Chapter: Essentials of Anatomy and Physiology: The Skeletal System
Embryonic Growth of Bone
EMBRYONIC GROWTH OF BONE
During embryonic development, the skeleton is first made of cartilage and fibrous connective tissue, which are gradually replaced by bone. Bone matrix is pro-duced by cells called osteoblasts (a blast cell is a “grow-ing” or “producing” cell, and osteo means “bone”). In the embryonic model of the skeleton, osteoblasts dif-ferentiate from the fibroblasts that are present. The production of bone matrix, called ossification, begins in a center of ossification in each bone.
The cranial and facial bones are first made of fibrous connective tissue. In the third month of fetal development, fibroblasts (spindle-shaped connective tissue cells) become more specialized and differentiate into osteoblasts, which produce bone matrix. From each center of ossification, bone growth radiates out-ward as calcium salts are deposited in the collagen of the model of the bone. This process is not complete at birth; a baby has areas of fibrous connective tissue remaining between the bones of the skull. These are called fontanels (Fig. 6–2), which permit com-pression of the baby’s head during birth without breaking the still thin cranial bones. The fontanels also permit the growth of the brain after birth. You may have heard fontanels referred to as “soft spots,” and indeed they are. A baby’s skull is quite fragile and must be protected from trauma. By the age of 2 years, all the fontanels have become ossified, and the skull becomes a more effective protective covering for the brain.
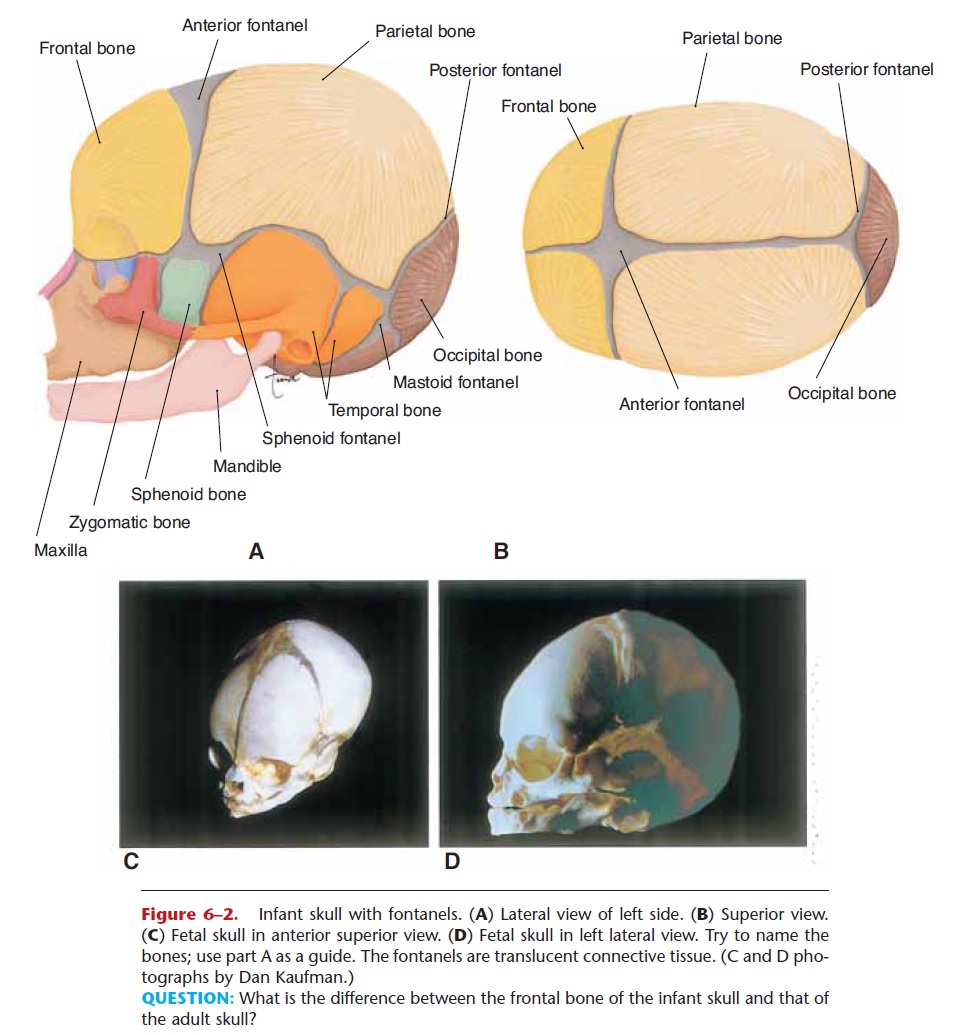
Figure 6–2. Infant skull with fontanels. (A) Lateral view of left side. (B) Superior view. (C) Fetal skull in anterior superior view. (D) Fetal skull in left lateral view. Try to name the bones; use part A as a guide. The fontanels are translucent connective tissue. (C and D pho-tographs by Dan Kaufman.)
QUESTION: What is the difference between the frontal bone of the infant skull and that of the adult skull?
The rest of the embryonic skeleton is first made of cartilage, and ossification begins in the third month of gestation in the long bones. Osteoblasts produce bone matrix in the center of the diaphyses of the long bones and in the center of short, flat, and irregular bones. Bone matrix gradually replaces the original cartilage (Fig. 6–3).
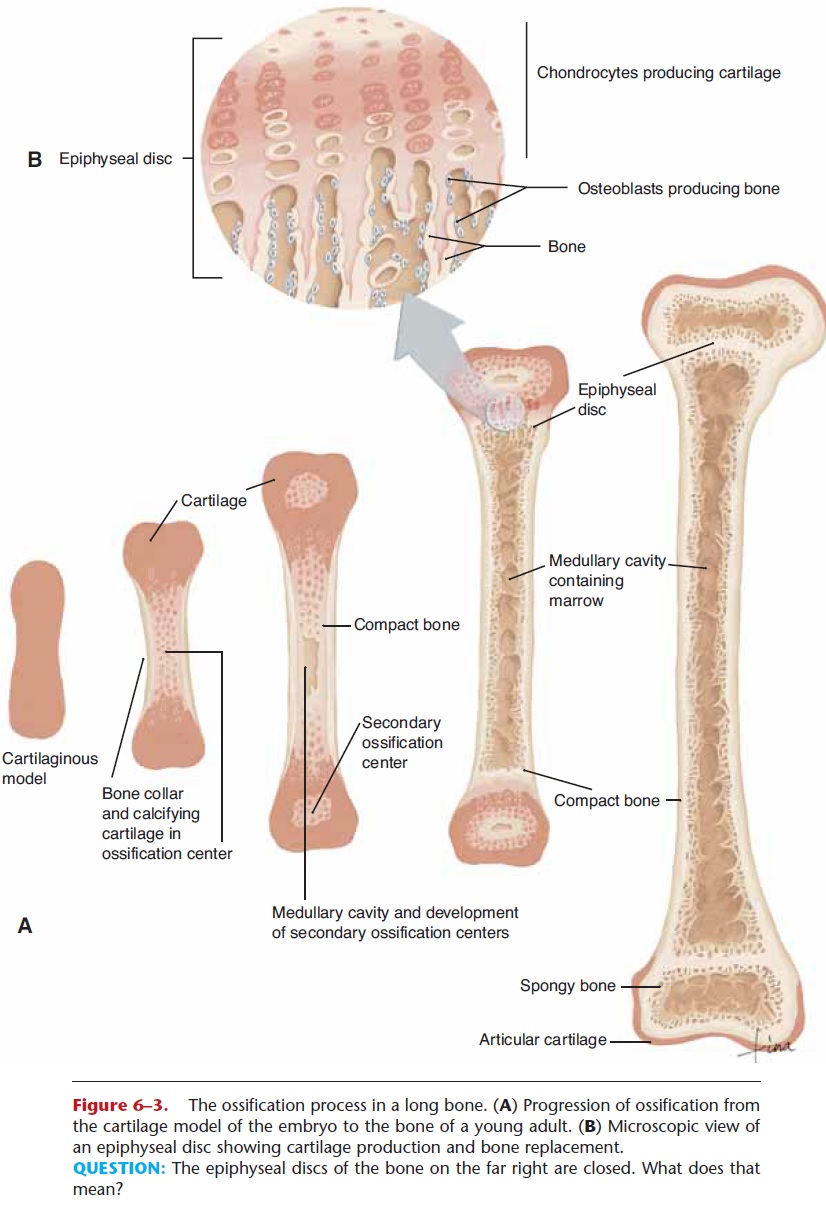
Figure 6–3. The ossification process in a long bone. (A) Progression of ossification from the cartilage model of the embryo to the bone of a young adult. (B) Microscopic view of an epiphyseal disc showing cartilage production and bone replacement.
QUESTION: The epiphyseal discs of the bone on the far right are closed. What does that mean?
The long bones also develop centers of ossification in their epiphyses. At birth, ossification is not yet com-plete and continues throughout childhood. In long bones, growth occurs in the epiphyseal discs at the junction of the diaphysis with each epiphysis. An epi-physeal disc is still cartilage, and the bone grows in length as more cartilage is produced on the epiphysis side (see Fig. 6–3). On the diaphysis side, osteoblasts produce bone matrix to replace the cartilage. Between the ages of 16 and 25 years (influenced by estrogen or testosterone), all of the cartilage of the epiphyseal discs is replaced by bone. This is called closure of the epiphyseal discs (or we say the discs are closed), and the bone lengthening process stops.
Also in bones are specialized cells called osteo-clasts (a clast cell is a “destroying” cell), which are able to dissolve and reabsorb the minerals of bone matrix, a process called resorption. Osteoclasts are very active in embryonic long bones, and they reabsorb bone matrix in the center of the diaphysis to form the marrow canal. Blood vessels grow into the marrow canals of embryonic long bones, and red bone marrow is established. After birth, the red bone marrow is replaced by yellow bone marrow. Red bone marrow remains in the spongy bone of short, flat, and irregu-lar bones. For other functions of osteoclasts and osteoblasts..
Related Topics