Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Pathogenesis, Immunity, and Chemotherapy of Fungal Infections
General Aspects of Fungal Disease
GENERAL ASPECTS OF FUNGAL DISEASE
EPIDEMIOLOGY
Fungal infections are acquired from the environment or may be endogenous in the few instances where they are members of the normal flora. Inhalation of infectious conidia generated from molds growing in the environment is a common mechanism. Some of these molds are ubiquitous, whereas others are restricted to geographic areas whose climate favors their growth. In the latter case, disease can be acquired only in the endemic area. Some environmental fungi produce disease after they are accidentally injected past the skin barrier. The pathogenic fungi represent only a tiny fraction of those found in the environment. Endogenous infections are restricted to a few yeasts, primarily Candidaalbicans. These yeasts have the ability to colonize by adhering to host cells and, given theopportunity, invade deeper structures.
PATHOGENESIS
Compared with bacterial, viral, and parasitic disease, less is known about the patho-genic mechanisms and virulence factors involved in fungal infections. Analogies with bacterial diseases come the closest because of the apparent importance of adherence to mucosal surfaces, invasiveness, extracellular products, and interaction with phagocytes. Most fungi are opportunists, producing serious disease only in individuals with impaired host defense systems. Only a few fungi are able to cause disease in previously healthy persons.
Adherence
A number of fungal species, particularly the yeasts, are able to colonize the mucosal sur-faces of the gastrointestinal and female genital tracts. It has been shown experimentally that the ability to adhere to buccal or vaginal epithelial cells is associated with coloniza-tion and virulence. Within the genus Candida , the species that adhere best to epithelial cells are those most frequently isolated from clinical infections. Adher-ence usually requires a surface adhesin on the microbe and a receptor on the epithelial cell. In the case of C. albicans, mannoprotein components extending from the cell wall have been implicated as the adhesin and fibronectin, and other components of the extra-cellular matrix as the receptor(s). A few binding mediators have been identified for other fungi, usually a surface mannoprotein.
Invasion
Passing an initial surface barrier, whether skin, mucous membrane, or respiratory epithe-lium, is an important step for most successful pathogens. Some fungi are introduced through mechanical breaks. For example, Sporothrix schenckii infection typically follows a thorn prick or some other obvious trauma . Fungi that initially infect the lung must produce conidia small enough to be inhaled past the upper airway defenses. For example, arthroconidia of Coccidioides immitis (2 to 6μm) can remain suspended in air for a considerable time and can reach the terminal bronchioles to initiate pulmonary coccidioidomycosis.
Triggered by temperature and possibly other cues, dimorphic fungi from the environ-ment undergo a metabolic shift similar to the heat shock response and completely change their morphology and growth to a more invasive form. Invasion directly across mucosal barriers by the endogenous yeast C. albicans is similarly associated with a mor-phologic change, the formation of hyphae. The triggering mechanisms of this change are unknown, but the new form is able to penetrate and spread. Extracellular enzymes (eg, proteases, elastases) are associated with the hyphal form of Candida and with the inva-sive forms of many of the dimorphic and other pathogenic fungi. Although these enzymes must contribute to some aspect of invasion or spread, their precise role is unknown for any fungus.
Tissue Injury
None of the extracellular products of opportunistic fungi or dimorphic pathogens have been shown to injure the host directly during infection in a manner analogous to bacter-ial toxins. Although the presence of necrosis and infarction in the tissues of patients with invasion by fungi such as Aspergillus suggests a toxic effect, direct evidence is lacking. A number of fungi do produce exotoxins, called mycotoxins, in the environ-ment but not in vivo. The structural components of the cell do not cause effects similar to those of the endotoxin of Gram-negative bacteria, although mannan is known to circulate widely in the body. The injury caused by fungal infections seems to be due primarily to the inflammatory and immune responses that are stimulated by the pro-longed presence of the fungus.
IMMUNITY
Phagocyte Interactions
There is considerable evidence that normal persons have a high level of natural resistance to most fungal infections. This is particularly true of opportunistic molds. An important component of this resistance is the ability of healthy neutrophils to kill hyphae of most fungi if they reach the tissues. A small number of species, all of which are dimorphic, are able to produce mild to severe disease in otherwise healthy individuals. In vitro studies have shown these fungi to be more resistant to killing by neutrophils than the oppor-tunists. C. albicans is able to bind complement components in a way that interferes with phagocytosis.
C. immitis, one of the best-studied species, has been shown to contain a component inthe wall of its conidial (infective) phase that is antiphagocytic. As the hyphae convert to the spherule (tissue) phase, they also become resistant to phagocytic killing because of their size and surface characteristics. The tissue yeast form of Histoplasma capsulatum is resistant to phagocytic killing after ingestion and, in fact, multiplies within macrophages. These mechanisms of avoiding phagocytic killing appear to allow many dimorphic fungi to multiply sufficiently to produce an infection that can be controlled only by the immune re-sponse.
Adaptive Immune Response
A recurrent theme with fungal infections is the importance of an intact immune response in preventing infection and progression of disease. Most fungi are incapable of producing even a mild infection in immunocompetent individuals.
A small number of species are able to cause clinically apparent infection that usually resolves once there is time for activation of normal immune responses. In most instances in which it has been investigated, the actions of neutrophils and T lymphocyte–mediated immune responses have been found to be of primary importance in this resolution. Progressive, debilitating, or life-threatening disease with these agents is commonly associated with depressed or absent cell-mediated immune responses, and the course of any fungal disease is worse in immunocompromised than previously healthy persons.
Humoral Immunity
Antibodies can be detected at some time during the course of almost all fungal infections,but for most there is little evidence that they contribute to immunity. The only encapsulatedfungus, Cryptococcus neoformans, is one example of a fungus against which antibody playsa role in controlling infection. Although the polysaccharide capsule of C. neoformans hasantiphagocytic properties similar to those of encapsulated bacterial pathogens (eg, Strepto-coccus pneumoniae, Haemophilus influenzae), it is less antigenic. Anticapsular antibody plays a role in resolving cryptococcal infection, but T cell–mediated responses are still dominant. Antibody also plays a role in control of C. albicans infections by enhancingfungus–phagocyte interactions, and this is probably true for other yeasts. In some other fun-gal infections, the lack of protective effect of antibody is striking. In coccidioidomycosis,for example, high titers of C. immitis–specific antibodies are associated with disseminationand a worsening clinical course.
Cellular Immunity
Considerable clinical and experimental evidence points toward the importance of cellular immunity in fungal infections. Most patients with severe systemic disease have neutrope-nia, defects in neutrophil function, or depressed T lymphocyte–mediated immune reactions. These can result from factors such as steroid treatment, leukemia, Hodgkin’s disease, and acquired immunodeficiency syndrome. In other cases, an immunologic deficit can usually be demonstrated by absence of delayed-type hypersensitivity re-sponses or by direct in vitro assays of T-cell responsiveness to the fungus in question. In the latter case, it is possible that hyporesponsiveness is due at least in part to activation ofsuppressor cells or continued circulation of fungal antigen.
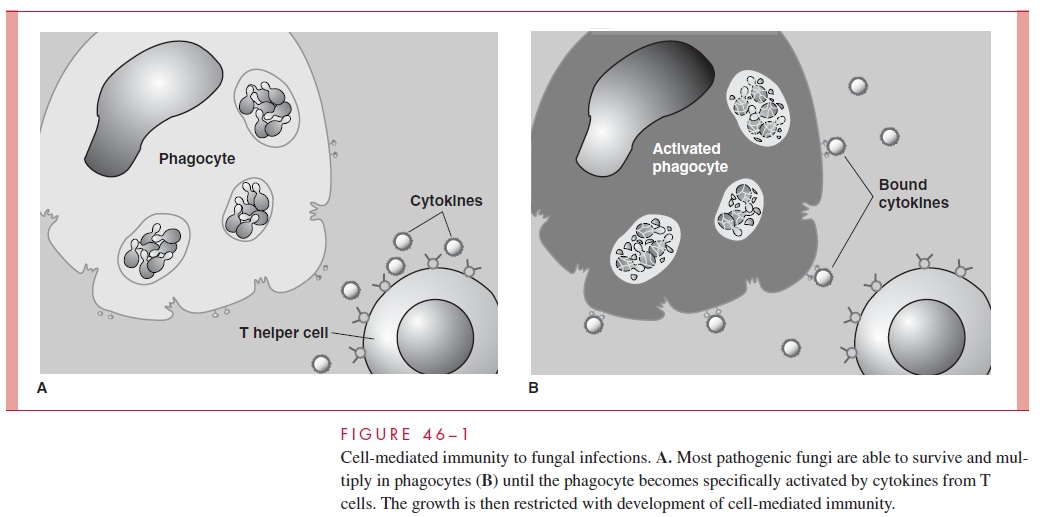
Although not all fungi have been studied to the same degree, a unified picture emerg-ing from clinical and experimental animal studies is illustrated in Figure 46–1. Whenhyphae or yeast cells of the fungus reach deep tissue sites, they are either killed by neu- trophils or resist destruction by one of the antiphagocytic mechanisms described earlier. Surviving cells continue to grow slowly or, if they are dimorphic, convert to their yeast,hyphal, or spherule tissue phases. The growth of these invasive forms may be slowed but not killed by macrophages. In healthy persons, the extent of infection is small, and any symptoms are caused by the inflammatory response. Everything awaits the specific im- mune response. The turning point comes when the macrophages are activated by cytokinemediators produced by T lymphocytes that have interacted with the fungal antigen. Where they have been identified, these mediators, which are associated with helper T-cell responses, are those such as interleukin 2 or interferon- γ . The activated macrophages arethen able to restrict the growth of the fungus, and the infection is controlled. Defects thatdisturb this cycle lead to progressive disease.
Related Topics