Chapter: Pharmaceutical Biotechnology: Fundamentals and Applications : Recombinant Human Deoxyribonuclease I
Clinical Use- Recombinant Human Deoxyribonuclease I
CLINICAL USE
Indication and Clinical Dosage
rhDNase I (Pulmozyme dornase alpha) is currently approved for use in CF patients, in conjunction with standard therapies, to reduce the frequency of respiratory infections requiring parenteral antibiotics and to improve pulmonary function (Fig. 1). The recommended dose for use in most CF patients is one 2.5 mg dose inhaled daily using a tested, recom-mended nebulizer.
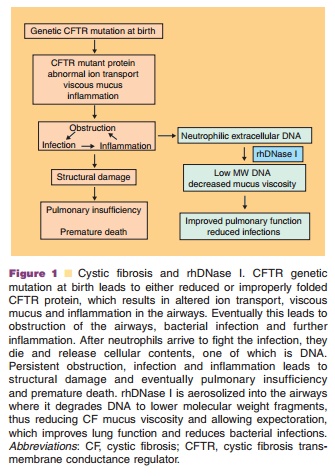
Cystic Fibrosis
rhDNase I has been evaluated in a large, randomized, placebo-controlled trial of clinically stable CF patients, 5 years of age or older, with baseline forced vital capacity (FVC) greater than or equal to 40% of predicted (Fuchs et al., 1994). All patients received additional standard therapies for CF. Patients were treated with placebo or 2.5 mg of rhDNase I once or twice a day for six months. When compared to placebo, both once daily and twice daily doses of
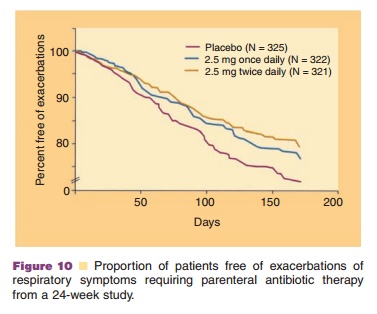
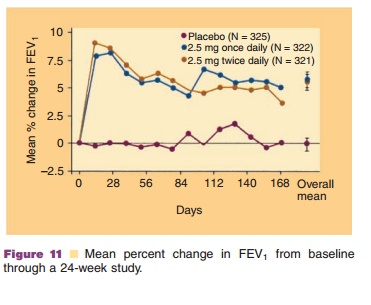
rhDNase I resulted in a 28% to 37% reduction in respiratory tract infections requiring use of parenteral antibiotics (Fig. 10). Within eight days of the start of treatment with rhDNase I, mean forced expiratory volume in one second (FEV1) increased 7.9% in patients treated once a day and 9.0% in those treated twice a day compared to the baseline values. The mean FEV1 observed during long-term therapy increased 5.8% from baseline at the 2.5 mg daily dose level and 5.6% from baseline at the 2.5 mg twice daily dose level (Fig. 11). The risk of respiratory tract infection was reduced even in patients whose pul-monary function (FEV1) was not improved. This finding is thought to result from improved clearance of mucus from the small airways in the lung, which may have little effect on FEV1 or FVC, but may significantly reduce the risk of exacerbations of infection (Shak, 1995). The administration of rhDNase I also lessened shortness of breath, increased the general perception of well-being, and reduced the severity of other CF-related symptoms.
rhDNase I did not produce a pulmonary func-tion benefit in short-term usage in the most severely ill CF patients (FVC less than 40% of predicted) (Shak, 1995; McCoy et al., 1996). These patients with end-stage lung disease represent approximately 7% of the CF population. Many are being prepared for lung transplantation but die while still awaiting an organ due to the shortage of donors. Studies are in progress to assess the impact of chronic usage on pulmonary function and infection risk in this population.
Since rhDNase I therapy may provide clinical benefit for young CF patients with mild disease by slowing progression of the disease, a study was done to investigate the safety and deposition of rhDNase I in the airways of CF patients <5 years old (3 months–5 years) compared with patients 5 to 10 years of age (Wagener et al., 1998). After two weeks of daily administration of 2.5 mg rhDNase I, comparable levels of rhDNase I were deposited in the lower airways of both age groups. Moreover, rhDNase I was well tolerated in the younger age group with an adverse event frequency similar to that in the older age group. Children with mild CF related lung disease were treated for 2 years in a randomized controlled trial to assess the effect of rhDNase I in early disease (Quan et al., 2001). Children with a mean age of 8.4 years and an FVC greater than 85% of predicted (Ramsey et al., 1999) were treated once daily with either placebo or 2.5 mg rhDNase I. After 96 weeks lung function was significantly better in the treated group compared with placebo, particularly for tests measuring function of smaller airways. Respiratory exacerbations were also reduced in the treated group.
Clinical trials have indicated that rhDNase I therapy can be continued or initiated during an acute respiratory exacerbation (Wilmott et al., 1996). Short-term dose ranging studies demonstrated that doses in excess of 2.5 mg twice daily did not provide further significant improvement in FEV1(Aitken et al., 1992; Hubbard et al., 1992; Ramsey et al., 1993). Patients who have received drug on a cyclical regimen (i.e., administration of rhDNase I 10 mg twice daily for 14 days, followed by a 14-day washout period) showed rapid improvement in FEV1 with the initiation of each cycle and a return to baseline with each rhDNase I withdrawal (Eisenberg et al., 1997).
Concomitant therapy with rhDNase I and other standard CF therapies often show additive effects. The intermittent administration of aerosolized tobramycin was approved for use in CF patients with or without concomitant use of rhDNase I (Ramsey et al., 1999). Aerosolized tobramycin was well tolerated, enhanced pulmonary function and decreased the density of Pseudomonas aeruginosa in sputum. In combination withrhDNase I a larger treatment effect was noted but did not reach statistical significance. No differences insafety profile were observed following aerosolized tobramycin in patients that did or did not use rhDNase I. Chronic use of azithromycin has also been studied in CF patients chronically infected with P. aeruginosa (Saiman et al., 2005). Similar improvement in lung function and reduction in respiratory exacerbations was seen in patients receiving rhDNase I as those not, suggesting an additive, but not synergistic, benefit of the two therapies used together. The combination of hypertonic saline therapy with chronic use of rhDNase I has similar additive benefit (Elkins et al., 2006), in agreement with the previously mentioned in vitro studies. Additionally there was no evidence of a change in adverse events related to combination therapy in either of these studies.
Non-CF Respiratory Disease
Although originally considered beneficial for the treatment of non-CF related bronchiectasis (Wills et al., 1996), rhDNase I had no effect on pulmonary function or the frequency of respiratory exacerbations in a randomized controlled trial (O’Donnell et al., 1998). In another randomized controlled trial of rhDNase I, young children had shorter periods of ventilatory support following cardiac surgery when rhDNase I was instilled twice daily into the endo-tracheal tube (Riethmueller et al., 2006). Complicating atelectasis was less frequent in the treated group, consistent with numerous case reports suggesting that rhDNase I decreases, and can be used to treat, atelectasis when directly instilled into the airway (Hendriks et al., 2005). Finally, limited benefit has been described in children with asthma (Puterman and Weinberg, 1997) or with bronchiolitis due to respiratory syncytial virus infection (Nasr et al., 2001).
Systemic Lupus Erythematosus
The presence of antibodies to nuclear antigens including DNA is a hallmark of SLE, an autoimmune disease that affects multiple organ systems; antibo-dies directed against double-stranded DNA appear to play a prominent role in lupus nephritis and are generally present at elevated serum levels in clini-cally active disease. The observation that DNase I-deficient mice develop a lupus-like syndrome sup-ports the hypothesis that a reduction in serum DNase may be a factor in the initiation of SLE (Napirei et al., 2000). Therefore, systemic adminis-tration of rhDNase I may be effective in the treatment of SLE (Lachmann, 2003). Degradation of extracellular DNA or the DNA component of DNA/ anti-DNA immune complexes may be of clinical benefit in SLE by decreasing inflammation in tissues or by reducing the antigen load leading to a decrease in production of antibodies to DNA. A clinical study demonstrated that systemic administration ofrhDNase I was well tolerated and did not induce the production of antibodies to rhDNase I (Davis et al., 1999). However, serum concentrations of rhDNase I were insufficient for catalytic activity of rhDNase I in serum (Prince et al., 1998). In a murine model of SLE, an actin/salt-resistant hyperactive DNase I variant showed significant protection from the development of anti-ssDNA and anti-histone antibodies, but not of renal disease (Manderson et al., 2006). Additional studies utilizing higher doses or more potent versions of rhDNase I variants are needed to determine whether systemic administration has any clinical benefit in SLE.
Other Medical Conditions
In principle rhDNase I may be useful for treating any condition where high levels of extracellular DNA and associated viscoelastic properties are pathological. A number of other clinical diseases with high potential for this have been investigated, although only to a limited extent. Pulmonary empyema involves the collection of purulent material in the pleural space and the use of rhDNase I instilled into the pleural space has been proposed (Simpson et al., 2003). rhDNase I has also been instilled into the nasal sinuses after surgery for chronic infections (Cimmino et al., 2005; Raynor et al., 2000).
Safety
The administration of rhDNase I has not been associated with an increase in major adverse events. Most adverse events were not more common with rhDNase I than with placebo treatment and probably reflect complications related to the underlying lung disease. Most events associated with dosing were mild, transient in nature, and did not require alterations in dosing. Observed symptoms included hoarseness, pharyngitis, laryngitis, rash, chest pain, and conjuncti-vitis. Within all the studies a small percentage (average 2–4%) of patients treated with rhDNase I developed serum antibodies to rhDNase I. None of these patients developed anaphylaxis, and the clinical significance of serum antibodies to rhDNase I is unknown.
Related Topics