Chapter: Biochemical Pharmacology : Pharmacokinetics
Drug application and uptake - Pharmacokinetics
Drug application and uptake
You are certainly aware that
drugs are applied by various routes; the choice depends largely on the
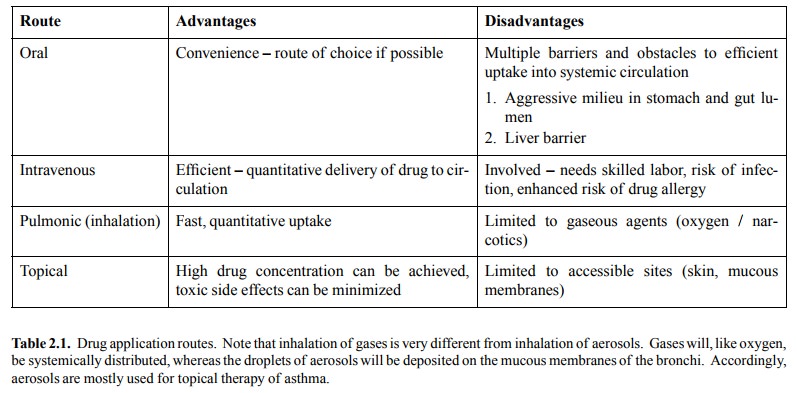
pharmacokinetic properties of the drug in question. Table 2.1 lists some
characteristics of the major routes.
We will look at the various
routes of application in turn. Oral uptake is the most common one, so let's
start with this one.
Oral drug application
Inside the digestive tract,
drug molecules encounter a quite aggressive chemical milieu. E.g., the acidic
pH in the stom-ach (pH ~2) and the presence of proteases and nucleases in the
gut preclude the application of proteins, nucleic acids, and other labile
molecules. The gut mucous membrane presents a barrier to uptake; many drugs are
not able to ef-ficiently cross it by way of diffusion.
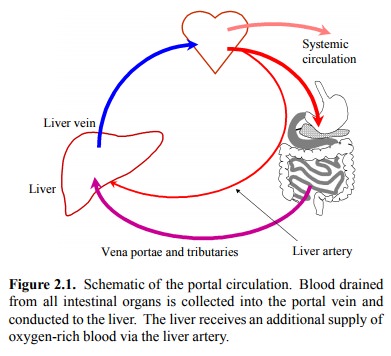
For those drugs that make it
from the gut lumen into the blood, the liver presents another formidable
barrier. All blood drained from the intestines (as well as the spleen and the
pancreas) is first passed through the liver before being released into the
general circulation. This is schematically depicted in figure 2.1.
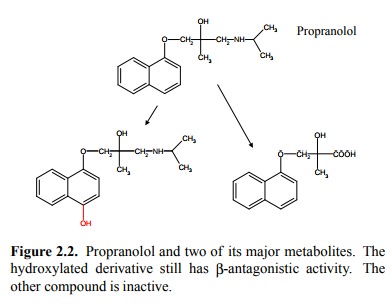
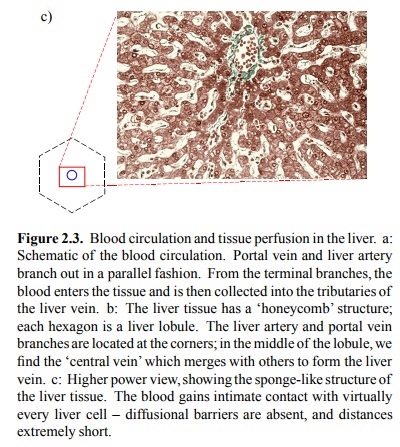
Inside the liver, the blood
leaves the terminal branches of the portal vein and the liver artery and is
filtered through the liver tissue (Figure 2.3a).
The liver tissue has a
characteristic honey-comb structure (Figure 2.3b). The individual hexagons of
the honeycomb are referred to as lobuli. The portal vein and liver artery
branches spread along the boundaries of the lobuli. The blood that leaves them
is filtered through the tissue towards the center of the lobulus, where it
reaches the central vein. The central veins then siphon the blood toward the
systemic circulation.
A notable feature of the
liver tissue is its lack of real blood vessel walls along the way from the
portal vein branches to the central veins. Therefore, the blood gets into
intimate contact with the liver epithelial cells, which therefore can very
efficiently extract from the blood any compound they see fit (Figure 2.3c).
The liver is a metabolically
very versatile organ and is ca-pable of chemically modifying a great many
substrates – including drugs – in a variety of ways and with great effi-ciency.
In fact, many drugs cannot be orally applied at all because even during the
initial passage the liver extracts them quantitatively from the portal venous
blood. This phe-nomenon is called the `first pass effect'. An example of a drug
that undergoes a substantial first-pass effect is propra-nolol (Figure 2.2).
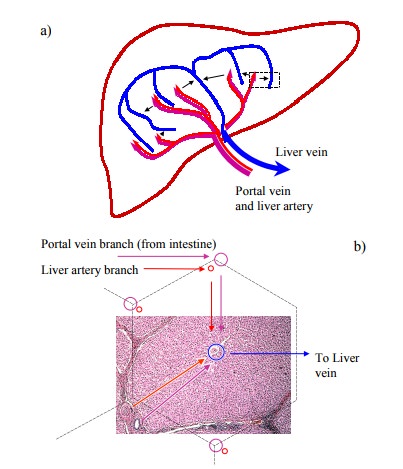
Propranolol, which blocks β-adrenergic receptors, is com-monly used in patients with
cardiovascular disease. Shown below are two metabolites. The left one
(4-hydroxypropra-nolol) is still active but not quantitatively very important.
The right one (naphthyloxymethyllactate) is entirely inac tive. Only about 30%
of the propranolol ingested actually shows up in the systemic circulation – the
rest is either not absorbed or metabolized in the liver during the first
pas-sage. The extent of this first pass effect shows considerable
inter-individual variation – which means that the required dosage may vary
considerably and has to be empirically determined with each patient. The
fraction that reaches the systemic circulation (~30% in our example) is
designated as the `bioavailability' of the drug.
To sum up: Oral application
has
• Advantages: Convenience – route of choice if
possible
• Disadvantages:
1.
Aggressive
chemical milieu in the digestive tract – precludes application of proteins,
nucleic acids..
2.
Gut
mucous membrane presents a barrier
3.
Blood
from the intestine is passed through the liver
– liver may immediately extract and metabolize
the drug (`first pass effect')
4.
Absorption
is slow (not suitable for emergency treat-ment) and variable
Intravenous drug application
With intravenous application,
we have the following advan-tages:
• `Absorption', even of large molecules, is
quantitative and instantaneous. This is essential if drug action is needed
immediately.
• Short-lived drugs can be continuously applied
by in-fusion, and the infusion rate can be controlled so as to `titrate'the
clinical effect. Examples: Muscle relaxation with succinylcholine during narcosis,
control of blood pressure in hypertonic crisis with sodium nitroprusside (both
drugs will be discussed later in this class).
• No exposure of drug to harsh conditions –
proteins can be applied this way
Disadvantages:
• Involved (needs trained professional for each
applica-tion – dangerous if not performed properly)
• Adverse reactions to drugs will be more
instantaneous and serious, too (example: penicillin allergy)
Other routes of drug
applicaton
Dermal application has two cases:
Topical application
(treatment of skin disease). No critical issues here; often preferable to
systemic therapy (high local drug concentrations, minimal side effects on the
rest of the body).
• Dermal application for systemic use.
– Uptake typically slow and
inefficient (Mother Nature gave us skin as a barrier, not as a conductor).
Notable exception: very hydrophobic compounds (organic solvents, nerve gases).
– Retarded uptake can be
utilized for sustaining pro-longed, slow delivery (example: Nicotine for
wean-ing smokers)
Mucosal application exploits
the fact that, compared to the skin, the barrier is much thinner. Moreover, the
veins un-derlying the mucous membranes in the two favorite places (nose and
rectum) are not drained into the liver – i.e., the first pass effect can be
circumvented. Examples:
1. Nose: Cocaine, antidiuretic hormone (ADH). ADH
is a peptide – so even peptides can make it across the mucosa
2. Rectum:
Acetaminophen. Rectal application
will in-
crease the bioavailability of
this drug as compared to oral uptake, because the first pass effect is absent. 1
Pulmonal application (Figure
2.4) has two modes:
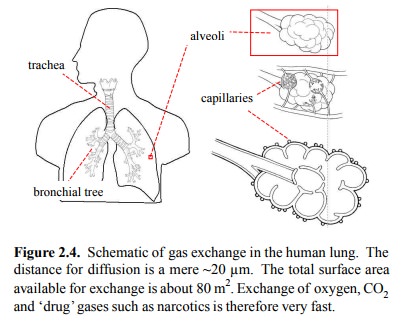
• Gaseous drugs reach the alveoli. This mainly
applies to inhalation anesthetics (chloroform, ether, N2O, and their
more modern replacements). Very rapid transition into the bloodstream – very
rapid onset of action.
• Non-gaseous drugs can be conveyed by aerosols.
The droplets are actually deposited in the bronchi but do not reach the alveoli
(topical / mucosal application). Exam ple: Steroids for asthma therapy (asthma
is an affliction of the bronchi).
Related Topics