Chapter: Civil : Mechanics Of Fluids : Fluid Properties And Fluid Statics
Fluid Properties
INTRODUCTION
TO FLUIDS
Definition
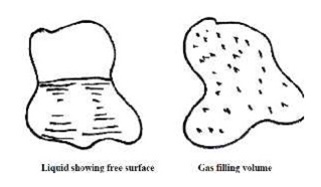
There are three states of matter: solids, liquids
and gases. Both liquids and gases are classified as fluids.
Fluids do not resist a change in shape. Therefore
fluids assume the shape of the container they occupy.
Liquids may be considered to have a fixed volume
and therefore can have a free surface.
Liquids are almost incompressible.
C o n v e r s e l y , gases are easily compressed
and will expand to fill a container they occupy.
We will usually be interested in liquids, either at rest or in
motion.
Definition
The strict definition of a fluid is: A fluid is
a substance which conforms continuously
under
the action of shearing forces.
To understand this, remind ourselves of what a shear force is:
2
Static Fluids
According to this definition, if we apply a shear
force to a fluid it will deform and take up
a state in which no shear force exists. Therefore,
we can say: If a fluid is at rest there can be no shearing forces
acting and therefore all forces in the fluid must be perpendicular to the
planes in which they act. Note here that we specify that the fluid must be
at rest. This is because, it is found experimentally that fluids in
motion can have slight resistance to shear force. This is
the source of viscosity.
3
Fluids
in Motion
For
example, consider the fluid shown flowing along a fixed surface. At the surface
there will be little movement of the fluid (it will 'stick' to the
surface), whilst furtheraway from the surface the fluid flows faster (has
greater velocity):
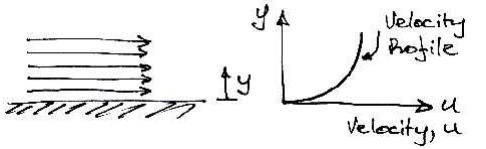
If one layer of is moving faster than another layer of fluid,
there must be shear forcesacting between them. For example, if we have fluid in
contact with a conveyor beltthat is moving we will get the behaviour shown:
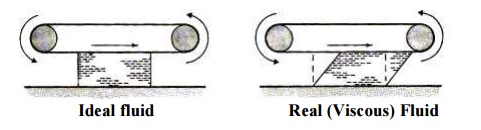
When fluid is in motion, any difference in
velocity between adjacent layers has the same effect as the conveyor belt does.
Therefore, to represent real fluids in motion we
must consider the action of shear forces
Consider the small element of fluid shown, which is subject to
shear force and has a dimension sinto the page. The force F acts
over an area A = BC� s. Hence we have a shear stress
applied:

Any stress causes a deformation, or strain, and a shear stress
causes a shear strain. This shear strain is measured by the angle ? .
Remember that a fluid continuously deforms when under the action of
shear. This is different to a solid: a solid has a single value of ? for each
value of ? . So the longer a shear stress is applied to
a fluid, the more shear strain occurs. However, what is known from experiments
is that the rate of shear strain (shear strain per unit time) is related to the
shear stress:

We need to know the rate of shear strain. From the diagram,
the shear strain is:

If we suppose that the particle of fluid at E moves a
distance x in time t, then, using S = R? for small
angles, the rate of shear strain is:
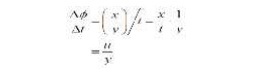
Where u is the velocity of the fluid. This term is also
the change in velocity with height. When we consider infinitesimally small
changes in height we can write this in differential form, du/ dy . Therefore
we have:
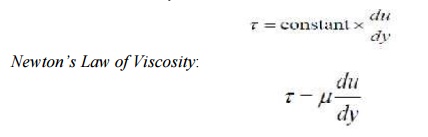
Newton's Law of
Viscosity:
Generalized
Laws of Viscosity
We have derived a law for the behaviour of fluids - that of Newtonian
fluids. However, experiments show that there are non-Newtonian
fluids that follow a generalized law of viscosity:
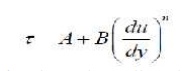
Where A, B and n are constants found
experimentally. When plotted these fluids show much different behaviour to a
Newtonian fluid:
Behaviour of Fluids and Solids
In this graph the Newtonian fluid is represent by
a straight line, the slope of which is ? . Some
of
the other fluids are:
Plastic: Shear stress must reach a certain
minimum before flow commences.
Pseudo-plastic: No minimum shear stress
necessary and the viscosity decreases with rate of shear, e.g.
substances like clay, milk and cement.
Dilatant substances; Viscosity
increases with rate of shear, e.g. quicksand.
Viscoelastic materials: Similar
to Newtonian but if there is a sudden large change in shear they behave
like plastic.
Solids: Real solids do have a slight
change of shear strain with time, whereas ideal solids (those we
idealise for our theories) do not. Lastly, we also consider the ideal
fluid. This is a fluid which is
assumed to have no viscosity and is very useful for developing
theoretical solutions. It helps achieve some practically useful solutions.
Properties
Further
Reading
Here we consider only the relevant properties of
fluids for our purposes. Find out about
surface
tension and capillary action elsewhere. Note that capillary action only
features in
pipes
of
?
10 mm diameter.
4.
FLUID PROPERTIES:
1.
Density or Mass density: Density
or mass density of a fluid is defined as the ratio of the mass of a
fluid to its volume. Thus mass per unit volume of a is called density.
Mass
density fluid / Mass of Density of fluid
The
unit of density in S.I. unit is kg/m3. The value of density for
water is 1000kg/m
2.Specific weight or weight density: Specific
weight or weight density of a fluid is the ratio between the weight of a
fluid to its volume. The weight per unit volume of a fluid is called weight
density.
Weight density = Weight
of fluid / Volume of fluid
w = Mass of fluid x g /
Volume of fluid
The unit of specific weight in S.I. units is N/m3.
The value of specific weight or weight density of water is
9810N/m3.
3.)Specific Volume: Specific volume of a fluid is
defined as the volume of a fluid occupied by a unit mass or volume per
unit mass of a fluid.
Specific volume = Volume
of a fluid / Mass of fluid
Thus specific volume is the reciprocal of mass
density. It is expressed as m3/kg. It is commonly applied to gases.
4.)Specific Gravity: Specific gravity is defined as
the ratio of the weight density of a fluid to the weight density of a
standard fluid.
Specific gravity = Weight density of liquid / Weight density
of water
VISCOSITY
Viscosity
is defined as the property of a fluid which offers resistance
to the movement of one layer of fluid over adjacent layer of
the fluid. When two layers of a fluid, a distance 'dy' apart,
move one over the other at different velocities, say u and u+du as shown in
figure. The viscosity together with relative velocity causes a shear stress
acting between the fluid layers.
COMPRESSIBILITY:
Compressibility is the reciprocal of the bulk
modulus of elasticity, K which is defined as the ratio of compressive stress to
volumetric strain.
Consider
a cylinder fitted with a piston as shown
in
figure. Let V= Volume of a gas enclosed
in the
cylinder
P=
Pressure of gas when volume is V
Let
the pressure is increased to p+dp, the volume of gas decreases
from
V to V-dV. Then increase in pressure =dp kgf/m2
Decrease
in volume= dV
Volumetric Strain = d / V
- ve sign means the volume decreases with increase
of pressure.
Bulk modulus K = Increase pressure / Volumetric
Strain
= dp / dV/V
Compressibility is given by = 1/K
Relationship
between K and pressure (p) for a Gas:
The relationship between bulk modulus of
elasticity (K) and pressure for a gas for two different processes of comparison
are as:
(i)
For Isothermal Process: The
relationship between pressure (p) and density (?) of a gas
as
p = Constant
V = Constant
Differentiating
this equation, we get (p and V are
variables)
PdV
+Vdp = 0
or
pdV=
- Vdp
or
p/dV = Vdp
Substituting
this value K =p
(ii) For adiabatic
process. For adiabatic
process
p Constant or pVk
= Constant
SURFACE
TENSION:
Surface tension is defined as the tensile force
acting on the surface of a liquid in contact with a gas or on the surface
between two two immiscible liquids such that the contact surface behaves like a
membrane under tension
CAPILLARITY
Capillarity
is defined as a phenomenon of rise or fall of a liquid surface in a small tube
relative to the adjacent general level of liquid when the tube is held
vertically in the liquid. The rise of liquid surface is known as capillary rise
while the fall of the liquid surface is known as capillary depression. It is
expressed in terms of cm or mm of liquid. Its value depends upon the specific
weight of the liquid, diameter of the tube and surface tension of the liquid.
Problem
1.
Calculate
the capillary effect in millimeters a glass tube of 4mm diameter, when immersed
in (a) water (b) mercury. The temperature of the liquid is 200 C and
the values of the surface tension of water and mercury at 200 C in
contact with air are 0.073575 and 0.51 N/m respectively. The angle of contact
for water is zero that for mercury 1300. Take specific weight of
water as 9790 N / m3

Problem
2.
A cylinder of 0.6 m3 in volume contains
air at 500C and 0.3 N/ mm2 absolute pressure. The air is
compressed to 0.3 m3. Find (i) pressure inside the cylinder assuming
isothermal process (ii) pressure and temperature assuming adiabatic process.
Take K = 1.4

Problem
3
If
the velocity profile of a fluid over a plate is a parabolic with the vertex 202
cm from the plate, where the velocity is 120 cm/sec. Calculate the velocity
gradients and shear stress at a distance of 0,10 and 20 cm from the plate, if
the viscosity of the fluid is 8.5 poise.

Problem
4
A
15 cm diameter vertical cylinder rotates concentrically inside another cylinder
of diameter 15.10 cm. Both cylinders are 25 cm high. The space between the
cylinders is filled with a liquid whose viscosity is unknown. If a torque of
12.0 Nm is required to rotate the inner cylinder at 100 rpm determine the
viscosity of the fluid.
Solution:
Diameter
of cylinder = 15 cm = 0.15 m
Diameter
of outer cylinder = 15.10 cm = 0.151 m
Length
of cylinder � L = 25 cm = 0.25 m
Torque
T= 12 Nm ; N = 100 rpm.
Viscosity
= m

Problem
5
The
dynamic viscosity of oil, used for lubrication between a shaft and sleeve is 6
poise. The shaft is of diameter 0.4 m and rotates at 190 rpm. Calculate the
power lost in the bearing for a sleeve length of 90 mm. The thickness of the
oil film is 1.5 mm.

Problem
6
If
the velocity distribution over a plate is given by u =2 y/3 -y 2 in which U is the velocity in m/s at a
distance y meter above the plate, determine the shear stress at y = 0 and y =
0.15 m. Take dynamic viscosity of fluid as 8.63 poise.

Problem
7
The diameters of a small piston and a large piston
of a hydraulic jack at3cm and 10 cm respectively. A force of 80 N is applied on
the small piston Find the load lifted by the large piston when:
�
The pistons are at the same level
�
Small piston in 40 cm above the large piston.
The
density of the liquid in the jack in given as 1000 kg/m3
Given: Dia of small piston d = 3 cm.

Related Topics