Chapter: Modern Pharmacology with Clinical Applications: Therapy of Human Immunodeficiency Virus
Human Immunodeficiency Virus
HUMAN
IMMUNODEFICIENCY VIRUS
Human immunodeficiency virus
(HIV) is a single-stranded RNA retrovirus that causes acquired
immun-odeficiency syndrome (AIDS), a condition in which in-dividuals are at
increased risk for developing certain infections and malignancies. The virus is
found in two major forms: HIV-1, the most prevalent worldwide, and HIV-2, the
most common in western Africa. More than 22 million people have died of HIV
infection, and 40 million are believed to be infected worldwide. AIDS epidemics
threaten populations in sub-Saharan Africa, Southeast Asia, Central and South
America, and Russia. In the United States about 450,000 deaths have occurred
and another 900,000 people are estimated to carry the virus. Although the
development of new drugs, complex multidrug regimens, and behavioral
modifica-tion have done much to combat the spread of HIV infection, AIDS
remains a serious threat because of the expense and inaccessibility of
antiretroviral agents in the developing countries in which the disease is most
prevalent. In addition, the effectiveness of antiretroviral drugs has been
diminished by the emergence of mul-tidrug-resistant virus.
Production of Immunodeficiency by HIV
HIV infects CD4+ T
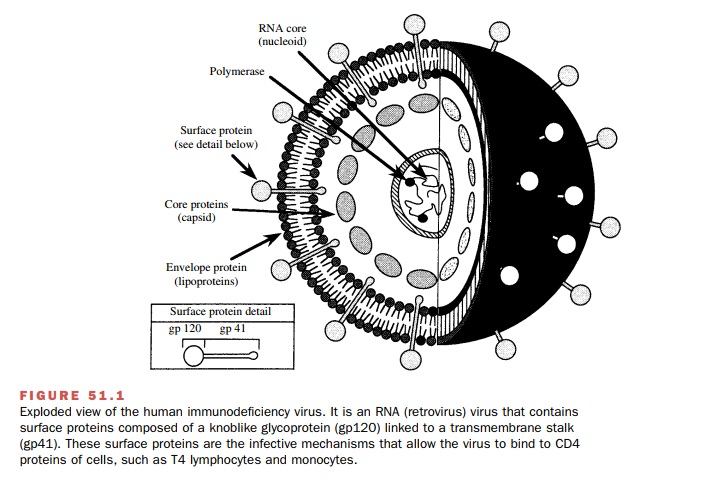
lymphocytes, macrophages, and dendritic cells. Viral entry is initiated when gp120 (SU), a glycoprotein on the
surface of the viral envelope, at-taches itself to the CD4 surface glycoprotein of the tar-get cell (Fig. 51.1). This
interaction produces a confor-mational change in gp120 that allows it to bind
to a chemokine coreceptor: CXCR4 for
CD-4 T (helper) cells or CCR5 for
macrophages.
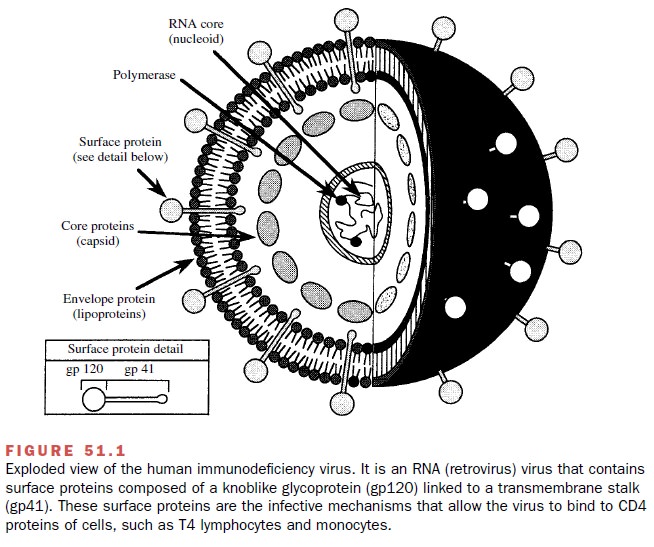
Chemokine coreceptor binding is required for viral
entry; individuals with ge-netic defects in these proteins are resistant to HIV
in-fection. The binding of gp120 to CXCR4 or CCR5 causes a rearrangement in the
envelope glycoproteins that allows the fusion of a viral transmembrane
glyco-protein (gp41) with the target
cell membrane. Fusion of the viral and cellular membranes follows as the virus
enters the target cell.
After entering the host cell
and uncoating, viral re-verse
transcriptase synthesizes DNA using viral RNA as a template. This DNA circularizes, enters the nucleus, and is
integrated into the host genome by another viral enzyme, integrase. The host cell then transcribes the vi-ral genes and
produces viral proteins and progeny viral RNA. New virions assemble, bud from
the cell mem-brane, and undergo a maturation process in which the gag-pol
polyprotein is cleaved by the viral enzyme pro-tease.
The resultant mature virus particles spread to in-fect other susceptible
cells.
The majority of viral
replication occurs in recently infected CD4+ lymphocytes and depletes them
during the first several years of infection. Macrophage popula-tions are
depleted or cease to function properly in 3 to 10 years or more. It is during
this time that an HIV-infected person becomes immunodeficient and can die of
infections that under normal conditions are not life threatening. Eventually
the macrophages of the brain (microglia) may become infected and an
inflammation-based dementia may occur.
Several pools of
nonreplicating virus serve as reser-voirs of infection and limit the
effectiveness of anti-retroviral therapy. HIV can live and multiply in
mono-cytes and macrophages; these cells are present in all tissues and can live
for many months. Infective virus can also reside in long-lived resting CD4+
lymphocytes.
Related Topics