Chapter: Medical Physiology: Physical Principles of Gas Exchange; Diffusion of Oxygen and Carbon Dioxide Through the Respiratory Membrane
Diffusion of Gases Through the Respiratory Membrane
Diffusion of Gases Through the Respiratory Membrane
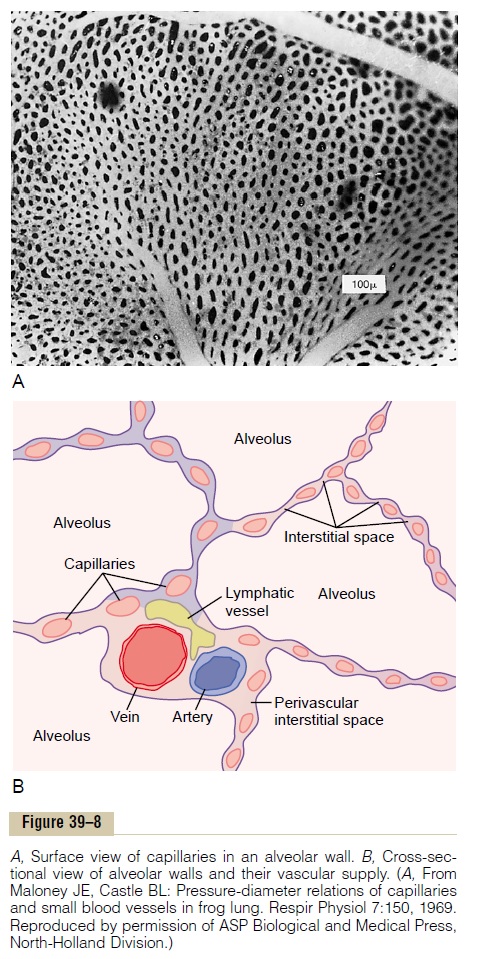
Respiratory Unit. Figure 39–7 shows therespiratory unit(also called “respiratory lobule”), which is composed of a respiratory bronchiole, alveolar ducts, atria, and alveoli. There are about 300 million alveoli in the twolungs, and each alveolus has an average diameter of about 0.2 millimeter. The alveolar walls are extremely thin, and between the alveoli is an almost solid network of interconnecting capillaries, shown in Figure 39–8. Indeed, because of the extensiveness of the capillary plexus, the flow of blood in the alveolar wall has been described as a “sheet” of flowing blood. Thus, it is obvious that the alveolar gases are in very close proximity to the blood of the pulmonary capil-laries. Further, gas exchange between the alveolar air
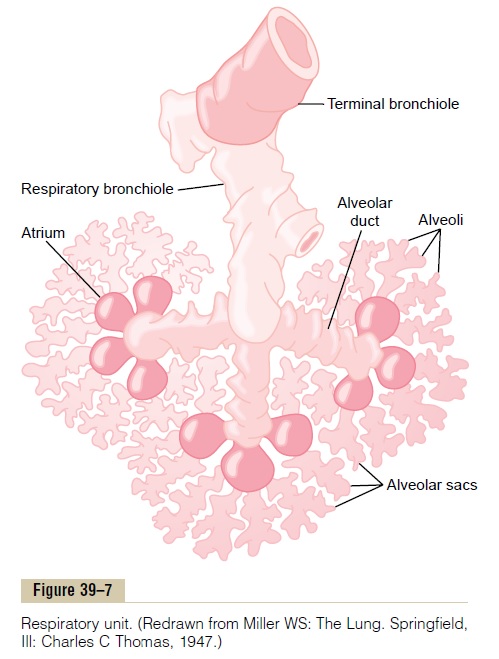
and the pulmonary blood occurs through the mem-branes of all the terminal portions of the lungs, not merely in the alveoli themselves. All these membranes are collectively known as the respiratory membrane, also called the pulmonary membrane.
Respiratory Membrane. Figure 39–9 shows the ultrastruc-ture of the respiratory membrane drawn in cross section on the left and a red blood cell on the right. It also shows the diffusion of oxygen from the alveolus into the red blood cell and diffusion of carbon dioxide in the opposite direction. Note the following different layers of the respiratory membrane:
1. A layer of fluid lining the alveolus and containing surfactant that reduces the surface tension of the alveolar fluid
2. The alveolar epithelium composed of thin epithelial cells
3. An epithelial basement membrane
4. A thin interstitial space between the alveolar epithelium and the capillary membrane
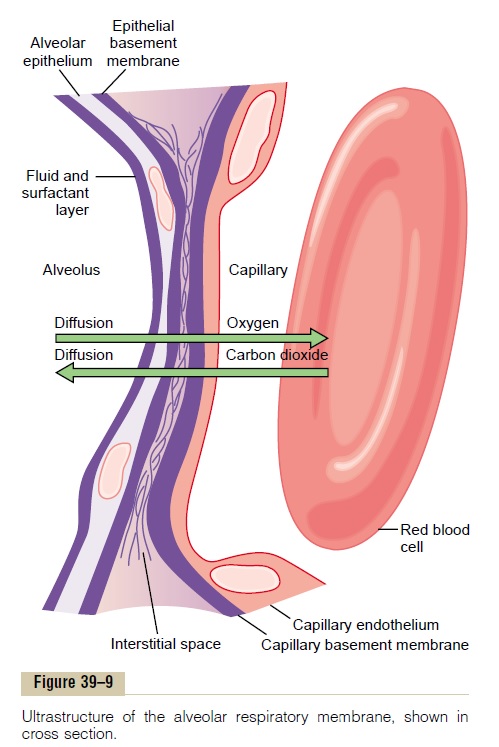
5. A capillary basement membrane that in many places fuses with the alveolar epithelial basement membrane
6. The capillary endothelial membrane
Despite the large number of layers, the overall thickness of the respiratory membrane in some areas is as little as 0.2 micrometer, and it averages about 0.6 micrometer, except where there are cell nuclei. From histological studies, it has been estimated that the total surface area of the respiratory membrane is about 70 square meters in the normal adult human male. This is equivalent to the floor area of a 25–by-30–foot room. The total quantity of blood in the capillaries of the lungs at any given instant is 60 to 140 milliliters. Now imagine this small amount of blood spread over the entire surface of a 25–by-30–foot floor, and it is easy to understand the rapidity of the respiratory exchange of oxygen and carbon dioxide.
The average diameter of the pulmonary capillaries is only about 5 micrometers, which means that red blood cells must squeeze through them. The red blood cell membrane usually touches the capillary wall, so that oxygen and carbon dioxide need not pass through significant amounts of plasma as they diffuse between the alveolus and the red cell. This, too, increases the rapidity of diffusion.
Factors That Affect the Rate of Gas Diffusion Through the Respiratory Membrane
Referring to the earlier discussion of diffusion of gases in water, one can apply the same principles and math-ematical formulas to diffusion of gases through the respiratory membrane. Thus, the factors that deter-mine how rapidly a gas will pass through the mem-brane are (1) the thickness of the membrane, (2) the surface area of the membrane, (3) the diffusion coeffi-cient of the gas in the substance of the membrane, and (4) the partial pressure difference of the gas between the two sides of the membrane.
The thickness of the respiratory membrane occa-sionally increases—for instance, as a result of edema fluid in the interstitial space of the membrane and in the alveoli—so that the respiratory gases must then diffuse not only through the membrane but also through this fluid. Also, some pulmonary diseases cause fibrosis of the lungs, which can increase the thickness of some portions of the respiratory mem-brane. Because the rate of diffusion through the mem-brane is inversely proportional to the thickness of the membrane, any factor that increases the thickness to more than two to three times normal can interfere sig-nificantly with normal respiratory exchange of gases.
The surface area of the respiratory membrane can be greatly decreased by many conditions. For instance, removal of an entire lung decreases the total surface area to one half normal. Also, in emphysema, many of the alveoli coalesce, with dissolution of many alveolar walls. Therefore, the new alveolar chambers are much larger than the original alveoli, but the total surface area of the respiratory membrane is often decreased as much as fivefold because of loss of the alveolar walls. When the total surface area is decreased to about one third to one fourth normal, exchange of gases through the membrane is impeded to a signifi-cant degree, even under resting conditions, and during competitive sports and other strenuous exercise, even the slightest decrease in surface area of the lungs can be a serious detriment to respiratory exchange of gases.
The diffusion coefficient for transfer of each gas through the respiratory membrane depends on the gas’s solubility in the membrane and, inversely, on the square root of the gas’s molecular weight. The rateof diffusion in the respiratory membrane is almost exactly the same as that in water, for reasons explained earlier. Therefore, for a given pressure difference, carbon dioxide diffuses about 20 times as rapidly as oxygen. Oxygen diffuses about twice as rapidly as nitrogen.
The pressure difference across the respiratory mem-brane is the difference between the partial pressure of the gas in the alveoli and the partial pressure of the gas in the pulmonary capillary blood. The partial pres-sure represents a measure of the total number of mol-ecules of a particular gas striking a unit area of the alveolar surface of the membrane in unit time, and the pressure of the gas in the blood represents the number of molecules that attempt to escape from the blood in the opposite direction. Therefore, the difference between these two pressures is a measure of the nettendency for the gas molecules to move through themembrane. When the partial pressure of a gas in the alveoli is greater than the pressure of the gas in the blood, as is true for oxygen, net diffusion from the alveoli into the blood occurs; when the pressure of the gas in the blood is greater than the partial pressure in the alveoli, as is true for carbon dioxide, net diffusion from the blood into the alveoli occurs.
Diffusing Capacity of the Respiratory Membrane
The ability of the respiratory membrane to exchange a gas between the alveoli and the pulmonary blood is expressed in quantitative terms by the respiratorymembrane’s diffusing capacity, which is defined asthe volume of a gas that will diffuse through the mem-brane each minute for a partial pressure difference of 1 mm Hg. All the factors discussed earlier that affectdiffusion through the respiratory membrane can affect this diffusing capacity.
Diffusing Capacity for Oxygen. In the average young man,the diffusing capacity for oxygen under resting condi-tions averages 21 ml/min/mm Hg. In functional terms, what does this mean? The mean oxygen pressure difference across the respiratory membrane during normal, quiet breathing is about 11 mm Hg. Multipli-cation of this pressure by the diffusing capacity (11 x 21) gives a total of about 230 milliliters of oxygen dif-fusing through the respiratory membrane each minute; this is equal to the rate at which the resting body uses oxygen.
Change in Oxygen Diffusing Capacity During Exercise. During strenuous exercise or other conditions that greatly increase pulmonary blood flow and alveolar ventilation, the diffusing capacity for oxygen increases in young men to a maximum of about 65 ml/min/ mm Hg, which is three times the diffusing capacity under resting conditions. This increase is caused by several factors, among which are (1) opening up of many previously dormant pulmonary capillaries or extra dilation of already open capillaries, thereby increasing the surface area of the blood into which the oxygen can diffuse; and (2) a better match between the ventilation of the alveoli and the perfusion of the alveolar capillaries with blood, called the ventilation-perfusion ratio, which is explained in detail later. Therefore, during exercise, oxygenation of the blood is increased not only by increased alveolar ventilation but also by greater diffusing capacity of the respiratory membrane for transporting oxygen into the blood.
Diffusing Capacity for Carbon Dioxide. The diffusing capac-ity for carbon dioxide has never been measured because of the following technical difficulty: Carbon dioxide diffuses through the respiratory membrane so rapidly that the average PCO2 in the pulmonary blood is not far different from the PCO2 in the alveoli—the average difference is less than 1 mm Hg—and with the available techniques, this difference is too small to be measured.
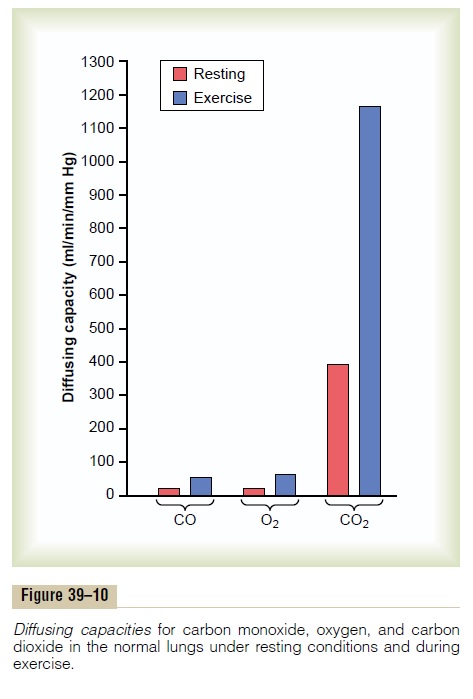
Nevertheless, measurements of diffusion of other gases have shown that the diffusing capacity varies directly with the diffusion coefficient of the particular gas. Because the diffusion coefficient of carbon dioxide is slightly more than 20 times that of oxygen, one would expect a diffusing capacity for carbon dioxide under resting conditions of about 400 to 450 ml/min/ mm Hg and during exercise of about 1200 to 1300 ml/ min/mm Hg. Figure 39–10 compares the measured or calculated diffusing capacities of carbon monoxide, oxygen, and carbon dioxide at rest and during exercise, showing the extreme diffusing capacity of carbon dioxide and the effect of exercise on the diffusing capacity of each of these gases.
MeasurementofDiffusing Capacity—The CarbonMonoxide Method. The oxygen diffusing capacity can be calculatedfrom measurements of (1) alveolar PO2, (2) PO2 in the pulmonary capillary blood, and (3) the rate of oxygen uptake by the blood. However, measuring the PO2 in the pulmonary capillary blood is so difficult and so impre-cise that it is not practical to measure oxygen diffusing capacity by such a direct procedure, except on an experimental basis.
To obviate the difficulties encountered in measuring oxygen diffusing capacity directly, physiologists usually measure carbon monoxide diffusing capacity instead and then calculate the oxygen diffusing capacity from this. The principle of the carbon monoxide method is the following: A small amount of carbon monoxide is breathed into the alveoli, and the partial pressure of the carbon monoxide in the alveoli is measured from appro-priate alveolar air samples. The carbon monoxide pres-sure in the blood is essentially zero, because hemoglobin combines with this gas so rapidly that its pressure never has time to build up. Therefore, the pressure difference of carbon monoxide across the respiratory membrane is equal to its partial pressure in the alveolar air sample. Then, by measuring the volume of carbon monoxide absorbed in a short period and dividing this by the alveolar carbon monoxide partial pressure, one can determine accurately the carbon monoxide diffusing capacity.
To convert carbon monoxide diffusing capacity to oxygen diffusing capacity, the value is multiplied by a factor of 1.23 because the diffusion coefficient for oxygen is 1.23 times that for carbon monoxide. Thus, the average diffusing capacity for carbon monoxide in young men at rest is 17 ml/min/mm Hg, and the diffusing capacity for oxygen is 1.23 times this, or 21 ml/min/mm Hg.
Related Topics