Chapter: Modern Pharmacology with Clinical Applications: Therapy of Human Immunodeficiency Virus
Drug Therapy of HIV Infection: Nucleotide Reverse Transcriptase Inhibitors
Nucleotide Reverse Transcriptase
Inhibitors
Tenofovir
Tenofovir disoproxil fumarate
(Viread) is a prodrug of tenofovir, a
phosphorylated adenosine nucleoside ana-logue, and is the only available agent
of its class . It is converted by cellular enzymes to tenofovir diphos-phate,
which competes with deoxyadenosine triphos-phate (dATP) for access to reverse
transcriptase and causes chain termination following its incorporation.
Tenofovir was approved as part of a combination ther-apy for HIV in adults who
failed treatment with other regimens; it appears to be effective against HIV
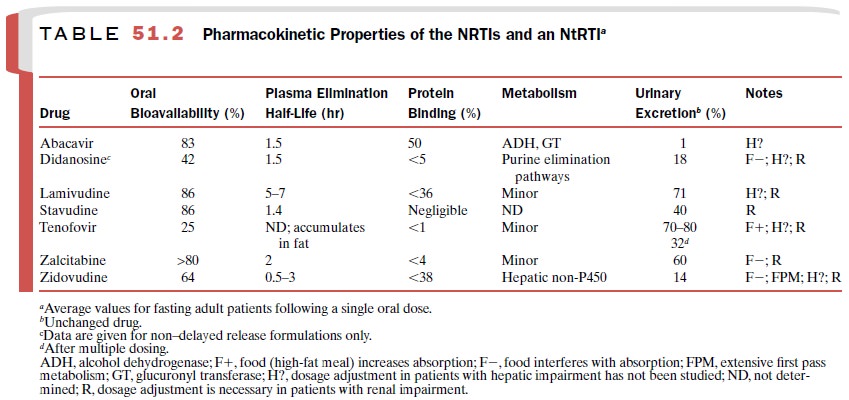
strains that are resistant to NRTIs. The pharmacokinetic prop-erties of
tenofovir are provided in Table 51.2.
Tenofovir is taken once daily
and is generally well tolerated, perhaps because it produces less
mitochondr-ial toxicity than the NRTIs. Nausea, vomiting, flatu-lence, and
diarrhea occur in 10% or fewer patients. Resistance to tenofovir has been
documented, and cross-resistance to NRTIs may occur.
Tenofovir should not be given
to patients with renal insufficiency. Its coadministration with didanosine
re-sults in increased plasma levels of didanosine that can produce toxicity.
Because lactic acidosis and severe he-patomegaly with steatosis have been
reported with NRTIs, it is important to monitor patients with known risk
factors during treatment with tenofovir.
Related Topics