Chapter: Biochemistry: Enzymes
Classification of enzymes
Classification of enzymes
The most comprehensive system for the
classification of enzymes was devised in 1961 by the Enzyme Commission of
International Union of Biochemistry (IUB).
The 6 major classes of enzymes are
1. oxidoreductases
Enzymes catalyzing oxidoreduction reactions
between two substrates A and B are called as oxidoreductases
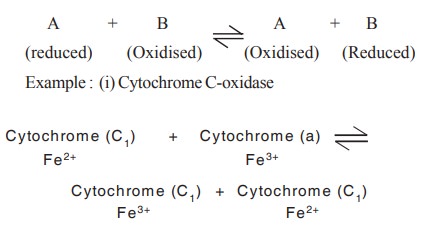
In this reaction cytochrome C1 is
oxidised and cytochrome a is reduced simultaneously by the action of cytochrome
C-oxidase.
This class includes several subclasses based on
the group on which the enzymes act. The enzymes acting on
-CH -OH (1.1)
-C=O (1.2)
-C=CH (1.3)
-CH-NH2 (1.4)
-CH-NH (1.5)
2. Transferases
Enzymes catalysing the transfer of a group (x)
from one substrate(AX ) to another are known as transferases.
AX + B < - - > A’ + BX
Example
The reaction catalysed by alanine transaminase
(ALT) is
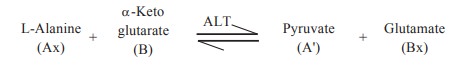
In this reaction the amino group from alanine
is transferred to α-ketoglutarate to form glutamate.
These enzymes are further divided into
subclasses on the basis of nature of the group transferred.
Transfer of
·
one
carbon compounds (2.1)
·
aldehyde
or ketonic groups (2.2)
·
acyl
groups (2.3)
·
glycosyl
groups (2.4)
·
Phosphate
groups (2.7)
·
Sulphur
containing groups (2.8)
3. Hydrolases
The hydrolases are those enzymes which catalyse
hydrolysis reactions i.e the direct addition of water molecule (s) across the
bond, which is to be cleaved. The substrate for these enzymes are esters,
ethers, peptides and glycosides.
Example : Pepsin. This enzyme is a gastro
intestinal enzyme which is proteolytic in nature and involve in the hydrolysis
of proteins present in the food.
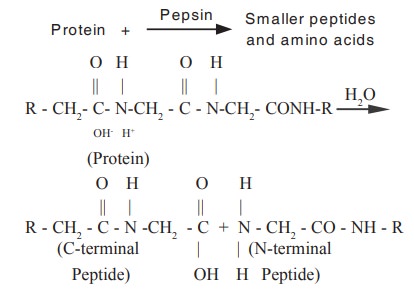
The hydrolases are divided into several
subclasses, depending on the nature of the group or bond being hydrolysed viz.,
(a) esterases etc. - hydrolyse ester bonds (3.1)
(b) glycosidases - hydrolyse glycosidic bonds (3.2)
(c) peptidases - hydrolyse peptide bonds (3.4)
4. Lyases
The lyases are a smaller class of enzymes that
catalyse the removal of a small molecule from a larger substrate molecule.
Since the reactions are reversible, lyases may also be considered to catalyse
the addition of small molecules to the substrate molecule
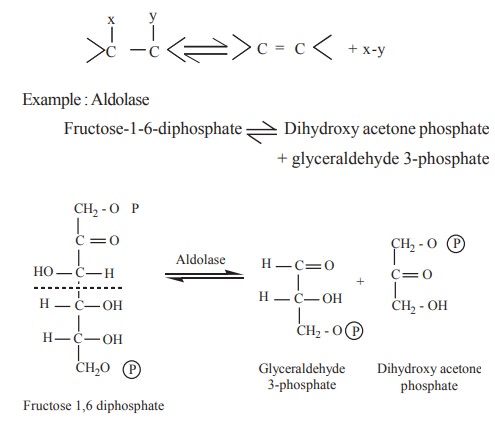
The lyases are further classified on the basis of the linkage they attack viz., acting on
a. C-C bond (4.1)
b. C-O bond (4.2)
c. C-N bond (4.3)
d. C-S bond (4.4)
e. C-halide bond (4.5)
5. Isomerases
This class includes all enzymes which catalyse
isomerization reactions i.e. interconversion of optical, geometrical or
position isomers.
Example
Retinene isomerase catalyses the conversion of
trans-retinene < - - > cis-retinene
Triose phosphate isomerase catalyses the
conversion of
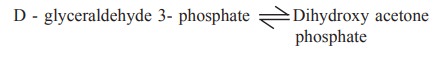
D - glyceraldehyde 3 phosphate < - - - > Dihydroxy
acetone phosphate
The isomerases are further divided into the
subclasses
a. racemases
b. epimerases
c. cis-trans isomerases
6. Ligases
These enzymes are otherwise known as
synthetases. They catalyse synthesis reactions by joining two molecules,
coupled with the breakdown of a phosphate bond of adenosine triphosphate. ATP
cleavage provides energy for the new bond formation.
Example : Formation of malonyl CoA from acetyl
CoA in the presence of acetyl CoA carboxylase.
ATP + acetyl CoA + CO2 < - - >
ADP + Pi + malonyl CoA
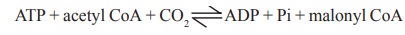
The subclasses of ligases are based on the
nature of bond formed in the product. Formation of
a. C-O bond
b. C-S bond
c. C-N bond
d. C-C bond
Class, subclass and enzyme numbers of some enzymes
1. Alcohol dehydrogenase (ADH)
Major class - oxidoreductase (1)
Subclass - acting on CH-OH (1)
Sub subclass - NAD or NADP as coenzymes (1)
Enzyme no - 1.1.1
2. Hexo kinase
Major class - Transferases (2)
Sub class - Transfering phosphate group (7)
Enzyme No. - 2.7.
3. Alkaline phosphatase
Major class Hydrolases (3)
Subclass - acting on esterbond (1)
Sub subclass - acting on phosphoric monoesters
(3)
Enzyme No. - 3.1.3
4. Fumarase
Major class - lyases (4)
Sub class - CO lyase (2)
Sub subclass - acting on aldehyde (1)
Enzyme No. - 4.2.1
5. Ribulose - 5 - phosphate epimerase
Major class - Isomerases (5)
Subclass - Racemases and Epimerases (1)
Sub subclass - acting on carbohydrates (3)
Enzyme no - 5.1.3
6. Acetyl CoA carboxylase
Major class - ligases (6)
Sub class - forming C-C bond (4)
Sub subclass - carboxyl group (1)
Enzyme No. - 6.4.1
Related Topics