Chapter: Introduction to Human Nutrition: Energy Metabolism
Energy expenditure related to physical activity - Energy Metabolism
Energy expenditure related to physical activity
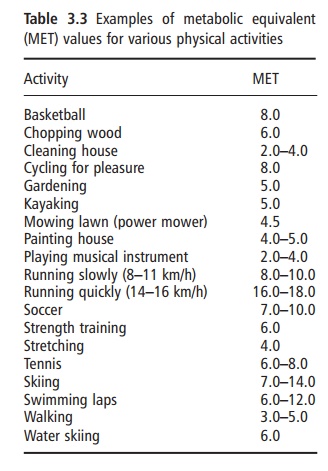
Physical activity energy expenditure encompasses all types of activity, including sports and leisure, work-related activities, general activities of daily living, and fidgeting. The metabolic rate of physical activity is determined by the amount or duration of activity (i.e., time), the type of physical activity (e.g., walking, running, typing), and the intensity at which the par-ticular activity is performed. The metabolic cost of physical activities is frequently expressed as metabolic equivalents (METs), which represent multiples of RMR. Thus, by definition, sitting quietly after a 12 hour fast is equivalent to 1 MET. Table 3.3 provides MET values for other typical physical activities. The cumulative total daily energy cost of physical activity is highly variable both within and between individuals. Therefore, physical activity provides the greatest source of plasticity or flexibility in the energy expenditure system, and is the component through which large changes in energy expenditure can be achieved.
Total energy expenditure: measurement by doubly labeled water
The integrated sum of all components of energy expenditure is termed total energy expenditure. Until recently, there was no good way to measure total energy expenditure in humans living under their habitual conditions. Total energy expenditure can be measured over 24 hours or longer in a metabolic chamber, but this environment is artificial and is not representative of the normal daily pattern of physical activity. The DLW technique can be used to obtain an integrated measure of all components of daily energy expenditure over extended periods, typically 7–14 days, while subjects are living in their usual environment. The technique was first introduced in the 1950s as an isotopic technique for measuring the carbon dioxide production rate in small animals. Unfortunately, it was not possible to apply the tech-nique to humans because the dose required was cost prohibitive given the relatively poor sensitivity of the required instrumentation at that time. It was not for another 20 years that the inventors of this technique described the feasibility of applying the technique to measure free-living energy expenditure in humans, and 10 years later this concept became a reality.
The DLW method requires a person to ingest small amounts of “heavy” water that is isotopically labeled with deuterium and oxygen-18 (2H2O and H218O). These forms of water are naturally occurring, stable (nonradioactive) isotopes of water that differ from the most abundant form of water. In deuterium-labeled water, the hydrogen is replaced with deute-rium, which is an identical form of water except that deuterium has an extra neutron in its nucleus com-pared with hydrogen, and is thus a heavier form of water; similarly, 18O-labeled water contains oxygen with an additional two extra neutrons. Thus, these stable isotopes act as molecular tags so that water can be tracked in the body. After a loading dose, deute-rium-labeled water is washed out of the body as a function of body water turnover; 18O is also lost as a function of water turnover, but is lost via carbon dioxide production as well. Therefore, using a number of assumptions, the rate of carbon dioxide produc-tion and energy expenditure can be assessed based on the different rates of loss of these isotopes from the body.
The major advantages of the DLW method are that the methodology is truly noninvasive and nonobtru-sive (subjects are entirely unaware that energy expen-diture is being measured), and measurement is performed under free-living conditions over extended periods (7–14 days). Moreover, when used in combi-nation with indirect calorimetry for assessment of resting metabolic rate, physical activity-related energy expenditure can be assessed by the difference (i.e., total energy expenditure minus resting metabolic rate, minus the thermic effect of meals = physical activity energy expenditure). The additional power of assessing total energy expenditure with the DLW method is that this approach can provide a measure of total energy intake in subjects who are in energy balance. This is because, by definition, in a state of energy balance, total energy intake must be equiva-lent to total energy expenditure. This aspect of the technique has been used as a tool to validate energy intakes using other methods such as food records and dietary recall. For example, it has been known for some time that obese subjects report a lower than expected value for energy intake. At one time it was thought that this was due to low energy requirements in the obese due to low energy expenditure and reduced physical activity. However, using DLW, it has now been established that obese subjects system-atically underreport their actual energy intake by 30– 40% and actually have a normal energy expenditure, relative to their larger body size.
The major disadvantages of the technique are the periodic nonavailability and expense of the 18O isotope (around €500–600 for a 70 kg adult), the need for and reliance on expensive equipment for analysis of samples, and that the technique is not well suited to large-scale epidemiological studies. Furthermore, although the technique can be used to obtain esti-mates of physical activity energy expenditure, it does not provide any information on physical activity pat-terns (i.e., type, duration, and intensity of physical activity periods during the day).
The DLW technique has been validated in humans in several laboratories around the world by compari-son with indirect calorimetry in adults and infants. These studies generally show the technique to be accurate to within 5–10%, relative to data derived by indirect calorimetry for subjects living in metabolic chambers. The theoretical precision of the DLW tech-nique is 3–5%. However, the experimental variability is ±12% under free-living conditions, owing to fluc-tuations in physical activity levels, and ±8% under more controlled sedentary living conditions. The good accuracy and reasonable precision of the tech-nique therefore allow the DLW method to be used as a “gold standard” measure of free-living energy expenditure in humans against which other methods can be compared.
Related Topics