, the thermodilution curve will be small (Fig. 7.9).
We can
also monitor the oxygen saturation of central venous blood either
intermittently by drawing samples for the laboratory, or continuously by
incor-porating an oximeter in the catheter. When oxygen content of arterial
blood and oxygen consumption are constant, a drop in venous oxygen saturation
indicates a decrease in tissue blood flow, i.e., cardiac output.
PA
catheters have come under much criticism because they may not reveal as much as
originally hoped for, and they are highly invasive and saddled with a
measurable rate of sometimes life-threatening complications including but not
limited to dysrhythmias, thrombosis, infection, and devastating pulmonary
artery rupture.4 Much less
invasively, transesophageal echocardiography offers a great advantage over the
PA catheter. PA catheters generate pressure and flow data; TEE shows volumes
and function of all four chambers and valves.
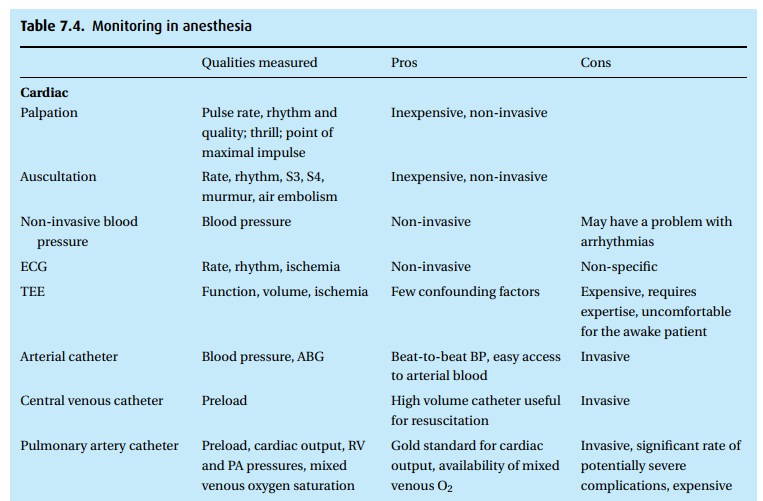
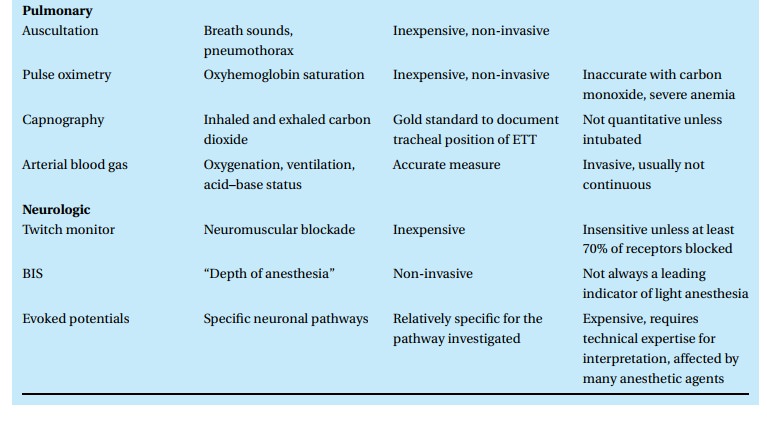
Thus we
have many monitors at our disposal (Table 7.4),
with new ones arriving regularly. Each has strengths, weaknesses, risks, and
potential benefits. No moni-tor is therapeutic in itself but requires the skill
and vigilance of a trained observer to interpret the information in the context
of the ever-changing clinical picture.
Related Topics