Chapter: 11th Biochemistry : Chapter 4 : Enzymes
Coenzyme
Coenzyme
· Some of the enzymes are simple proteins. Examples:
amylase, trypsin. Many enzymes require one or more non-protein components
called cofactors. If the cofactor is an organic molecule, it is known as
coenzyme. The cofactor may also be a metal ion.
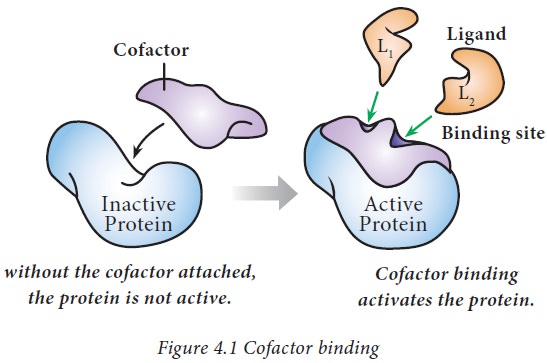
· Coenzymes may be defined as non-protein, low
molecular weight organic compounds required for the activity of enzymes.
Example: Thiamine pyrophosphate (TPP).
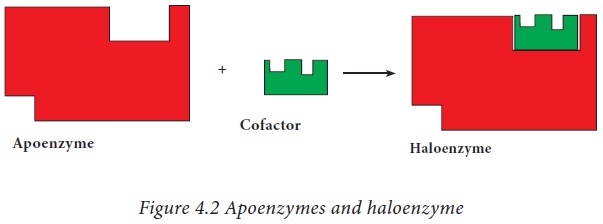
· The protein part of the enzyme is known as ‘apoenzyme’.
The entire enzyme system consisting of the apoenzyme and coenzyme (or)
prosthetic group is known as holoenzyme.
· Most of the coenzymes are linked to their
apoenzymes by noncovalent forces. Example: The coenzyme
ATP is attached to its apoenzyme hexokinase through weak non-bonding
interactions.
· Some of the coenzymes are tightly bound to their
apoenzymes through covalent bonds. These are termed as prosthetic groups.
Example: The prosthetic group biotin is attached to its apoenzyme carboxylase
through a covalent bond.
· The coenzymes undergo alterations during enzymatic
reaction. Hence the coenzymes are regarded as second substrates (or)
cosubstrates.
· Many coenzymes are the derivatives of water soluble
B-complex vitamins. Examples: Niacin.
· The Coenzymes can also be organic substances other
than vitamins. Example : ATP (adenosine triphosphate), CDP (Cytidine
diphosphate)
· Nucleotides and their derivatives can act as
coenzymes.
· Example: NAD, FMN, FAD, coenzyme-A etc.
· The specificity of an enzyme is mostly dependent on
the apoenzyme and not on the coenzyme. For example, NAD+ functions
as coenzyme for several enzymes like alcohol dehydrogenase and lactate
dehydrogenase.
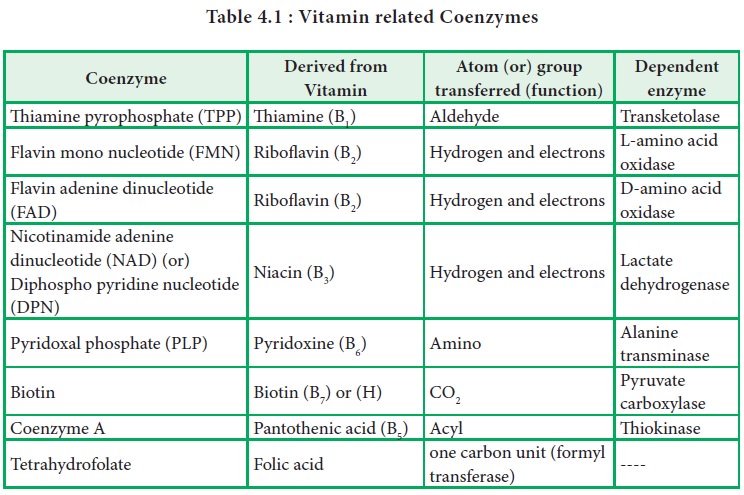
· Coenzymes functions as group transfer agents (Table
4.1).
Related Topics