Chapter: Medical Microbiology: An Introduction to Infectious Diseases: Characteristics of Fungi
Fungal Morphology and Growth
FUNGAL MORPHOLOGY AND GROWTH
The size of fungi varies immensely. A single cell without transverse septa may range from bacterial size (2– 4μm) to a macroscopically visible structure. The morphologic forms of growth vary from colonies superficially resembling those of bacteria to some of the most complex, multicellular, colorful, and beautiful structures seen in nature. Mush-rooms are an example and can be regarded as a complex organization of cells showing structural differentiation.
Mycology, the science devoted to the study of fungi, has many terms to describe themorphologic components that make up these structures. Fortunately, the terms and con-cepts that must be mastered can be limited by considering only the fungi of medical im-portance and accepting some simplification.
YEASTS AND MOLDS
Initial growth from a single cell may follow either of two courses, yeast or mold (Fig 45–2). The first and simplest is the formation of a bud, which extends out from a
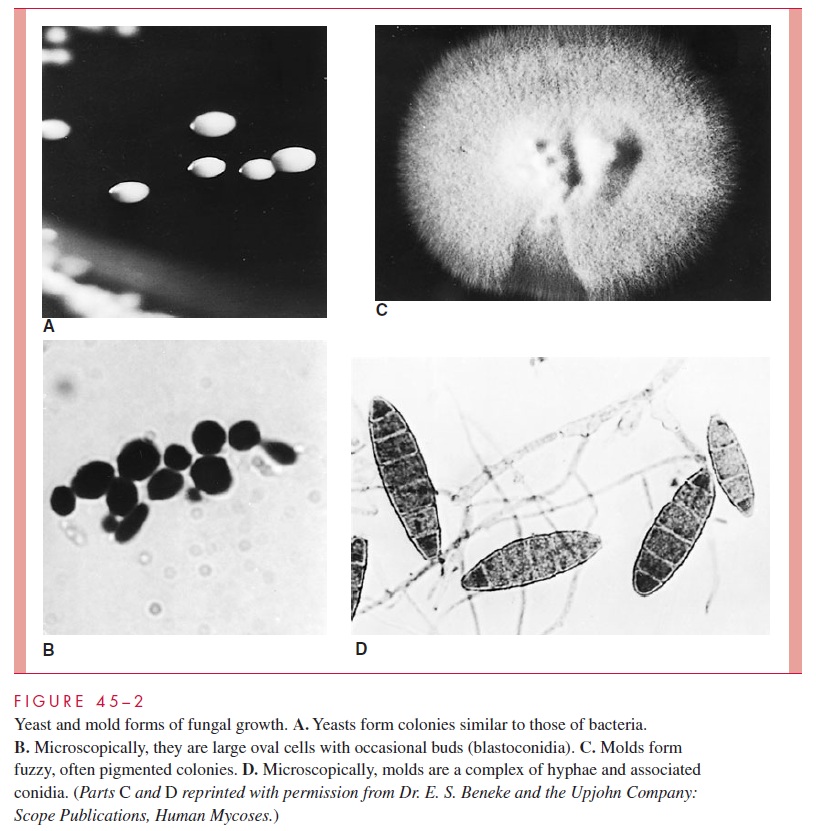
round or oblong parent, constricts, and forms a new cell. These buds are called blastoconidia (see Fig 45–2), and fungi that reproduce in this manner are called yeasts. On plates, yeasts form colonies that resemble those of bacteria. In broth, yeasts produce diffuse turbidity or grow as sediments in unshaken cultures.
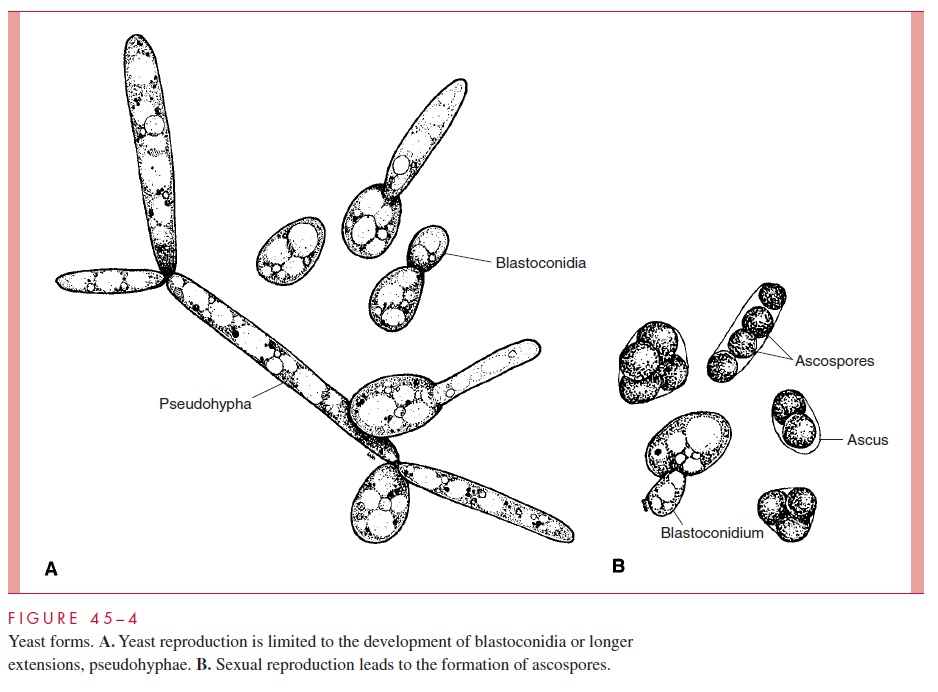
Fungi may also grow through the development of hyphae (singular, hypha), which are tube-like extensions of the cell with thick, parallel walls. As the hyphae extend, they form an intertwined mass called a mycelium. Most fungi form hyphal septa (singular, septum), which are cross-walls perpendicular to the cell walls that divide the hypha into subunits (Fig 45–3). These septa may not form complete walls and vary among species in the extent to which they restrict movement of organelles and nuclei. Some species are nonseptate; they form hyphae and mycelia as a single, continuous cell. In both septate and nonseptate hyphae, multiple nuclei are present, with free flow of cytoplasm along the hyphae or through pores in any septum. A portion of the mycelium (vegetative mycelium) usually grows into the medium or organic substrate (eg, soil) and functions like the roots of plants as a collector of nutrients and moisture. The more visible surface growth may assume a fluffy character as the mycelium becomes aerial. The hyphal walls are rigid enough to support this extensive, intertwining network, commonly called a mold. The aerial hyphae bear the reproductive structures of this class of fungi. Some fungi form structures called pseudohyphae (Fig 45– 4), which differ from true hyphae in having re-curring bud-like constrictions and less rigid cell walls.
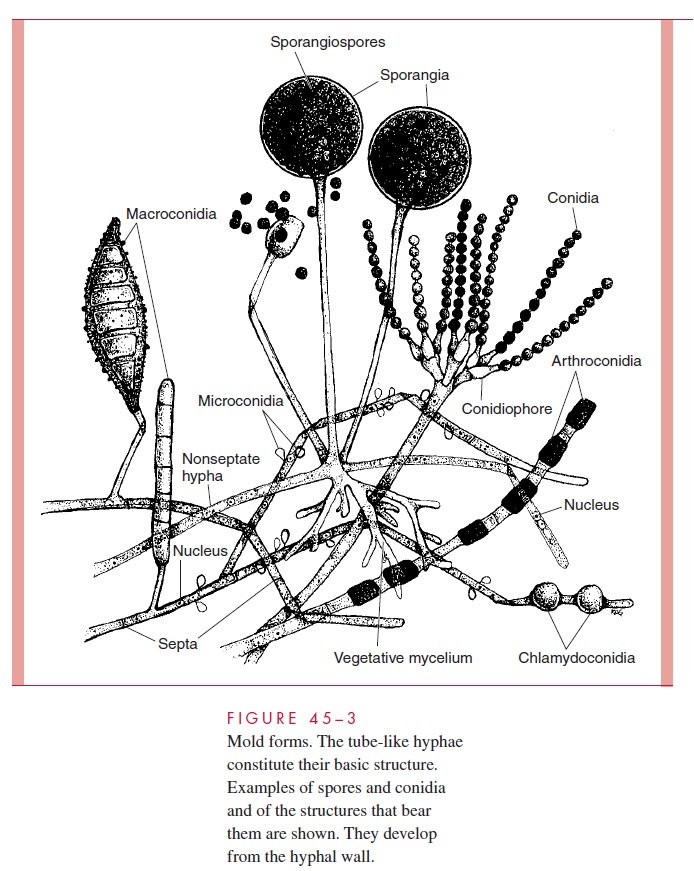
The reproductive conidia and spores of the molds and the structures that bear them assume a great variety of sizes, shapes, and relationships to the parent hyphae, and the morphology and development of these structures are the primary basis of identification of medically important molds. The mycelial structure plays some role in identification, depending on whether the hyphae are septate or nonseptate, but differences are not suffi-ciently distinctive to identify or even suggest a fungal genus or species.
Exogenously formed conidia may arise directly from the hyphae or on a special stalk-like structure, the conidiophore. Occasionally, terms such as macroconidia andmicro-conidia are used to indicate the size and complexity of these conidia. Conidia that developwithin the hyphae are called either chlamydoconidia or arthroconidia.Chlamydoconidia become larger than the hypha itself; they are round, thick-walled structures that may be borne on the terminal end of the hypha or along its course. Arthroconidia conform more to the shape and size of the hyphal units but are thickened or otherwise differentiated. Arthro-conidia may form a series of delicately attached conidia that break off and disseminate when disturbed. The most common sexual spore is termed an ascospore. Four or eight as-cospores may be found in a sac-like structure, the ascus. The structures are illustrated in Figures 45–3 and 45– 4.
DIMORPHISM
In general, fungi grow either as yeasts or as molds; mold forms show the greatest diver-sity. Some species can grow in either a yeast or a mold phase, depending on environmen-tal conditions. These species are known as dimorphic fungi. Several human pathogens demonstrate dimorphism; they grow in the mold form in their environmental reservoir and in culture at ambient temperatures, but convert to the yeast or some other form in in-fected tissue. For most, it is possible to manipulate the cultural conditions to demonstrate both yeast and mold phases in vitro. Yeast phase growth requires conditions similar to those of the parasitic in vivo environment, such as 35 to 37°C incubation and enriched medium. Mold growth requires minimal nutrients and ambient temperatures. The conidia produced in the mold phase may be infectious and serve to disseminate the fungus.
CLASSIFICATION
Although conidia are more readily observed, the major classification of fungi primarily depends on the nature of sexual spores and septation of hyphae as its differential charac-teristics. On this basis, fungi have been organized into four to six classes or phyla. A ma-jor problem of classifying the medically important fungi using these groups is that for most species, no sexual form has been demonstrated. This may be due to its loss during evolution or because the spores are so rarely produced that they have not been detected. One approach has been to give these fungi their own class (Deuteromycetes, or fungi im-perfecti) and wait for the discovery of the sexual form to place it in one of the legitimategroups—the Ascomycetes, Basidiomycetes, or Zygomycetes. The application of molecu-lar methods such as analysis of ribosomal RNA genes has allowed the placement of species pending discovery of the sexual forms. The medically important genera are shown in Table 45–1. Discovery of the sexual form may not bring immediate clarity from the student’s standpoint; for instance, when the sexual stage of Trichophyton mentagrophytes was demonstrated, it was found to be identical to that of an already named ascomycete (Arthroderma benhamiae). Most medically important species are now assigned to the As-comycetes and a few to the Basidiomycetes or Zygomycetes.
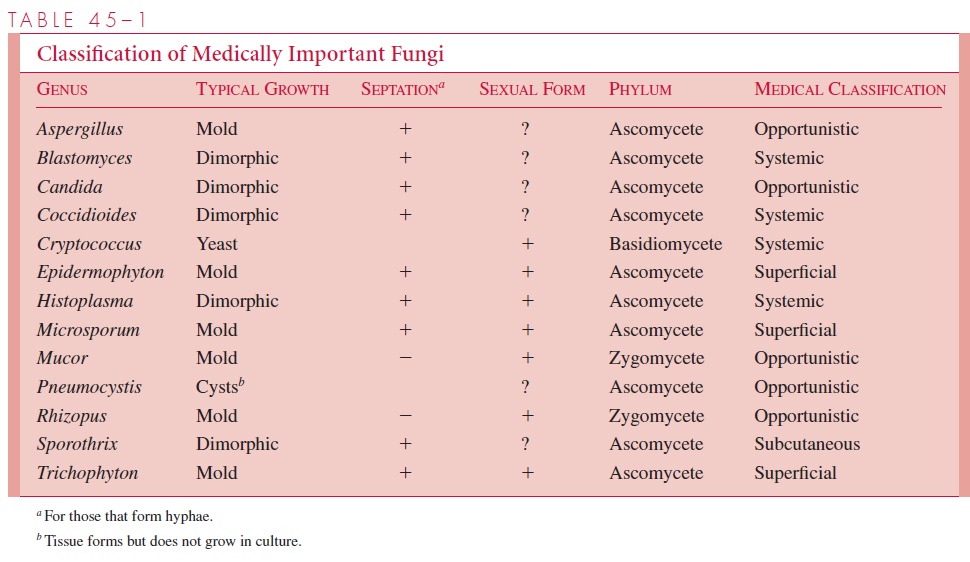
The grouping of medically important fungi used is based on the types of tissues they parasitize and the diseases they produce, rather than on the prin-ciples of basic mycologic taxonomy. The superficial fungi, such as the dermatophytes, cause indolent lesions of the skin and its appendages, commonly known as ringworm and athlete’s foot. The subcutaneous pathogens characteristically cause infection through the skin, followed by subcutaneous spread, lymphatic spread, or both. The opportunisticfungi are those found in the environment or in the normal flora that occasionally produce disease, usually in the compromised host. The systemic pathogens are the most virulent fungi and may cause serious progressive systemic disease in previously healthy persons. They are not members of the normal human flora. Although their major potential is to produce deep-seated visceral infections and systemic spread (systemic mycoses), they may also produce superficial infections as part of their disease spectrum or as the initiat-ing event. The superficial mycoses do not spread to deeper tissues. As with all clinical classifications, overlaps and exceptions occur. In the end, the organism defines the dis-ease, and it must be isolated or otherwise demonstrated.
EPIDEMIOLOGY
Most fungal infections arise from contact with an environmental reservoir or from the pa-tient’s own fungal flora. Some superficial mycoses can be transmitted from person to person by very close contact, such as sharing a comb with an individual who has scalp ringworm; others can be acquired from ringworm infections of animals. Other fungal infections are not communicable between humans or animals, and infected patients need not be isolated.
DIAGNOSIS
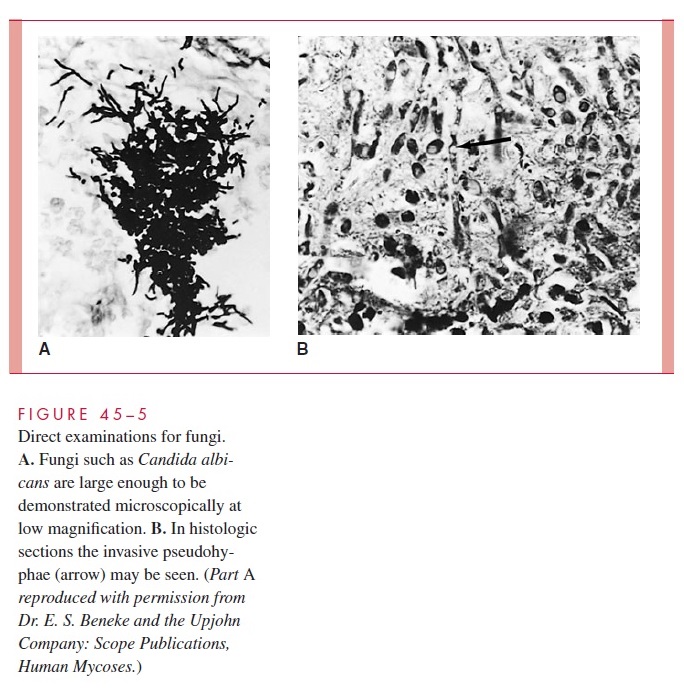
Because of their large size, fungi often demonstrate distinctive morphologic features on di-rect microscopic examination of infected pus, fluids, or tissues. The simplest method is to mix the specimen with a 10% solution of potassium hydroxide (KOH) preparation and place it under a coverslip. The strong alkali digests or clears the tissue elements (epithelial cells, leukocytes, debris) but not the rigid cell walls of both yeasts and molds. After diges-tion of the material, the fungi can be observed under the light microscope with or without staining (see Fig 47–1B). Some yeasts stain with common stains such as the Gram stain, to which they are usually positive. Direct examinations can be aided by the use of calcifluor white, a dye that binds to polysaccharides in cellulose and chitin. Under ultraviolet light, calcifluor white fluoresces, enhancing detection of fungi in fluids or tissue sections.
Histopathologic examination of tissue biopsy specimens is widely used and shows the relationship of the organism to tissue elements and responses (blood vessels, phagocytes, granulomatous reactions). Most fungi can be seen in sections stained with the hema-toxylin and eosin (H&E) method routinely used in histology laboratories (Fig 45–5). Spe-cialized staining procedures such as the silver impregnation methods are frequently used because they stain almost all fungi strongly but only a few tissue components. The pathol-ogist should be alerted to the suspicion of fungal infection when tissues are submitted, because special stains and searches for fungi are not made routinely.
Fungi can be grown by methods similar to those used to isolate bacteria. Growth oc-curs readily on enriched bacteriologic media commonly used in clinical laboratories (eg, blood agar and chocolate agar). Many fungal cultures, however, require days to weeks of incubation for initial growth; bacteria present in the specimen grow more rapidly and may interfere with isolation of a slow-growing fungus. Therefore, the culture procedures of di-agnostic mycology are designed to favor the growth of fungi over bacteria and to allow incubation to continue for a sufficient time to isolate slow-growing strains.
The most commonly used medium for cultivating fungi is Sabouraud’s agar, which contains only glucose and peptones as nutrients. Its pH is 5.6, which is optimal for growth of dermatophytes and satisfactory for growth of other fungi. Most bacteria associ-ated with humans fail to grow or grow poorly on Sabouraud’s agar.
Blood agar or another enriched bacteriologic agar medium is used when pure cultures would be expected. It is made selective for fungi by the addition of antibacterial antibi-otics such as chloramphenicol and gentamicin. Cycloheximide, an antimicrobic that inhibits some saprophytic fungi, is sometimes added to Sabouraud’s agar to prevent over-growth of contaminating molds from the environment, particularly for skin cultures. Media containing these selective agents cannot be relied on exclusively because they can interfere with growth of some pathogenic fungi or because the “contaminant” may be producing an opportunistic infection. For example, cycloheximide inhibits Cryptococcusneoformans, and chloramphenicol may inhibit the yeast forms of some dimorphic fungi.Selective media are not needed for growing fungi from sterile sites such as cerebrospinal fluid or tissue biopsy specimens. In contrast to most parasitic bacteria, many fungi grow best at 25 to 30°C, and temperatures in this range are used for primary isolation. Paired cultures incubated at 30 and 35°C may be used to demonstrate dimorphism.
Once a fungus is isolated, identification procedures depend on whether it is a yeast or mold. Yeasts are identified by biochemical tests analogous to those used for bacteria, in-cluding some that are identical (eg, urease production). The ability to form pseudohyphae is also taxonomically useful among the yeasts.
Molds are most often identified by the morphology of their conidia and conidio-phores. Other features such as the size, texture, and color of the colonies help characterize molds, but without demonstrating conidiation they are not sufficient for identification. The ease and speed with which various fungi produce conidia vary greatly. Minimal nu-trition, moisture, good aeration, and ambient temperature favor development of conidia.
Microscopic fungal morphology is usually demonstrated by methods that allow in situ microscopic observation of the fragile asexual conidia and their shape and arrangement. Morphology may also be examined in fragments of growth teased free of a mold and ex-amined moist in preparations containing a dye called lactophenol cotton blue. The dye stains the hyphae, conidia, and spores. Conidium production may not occur for days or weeks after the initial growth of the mold. It is somewhat like waiting for flowers to bloom, and it can be frustrating when the result has immediate clinical application.
It is desirable, but not always possible, to demonstrate both the yeast and mold phases with dimorphic fungi. In some cases, this result can be achieved with parallel cultures at 30° and 35°C. The tissue form of Coccidioides immitis is not readily produced in vitro. An alternate approach has been developed for identification of some of the dimorphic systemic fungi, based on soluble antigens prepared from mycelial growth (exoantigens) and called the exoantigen test. When these exoantigens react with specific antibody in an immunodiffusion procedure, precipitin lines are formed between the unknown antigen and its homologous antibody. Results are usually available much more rapidly than are results of cultural tests. For a few fungi, DNA probes are available for rapid speciation.
Serum antibodies directed against a variety of fungal antigens can be detected in pa-tients infected with those agents. Except for some of the systemic pathogens, the sensitiv-ity, specificity, or both, of these tests have not been sufficient to recommend them for use in diagnosis or therapeutic monitoring of fungal infections. The tests of value are dis-cussed in sections on specific agents.
Related Topics