Chapter: Basic & Clinical Pharmacology : Clinical Use of Antimicrobial Agents
Empiric Antimicrobial Therapy
EMPIRIC ANTIMICROBIAL THERAPY
Antimicrobial agents
are frequently used before the pathogen responsible for a particular illness or
the susceptibility to a particular antimicrobial agent is known. This use of
antimicrobial agents is called empiric (or presumptive) therapy and is based on
experience with a particular clinical entity. The usual justification for
empiric therapy is the hope that early intervention will improve the outcome;
in the best cases, this has been established by placebo-controlled,
double-blind prospective clinical trials. For example, treatment of febrile
episodes in neutropenic cancer patients with empiric antimicrobial therapy has
been demon-strated to have impressive morbidity and mortality benefits even
though the specific bacterial agent responsible for fever is deter-mined for
only a minority of such episodes.
Finally, there are
many clinical entities, such as certain epi-sodes of community-acquired
pneumonia, in which it is diffi-cult to identify a specific pathogen. In such
cases, a clinical response to empiric therapy may be an important clue to the
likely pathogen.
Frequently, the signs
and symptoms of infection diminish as a result of empiric therapy, and
microbiologic test results become available that establish a specific
microbiologic diagnosis. At the time that the pathogenic organism responsible
for the illness is identified, empiric therapy is optimally modified to definitivetherapy, which is typically
narrower in coverage and is given foran appropriate duration based on the
results of clinical trials or experience when clinical trial data are not
available.
Approach to Empiric Therapy
Initiation of empiric
therapy should follow a specific and system-atic approach.
A. Formulate a Clinical Diagnosis of Microbial Infection
Using all available
data, the clinician should determine that there is anatomic evidence of
infection (eg, pneumonia, cellulitis, sinusitis).
B. Obtain Specimens for Laboratory Examination
Examination
of stained specimens by microscopy or simple examination of an uncentrifuged
sample of urine for white blood cells and bacteria may provide important
etiologic clues in a very short time. Cultures of selected anatomic sites
(blood, sputum, urine, cerebrospinal fluid, and stool) and nonculture methods
(antigen testing, polymerase chain reaction, and serology) may also confirm
specific etiologic agents.
C. Formulate a Microbiologic Diagnosis
The history, physical
examination, and immediately available laboratory results (eg, Gram stain of
urine or sputum) may pro-vide highly specific information. For example, in a
young man with urethritis and a Gram-stained smear from the urethral meatus
demonstrating intracellular gram-negative diplococci, the most likely pathogen
is Neisseria gonorrhoeae. In the
latter instance, however, the clinician should be aware that a significant
number of patients with gonococcal urethritis have uninformative Gram stains
for the organism and that a significant number of patients with gonococcal
urethritis harbor concurrent chlamydial infection that is not demonstrated on
the Gram-stained smear.
D. Determine the Necessity for Empiric Therapy
Whether
or not to initiate empiric therapy is an important clinical decision based
partly on experience and partly on data from clini-cal trials. Empiric therapy
is indicated when there is a significant risk of serious morbidity if therapy
is withheld until a specific pathogen is detected by the clinical laboratory.
In
other settings, empiric therapy may be indicated for public health reasons
rather than for demonstrated superior outcome of therapy in a specific patient.
For example, urethritis in a young sexually active man usually requires
treatment for N gonorrhoeae and Chlamydia trachomatis despite the
absence of microbiologic confirmation at the time of diagnosis. Because the
risk of noncom-pliance with follow-up visits in this patient population may
lead to further transmission of these sexually transmitted pathogens, empiric
therapy is warranted.
E. Institute Treatment
Selection
of empiric therapy may be based on the microbiologic diagnosis or a clinical
diagnosis without available microbiologic clues. If no microbiologic
information is available, the antimicro-bial spectrum of the agent or agents
chosen must necessarily be broader, taking into account the most likely
pathogens responsible for the patient’s illness.
Choice of Antimicrobial Agent
Selection from among several drugs depends on host factors that include the following: (1) concomitant disease states (eg, AIDS, neutropenia due to the use of cytotoxic chemotherapy, organ transplantation, severe chronic liver or kidney disease) or the use of immunosuppressive medications; (2) prior adverse drug effects; (3) impaired elimination or detoxification of the drug (may be genetically predetermined but more frequently is associated with impaired renal or hepatic function due to underlying disease); (4) age of the patient; (5) pregnancy status; and (6) epidemiologic exposure (eg, exposure to a sick family member or pet, recent hospitalization, recent travel, occupational exposure, or new sexual partner).
Pharmacologic factors include (1) the kinetics of
absorption,distribution, and elimination; (2) the ability of the drug to be
delivered to the site of infection; (3) the potential toxicity of an agent; and
(4) pharmacokinetic or pharmacodynamic interactions with other drugs.
Knowledge of the susceptibility of an organism to a
specific agent in a hospital or community setting is important in the
selec-tion of empiric therapy. Pharmacokinetic differences among agents with
similar antimicrobial spectrums may be exploited to reduce the frequency of
dosing (eg, ceftriaxone may be conveniently given once every 24 hours).
Finally, increasing consideration is being given to the cost of antimicrobial
therapy, especially when multiple agents with comparable efficacy and toxicity
are available for a specific infection. Changing from intravenous to oral
antibiotics for prolonged administration can be particularly cost-effective.
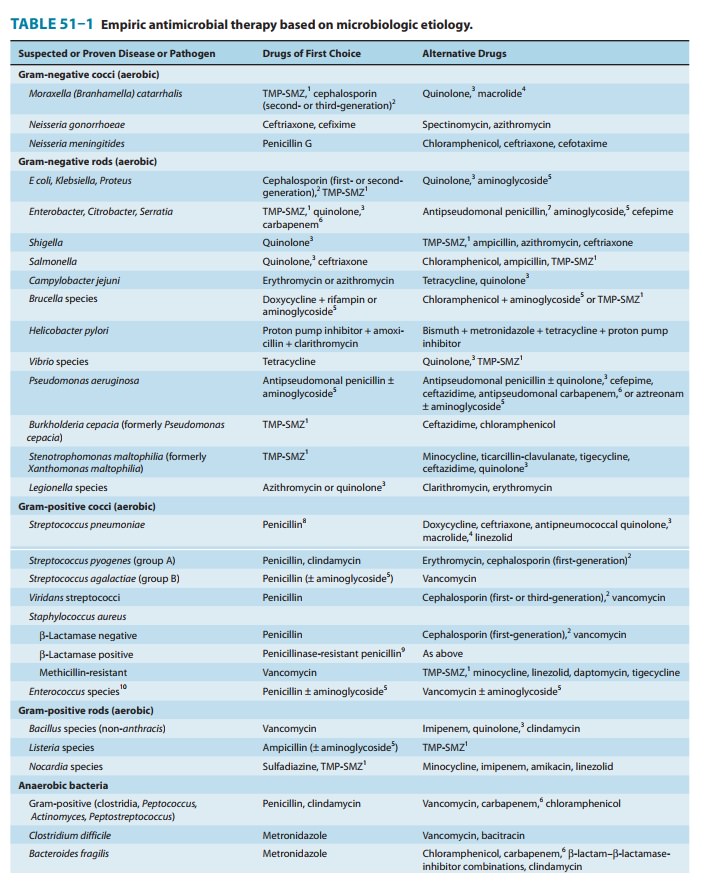
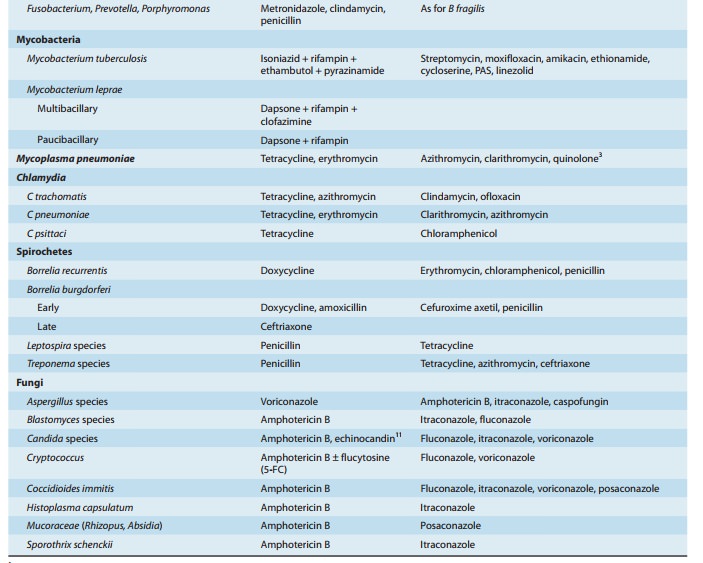
Brief guides to
empiric therapy based on presumptive microbial diagnosis and site of infection
are given in Tables 51–1 and 51–2.
Related Topics