Chapter: Medical Surgical Nursing: Genetics Perspectives in Nursing
Genes and Their Role in Human Variation
Genetics Concepts
Scientists and philosophers have long speculated about heredity and developed theories to explain how traits are transmitted to offspring. Developments in technology and research have accel-erated progress in our understanding of genetics, allowing scientists to better understand relatively rare diseases such as phenylketonuria (PKU) or hemophilia that are related to muta-tions of a single gene inherited in families. New technologies and tools allow scientists to characterize inherited metabolic varia-tions that interact over time and lead to common diseases such as cancer, heart disease, and dementia. This transition from genetics to genomics highlights how our understanding of single genes and their individual functions has evolved to understanding how multiple genes act and control biologic processes. Most health conditions are now believed to be the result of a combination of genetic and environmental influences and interactions (Billings, 2000).
GENES
AND THEIR ROLEIN HUMAN VARIATION
Genes are central components of human health and disease. Work on the Human Genome Project (an international
research effort to map and sequence the human genome in its entirety) has shown
how basic human genetics is to human development, health, and disease.
Knowledge that specific genes are associated with specific genetic conditions
makes diagnosis possible, even in the unborn. Research continues to demonstrate
how many common conditions have genetic causes. Many more associa-tions between
genetics, health, and disease will likely be identi-fied as scientists complete
and refine human genome mapping and sequencing.
Genes and Chromosomes
A person’s unique genetic constitution, called a genotype, is made up of some 30,000 to 40,000 genes. A person’s phenotype, the observable
characteristics of his or her genotype, includes physical appearance and other
biologic, physiologic, and molec-ular traits. Environmental influences modify
every individual’s phenotype, even those with a major genetic component.
Human growth, development, and disease occur as a result of both genetic
and environmental influences and interactions. The contribution of genetic
factors may be large or small. For exam-ple, in a person with cystic fibrosis
or PKU, the genetic contribu-tion is significant. In contrast, the genetic
contribution underlying a person’s response to infection may be less so.
An individual gene is conceptualized as a unit of heredity. A gene is
composed of a segment of deoxyribonucleic
acid (DNA) that contains a specific set of instructions for making the
protein or proteins needed by body cells for proper functioning. Genes regulate
both the types of proteins made and the rate at which proteins are produced.
The structure of the DNA molecule is re-ferred to as the double helix. The
essential components of the DNA molecule are sugar-phosphate molecules and
pairs of ni-trogenous bases. Each nucleotide
contains a sugar (deoxyribose), a phosphate group, and one of four nitrogenous
bases: adenine (A), cytosine (C), guanine (G), and thymine (T). DNA is
com-posed of two-paired strands, each made up of a number of nu-cleotides. The
strands are held together by hydrogen bonds between pairs of bases (Fig. 9-1).
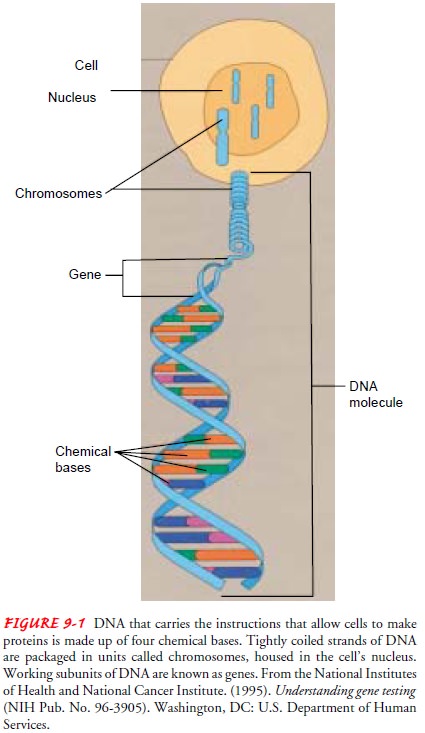
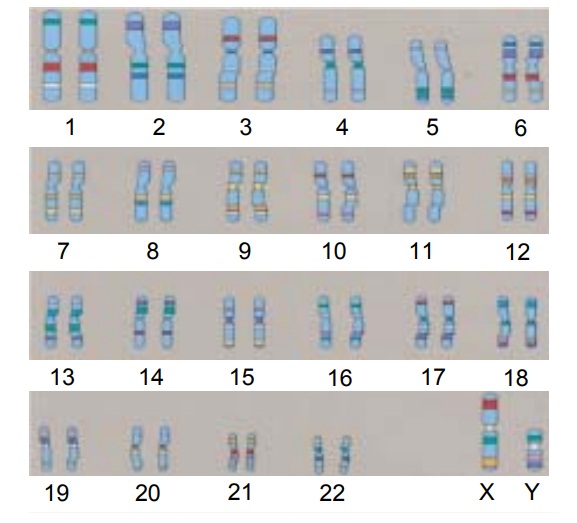
Genes are packaged and arranged in a linear order within chromosomes, which are located in the
cell nucleus. In humans,46 chromosomes occur in pairs in all body cells except
oocytes (eggs) and sperm, which each contain only 23 chromosomes. Twenty-two
pairs of chromosomes, called autosomes,
are the same in females and males. The 23rd pair is referred to as the sex
chromosomes. A female has two X chromosomes, while a male has one X and one Y
chromosome. At conception, each parent normally gives one chromosome of each
pair to his or her chil-dren. As a result, children receive half of their
chromosomes from their fathers and half from their mothers (Fig. 9-2).

Careful examination of DNA sequences from many individ-uals shows that these sequences have multiple versions in a pop-ulation. These different versions, or sequence variations, are called alleles. Sequences found in many forms are said to be poly-morphic, meaning that there are at least two common forms of a particular gene.
Cell Division
The human body grows and develops through a process of cell di-vision.
Mitosis and meiosis, two distinctly different types of cell division,
contribute to these processes.
Mitosis is the process of cell division involved in cell growth,differentiation,
and repair. During mitosis, the chromosomes of each cell duplicate. The result
is two cells, called daughter cells, each containing the same number of
chromosomes as the parent cell. The daughter cells are said to be diploid because they con-tain 46
chromosomes in 23 pairs. Mitosis occurs in all cells of the body except oocytes
(eggs) and sperm.
Meiosis, in contrast, occurs only in reproductive cells and isthe process by which oocytes and sperm are formed. During meiosis a reduction in the number of chromosomes takes place, resulting in oocytes or sperm that contain half the usual number or 23 chromosomes. Oocytes and sperm are referred to as hap-loid because they contain a single copy of each chromosome,compared to the usual two chromosomes in all other body cells.
During the initial phase
of meiosis, paired chromosomes come together in preparation for cell division,
portions cross over, and an exchange of genetic material occurs. This event,
called re-combination, creates greater diversity in the makeup of oocytes and
sperm.
During meiosis, a pair of chromosomes may fail to separate completely,
creating a sperm or oocyte that contains either two copies or no copy of a
particular chromosome. This sporadic event, called nondisjunction, can lead to either a trisomy or a monosomy. Down
syndrome is an example of trisomy.
Anindividual with Down syndrome has three number 21 chromo-somes. Turner
syndrome is an example of monosomy.
Girls who have Turner syndrome usually have a single X chromo-some, causing
them to have short stature and infertility (Lash-ley, 1998).
Gene Mutations
Within each cell, many intricate and complex interactions regu-late and
express human genes. Gene structure and function, tran-scription and translation, and protein synthesis are all
involved.Alterations in gene structure and function and the process of pro-tein
synthesis may influence a person’s health. Changes in gene structure, called mutations, permanently change the
sequence of DNA, which in turn can alter the nature and type of proteins made
(Fig. 9-3).
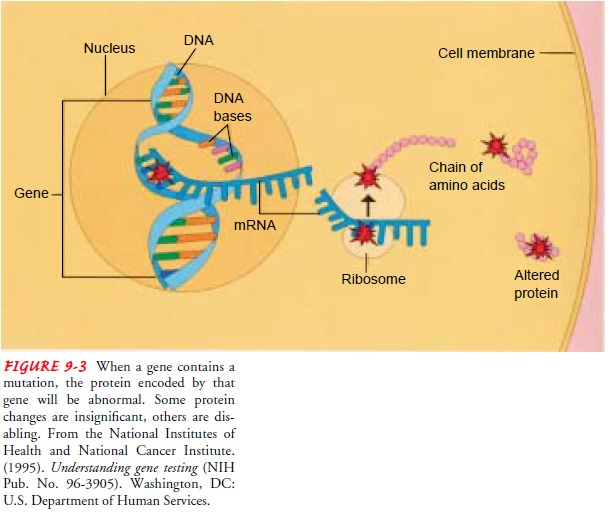
Some gene mutations have no significant effect on the pro-tein product
made, while others cause partial or complete changes. How a protein is altered
and its importance to proper body functioning determine the mutation’s impact.
Gene mu-tations may occur in hormones or enzymes or important pro-tein
products, thereby having significant implications for health and disease.
Sickle cell anemia is an
example of a genetic condition caused by a small gene mutation that affects
protein structure, producing hemoglobin S. A person who inherits two copies of
the hemoglobin S gene mutation has the condition sickle cell anemia and
experiences the symptoms of severe anemia and thrombotic organ damage resulting
from hypoxia (Lashley, 1998; Lea, 2000).
Other gene mutations may be larger, such as a deletion (loss), insertion (addition), duplication (multiplication), or rearrange-ment (translocation) of a longer DNA segment. Duchenne mus-cular dystrophy, an inherited form of muscular dystrophy, is an example of a genetic disorder caused by structural gene mutations such as deletions or duplications in the dystrophin gene. Another type of gene mutation, called a triplet or trinucleotide repeat, involves the expansion of more than the usual number of a triplet repeat sequence within a gene. Myotonic dystrophy, Huntington disease, and fragile X syndrome are examples of conditions caused by this type of gene mutation.
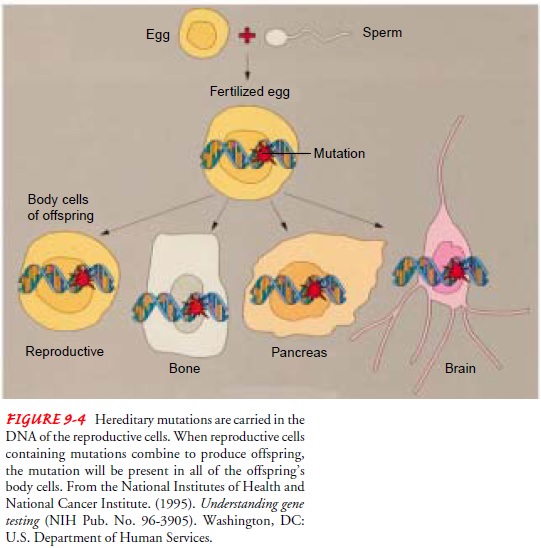
Gene mutations may be
inherited or acquired. Inherited or germ-line gene mutations are present in the
DNA of all body cells and are passed on in reproductive cells from parent to
child. Germ-line mutations are passed on to all daughter cells when body cells
replicate (Fig. 9-4). The gene that causes Huntington disease is one example of
a germ-line mutation.
Spontaneous gene
mutations take place in individual oocytes or sperm at the time of conception.
These mutations are not in-herited in other family members. A person who
carries the new “spontaneous” mutation, however, may pass on the gene muta-tion
to his or her children. Achondroplasia, Marfan syndrome, and neurofibromatosis type
1 are examples of genetic conditions that may occur in a single family member
as a result of sponta-neous mutation.
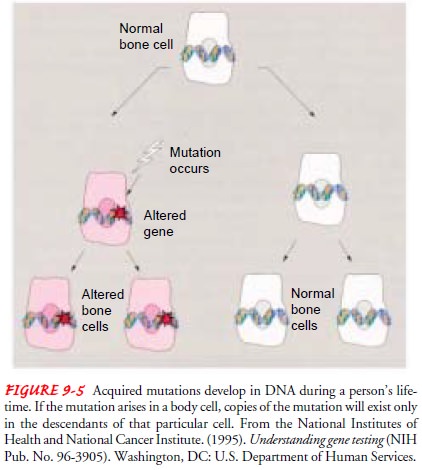
Acquired mutations take
place in somatic cells and involve changes in DNA that occur after conception,
during a person’s lifetime. Acquired mutations develop as a result of
cumulative changes in body cells other than reproductive cells (Fig. 9-5).
So-matic gene mutations are passed on to the daughter cells derived from that
particular cell line.
Gene mutations occur in the human body all the time. Cells have built-in
mechanisms by which they can recognize mutations in DNA, and in most situations
they correct the change before it is passed on by cell division. However, over
time, body cells may lose their ability to repair damage from gene mutations,
causing an accumulation of genetic changes that may ultimately result in
diseases such as cancer and possibly other conditions of aging, such as
Alzheimer’s disease (Lashley, 1998).
Genetic Variation
Sorting out the genetic
components of complex conditions (eg, heart disease, diabetes, common cancers,
psychiatric disorders) that result from the interaction of environment,
lifestyle, and the small effects of many genes is ongoing. New studies of
genetic
Genetic variations occur among individuals of all populations. Polymorphisms and single nucleotide
polymor-phisms (SNPs, pronounced “snips”) are the terms used for com-mon
genetic variations that occur most frequently throughout the human genome. Some
SNPs may contribute directly to a trait or disease expression by altering
function. SNPs are becoming in-creasingly important for the discovery of DNA
sequence varia-tions that affect biologic function. Such knowledge will allow
clinicians to subclassify diseases and adapt therapies to the indi-vidual
patient (Collins, 1999; Collins & McKusick, 2001). For example, a
polymorphism or SNP can alter a protein or enzyme activity and can thus affect
drug efficacy and safety when it oc-curs in proteins that are targets of
medication regimens or that are involved in drug transport or drug metabolism
(McCarthy & Hilfiker, 2000; Schafer & Hawkins, 1998).
Related Topics