Chapter: Plant Biology : Plants, water and mineral nutrition
Functions of mineral nutrients
FUNCTIONS OF MINERAL NUTRIENTS
Key Notes
Essential macro- and micronutrients
Plants depend on a range of essential mineral nutrients, which are extracted from the soil by the roots. These are categorized as macro- and micronutrients depending on the quantity required. Examples of macronutrients are sulfur, phosphorus, nitrogen, magnesium, potassium and calcium.
Essential, beneficial and toxic elements
Minerals may be categorized according to their effects on the plant. Essential elements are those without which a plant cannot reproduce; beneficial elements have beneficial effects on plant growth, but the plant can complete its life cycle without them. Toxic elements are deleterious to growth. Some elements are essential at low concentrations, but toxic at higher concentrations.
Key macronutrients
Nitrogen is a constituent of amino acids and proteins and is taken up as either nitrate or ammonium from soils or by nitrogen-fixing organisms. Nitrogen is transported as reduced nitrogen compounds. Sulfur is required in sulfur-containing amino acids to maintain protein structure. Phosphorus is required for membranes, nucleic acids and ATP. It is transported either as inorganic phosphate or as sugar phosphates. Its uptake is enhanced by mycorrhizal fungi. K+, Mg2+ and Ca2+ are all watersoluble cations. K+ is required for enzyme activity and osmo-regulation, Ca2+ for membrane stability and as an intracellular regulator and Mg2+ for chlorophyll and enzyme activity.
Nutrient deficiency and toxicity
Plants growing without enough of a nutrient show deficiency symptoms related to the function of the nutrient in the plant. Toxic ions such as aluminum may result in deficiency symptoms as they act by restricting the availability or uptake of nutrients. Good agricultural practice seeks to maximize the availability of nutrients and minimize toxic ions.
Essential macroand micronutrients
A nutrient is essential if: (i) it is required for the plant to complete a normal life cycle; (ii) it can be shown to be a component of the plant, either as part of structure or metabolism; and (iii) its function cannot be substituted for another element. Nutrients are either macronutrients (required in large amounts) or micronutrients (trace elements) required in much smaller amounts. Table 1 lists a range of both macro- and micronutrients. Theavailable form (i.e. the form in which the nutrient is transported into the plant) is usually either an anion
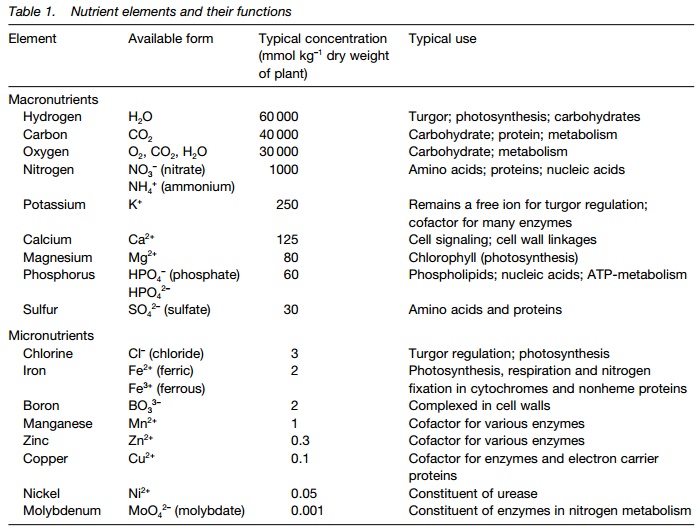
(negative) or a cation (positive). The distinction between macro- and micronutrients is to some extent arbitrary as some species contain more and others less of a particular nutrient; macronutrients are conveniently defined as those present at >10 mmol kg–1 dry matter.
Essential, beneficial and toxic elements
Some minerals are important in plant nutrition, but not essential. Elements which result in improved growth or reduced disease susceptibility, but without which the plant can still complete its life cycle are known as beneficial. An example of such an element is silicon (Si), which causes increased structural strength of cell walls, increased tolerance of toxic elements in soils and improved resistance to fungal pathogens. Some other elements are usually toxic, or may be toxic at high concentrations. Aluminum is almost always toxic when available (at acidic soil pH) as it complexes phosphate. Sodium, zinc, copper, manganese, boron, molybdenum and iron can all be toxic if present at high concentrations in the soil. Species differ in their ability to tolerate toxic ions and in some instances ions, which are normally toxic, are known to be beneficial. The growth of the tea plant, for instance, is enhanced by the presence of available soil aluminum, which it takes up as an organic acid complex, and the growth of many halophytes by sodium.
Key macronutrients
Nitrogen
Nitrogen gas is abundant in the aerial and soil environment, but unlike oxygen cannot be used directly. Nitrogen is fixed from the atmosphere by a number of microorganisms, which may be free-living, or in symbiotic association with some species of plants, mainly legumes . Other sources of available soil nitrogen may be decaying organic material, animal excreta and chemical fertilizers, frequently added to agricultural land.
Nitrogen assimilation
Plants take up either nitrate (NO3 –) or ammonium (NH4 +) from the soil, dependingon availability and species. Nitrate will be more abundant in well-oxygenated, nonacidic soils, whilst ammonium will predominate in acidic or waterlogged soils.After uptake, nitrate is reduced to ammonium in two stages by nitrate reductase(NR) and nitrite reductase before assimilation into amino acids.

NR is a dimer of two identical subunits. Expression of the gene for NR requires light and nitrate. The gene is expressed in both shoots and roots, but in low nitrate conditions, almost all nitrate assimilation will occur in the roots. Crops grown at high temperatures but only moderate light intensity may accumulate nitrate in the vacuole as a result of low induction of the NR gene.
Ammonium is toxic to plants, and is therefore rapidly incorporated into amino acids via the glutamine synthase-glutamate synthase (GS-GOGAT) pathway:
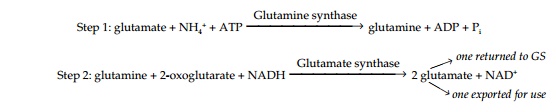
The process runs as a cycle, one of the glutamates produced being used as substrate by GS while the other is exported to the plant. Inputs to the cycle are ammonium and 2-oxoglutarate (from photosynthate imported via the phloem).
In plants with nitrogen-fixing nodules, nitrogen is exported to the plant from the nodule in the xylem flow as high-nitrogen containing compounds such as amino acids or ureides.
Sulfur
Sulfur is taken up in the form of sulfate (SO4 2–) by high-affinity transportproteins at the plasma membrane. The expression of these proteins varies withsulfate availability, the genes being repressed by high sulfate and activated bylow sulfate. Once in the plant, sulfate is reduced to the sulfur-containing aminoacid cysteine. The entire process involves the donation of 10 electrons and avariety of electron donors is required. It is more active in photosynthesizingleaves as the chloroplast provides a supply of electron donors. Sulfur is predominantly transported around the plant in thephloem, as glutathione. Glutathione (GSH) is a tripeptide formed of three amino acids (γ-glutamyl–cysteinyl– glycine) and acts as a storage form of sulfur in the plant.
Phosphate
Phosphate is not reduced in the plant. Root cells contain phosphate transporters at the plasma membrane and after uptake it either travels in xylem asinorganic phosphate (Pi) or is esterified through a hydroxyl group on a sugar orother carbon compound. Phosphate taken up by the root is rapidly incorporatedinto sugar phosphates, but is released as Pi into the xylem. Roots of plants arefrequently in symbiotic association with mycorrhizal fungi that extract phosphateefficiently from the soil . Phosphate is an essential constituentof nucleic acids and of many of the compounds of energy metabolism and it isutilized throughout the plant.
Nutrient cations
The nutrient cations (e.g. potassium, magnesium and calcium) are water soluble and transported as cations in the xylem. Potassium is very soluble and highly mobile, calcium being the least mobile of the three. Potassium is required for enzyme activity and osmotic-regulation, calcium maintains cell membrane stability, is an intracellular regulator and forms calcium pectate links in cell walls at middle lamellae . Magnesium is required as a central component of chlorophyll and for the activity of some enzymes.
Nutrient deficiency and toxicity
Plants grown in the absence of sufficient quantities of a particular nutrient show visible deficiency symptoms that relate to the function of that nutrient in the plant. Key terms are chlorosis, a lack of chlorophyll and yellowing of the leaves, and necrosis, the death of cells, often the growing tip or in lesions in the leaf surface. The extent and nature of the symptoms observed depend on where the nutrient is required and whether it can be redistributed in the plant. Potassium deficiency, for instance, causes necrosis of leaf margins and tips, whereas deficiency of manganese (a micronutrient) causes necrosis of tissue between the leaf veins (interveinal necrosis).
Development of symptoms such as those for deficiency can result from the presence of toxic elements, which interfere with the availability or transport of nutrients. Aluminum is more available in acidic soils and complexes with phosphate, creating phosphate deficiency. Plants show adaptations for the nutritional characteristics of the soils in which they grow. Calcicoles are adapted for growth in an alkaline, high calcium environment, where other nutrients are of low availability, while calcifuges are adapted to acidic soils, with high levels of aluminum and low levels of phosphate. Sodium competes with potassium for uptake resulting in potassium deficiency and failure to osmoregulate. Halophytes show a range of adaptations for growth in saline conditions, including salt extruding glands, high levels of discrimination between sodium and potassium for transport and xerophytic characteristics to minimize transpiration and salt uptake.
Many agricultural crops make severe demands on the nutrients in the soils in which they are grown; growth of cereals, for instance, results in the removal of large quantities of soil nitrogen and sulfur in seed protein. Good agricultural practice aims to maintain soil nutrient levels and minimize the availability of toxic compounds. For instance liming, the application of calcium or magnesium oxides, hydroxides or carbonates, is used to neutralize acidic soils and reduce aluminum toxicity, thereby increasing phosphate availability.
Related Topics