Chapter: Biochemistry: Enzymes
Chemical nature of enzymes
Chemical nature of enzymes
All the enzymes are proteins with no exception.
Some enzymes are simple proteins i.e. their molecules consist of only amino
acids. Many enzymes are conjugated proteins and their molecules consist of
aminoacids and a non-protein part (not made up of amino acids). The non-protein
part of the enzyme is known as coenzyme or prosthetic group, without which the
enzyme is inactive. The protein part of the enzyme (in conjugated type) is
known as the apoenzyme. The coenzyme and the apoenzyme complex is called as
holoenzyme.
Coenzyme + Apoenzyme - - > Holoenzyme
For example, pyruvate decarboxylase is an enzyme
which catalyses the decarboxylation of pyruvate to form acetaldehyde. The
non-protein part of the enzyme is thiamine pyro phosphate without which the
reaction can not be proceeded.

The non-proteinaceous part is either loosely or
tightly bound with the apoenzyme. If the coenzymes are tightly bound with the
apoenzymes the coenzymes are called as prosthetic groups. The losely attached
coenzymes are simply called as coenzymes. A biologically active enzyme exist in
3-dimensional folded structure (conformation).
Active site
A restricted region of the enzyme to which the
substrate comes and binds and concernes with the process of catalysis is called
as the active site. In some enzymes, the active site is a deep groove into
which the substrate binds. Specific amino acids are present in the active site
which are responsible for the catalytic action. These amino acids are called as
‘catalytic’ or ‘active’ amino acids. For example lysozyme has glutamic acid and
aspartic acid as catalytic aminoacids. Chymotrypsin which is a proteolytic
enzyme contains serine and histidine as catalytic amino acids.
The active site and the other part of the
enzyme undergo conformational modification when they come in contact with the
substrate
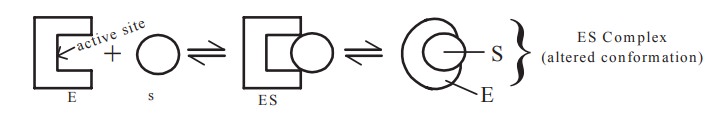
Koshland’s induced fit hypothesis of enzyme-substrate
interaction postulates that the active site of the enzyme consists of a number
of ‘active’ contact amino acids which permit the substrate to come close to the
reactive groups of the enzyme which thereupon undergoes a conformational change,
binding the substrate firmly to the enzyme and promoting catalytic activity.
Related Topics