Chapter: Medical Physiology: Physical Principles of Gas Exchange; Diffusion of Oxygen and Carbon Dioxide Through the Respiratory Membrane
Composition of Alveolar Air - Its Relation to Atmospheric Air
Composition of Alveolar Air - Its Relation to Atmospheric Air
Alveolar air does not have the same concentrations of gases as atmospheric air by any means, which can readily be seen by comparing the alveolar air compo-sition in Table 39–1 with that of atmospheric air. There are several reasons for the differences. First, the alve-olar air is only partially replaced by atmospheric air with each breath. Second, oxygen is constantly being absorbed into the pulmonary blood from the alveolar air. Third, carbon dioxide is constantly diffusing from the pulmonary blood into the alveoli. And fourth, dry atmospheric air that enters the respiratory passages is humidified even before it reaches the alveoli.
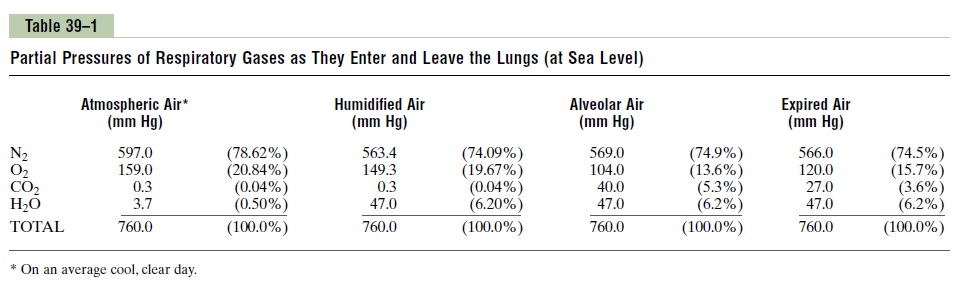
Humidification of the Air in the Respiratory Passages. Table39–1 shows that atmospheric air is composed almost entirely of nitrogen and oxygen; it normally contains almost no carbon dioxide and little water vapor. However, as soon as the atmospheric air enters the res-piratory passages, it is exposed to the fluids that cover the respiratory surfaces. Even before the air enters the alveoli, it becomes (for all practical purposes) totally humidified.
The partial pressure of water vapor at a normal body temperature of 37°C is 47 mm Hg, which is there-fore the partial pressure of water vapor in the alveo-lar air. Because the total pressure in the alveoli cannot rise to more than the atmospheric pressure (760 mm Hg at sea level), this water vapor simply dilutes all the other gases in the inspired air. Table 39–1also shows that humidification of the air dilutes the oxygen partial pressure at sea level from an average of 159 mm Hg in atmospheric air to 149 mm Hg in the humidified air, and it dilutes the nitrogen partial pres-sure from 597 to 563 mm Hg.
Rate at Which Alveolar Air Is Renewed by Atmospheric Air
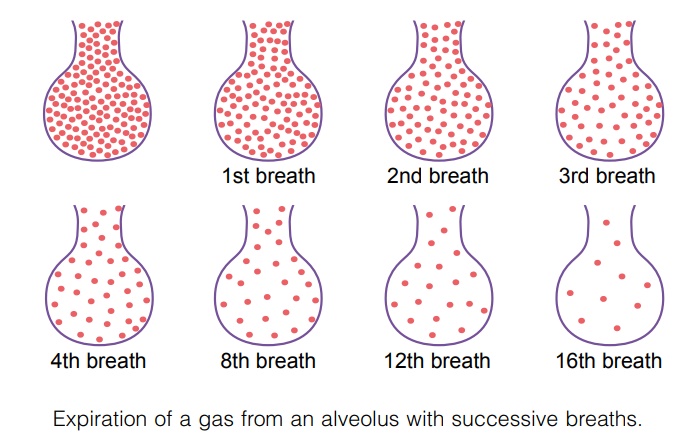
It was pointed out that the average male functional residual capacity of the lungs (the volumeof air remaining in the lungs at the end of normal expi-ration) measures about 2300 milliliters. Yet only 350 milliliters of new air is brought into the alveoli with each normal inspiration, and this same amount of old alveolar air is expired. Therefore, the volume of alve-olar air replaced by new atmospheric air with each breath is only one seventh of the total, so that multi-ple breaths are required to exchange most of the alve-olar air. Figure 39–2 shows this slow rate of renewal of the alveolar air. In the first alveolus of the figure, an excess amount of a gas is present in the alveoli, but note that even at the end of 16 breaths, the excess gas still has not been completely removed from the alveoli.
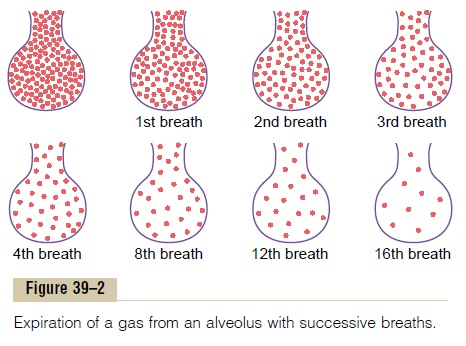
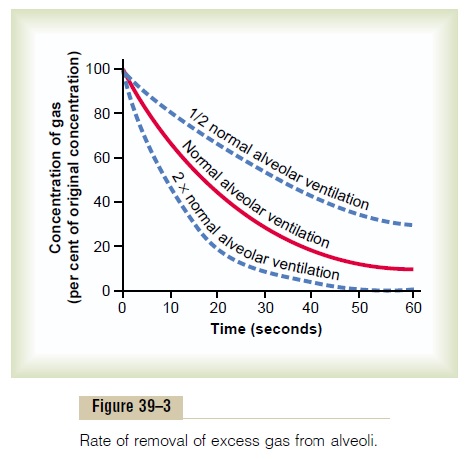
Figure 39–3 demonstrates graphically the rate at which excess gas in the alveoli is normally removed, showing that with normal alveolar ventilation, about one half the gas is removed in 17 seconds. When a person’s rate of alveolar ventilation is only one half normal, one half the gas is removed in 34 seconds, and when the rate of ventilation is twice normal, one half is removed in about 8 seconds.
Importance of the Slow Replacement of Alveolar Air. The slowreplacement of alveolar air is of particular importance in preventing sudden changes in gas concentrations in the blood. This makes the respiratory control mecha-nism much more stable than it would be otherwise, and it helps prevent excessive increases and decreases in tissue oxygenation, tissue carbon dioxide concentra-tion, and tissue pH when respiration is temporarily interrupted.
Oxygen Concentration and Partial Pressure in the Alveoli
Oxygen is continually being absorbed from the alveoli into the blood of the lungs, and new oxygen is contin-ually being breathed into the alveoli from the atmos-phere. The more rapidly oxygen is absorbed, the lower its concentration in the alveoli becomes; conversely, the more rapidly new oxygen is breathed into the alveoli from the atmosphere, the higher its concentra-tion becomes. Therefore, oxygen concentration in the alveoli, and its partial pressure as well, is controlled by
(1) the rate of absorption of oxygen into the blood and
(2) the rate of entry of new oxygen into the lungs by the ventilatory process.
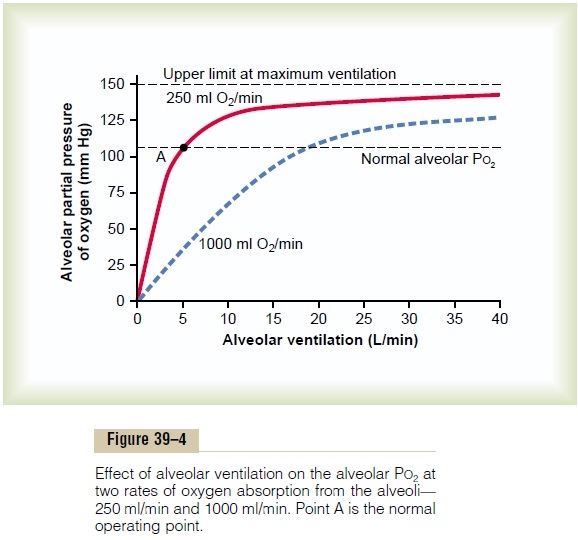
Figure 39–4 shows the effect of both alveolar venti-lation and rate of oxygen absorption into the blood on the alveolar partial pressure of oxygen (PO2). One curve represents oxygen absorption at a rate of 250 ml/min, and the other curve represents a rate of 1000 ml/min. At a normal ventilatory rate of 4.2 L/min and an oxygen consumption of 250 ml/min, the normal operating point in Figure 39–4 is point A. The figure also shows that when 1000 milliliters of oxygen is being absorbed each minute, as occurs during moderate exercise, the rate of alveolar ventilation must increase fourfold to maintain the alveolar PO2 at the normal value of 104 mm Hg.
Another effect shown in Figure 39–4 is that an extremely marked increase in alveolar ventilation can never increase the alveolar PO2above 149 mm Hg as long as the person is breathing normal atmospheric air at sea level pressure, because this is the maximum PO2 in humidified air at this pressure. If the person breathes gases that contain partial pressures of oxygen higher than 149 mm Hg, the alveolar PO2 can approach these higher pressures at high rates of ventilation.
CO2 Concentration and Partial Pressure in the Alveoli
Carbon dioxide is continually being formed in the body and then carried in the blood to the alveoli; it is continually being removed from the alveoli by venti-lation. Figure 39–5 shows the effects on the alveolar partial pressure of carbon dioxide (PCO2) of both alve-olar ventilation and two rates of carbon dioxide excre-tion, 200 and 800 ml/min. One curve represents a normal rate of carbon dioxide excretion of 200 ml/min. At the normal rate of alveolar ventilation of 4.2 L/min, the operating point for alveolar PCO2 is at point A in Figure 39–5—that is, 40 mm Hg.
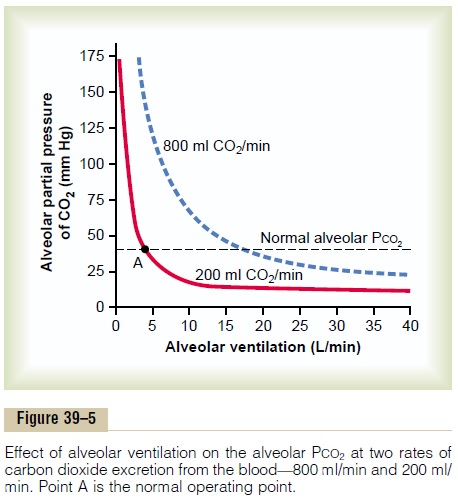
Two other facts are also evident from Figure 39–5: First, the alveolar PCO2increases directly in proportion to the rate of carbon dioxide excretion, as representedby the fourfold elevation of the curve (when 800 milliliters of CO2 are excreted per minute). Second, the alveolar PCO2 decreases in inverse proportion to alveolar ventilation. Therefore, the concentrations andpartial pressures of both oxygen and carbon dioxide in the alveoli are determined by the rates of absorption or excretion of the two gases and by the amount of alveolar ventilation.
Expired Air
Expired air is a combination of dead space air and alve-olar air; its overall composition is therefore determined by (1) the amount of the expired air that is dead space air and (2) the amount that is alveolar air. Figure 39–6 shows the progressive changes in oxygen and carbon dioxide partial pressures in the expired air during the course of expiration. The first portion of this air, the dead space air from the respiratory passageways, is typical humidified air, as shown in Table 39–1. Then, pro-gressively more and more alveolar air becomes mixed with the dead space air until all the dead space air has finally been washed out and nothing but alveolar air is expired at the end of expiration. Therefore, the method of collecting alveolar air for study is simply to collect a sample of the last portion of the expired air after forceful expiration has removed all the dead space air.
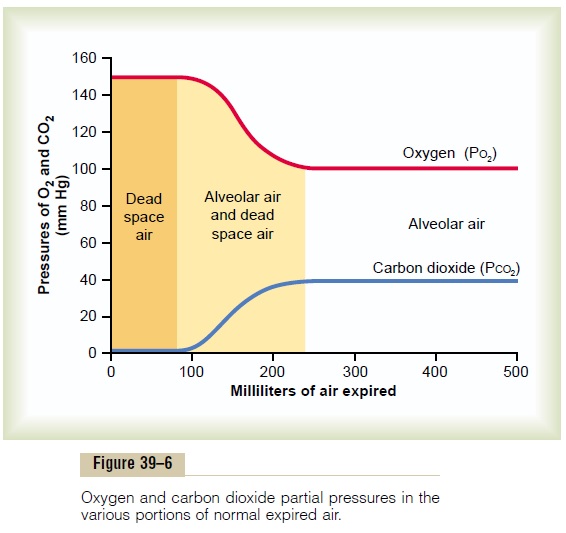
Normal expired air, containing both dead space air and alveolar air, has gas concentrations and partial pressures approximately as shown in Table 39–1—that is, concentrations between those of alveolar air and humidified atmospheric air.
Related Topics