Chapter: High Voltage Engineering : Electrical breakdown is gases, solids& Liquids
Classical Gas Laws
CLASSICAL GAS LAWS
In the
absence of electric or magnetic fields charged particles in weakly ionized
gases participate in molecular collisions. Their motions follow closely the
classical kinetic gas theory.
The
oldest gas law established experimentally by Boyle and Mariotte states that for
a given amount of enclosed gas at a constant temperature the product of
pressure (p) and volume (V) is constant or
pV = C =
const. 2.1
In the
same system, if the pressure is kept constant, then the volumes V and V0
are related to their absolute temperatures T and T0 (in K) by
Gay–Lussac’s law:

When
temperatures are expressed in degrees Celsius, eqn (2.2) becomes;
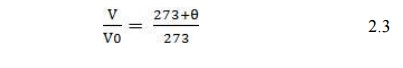
Equation
(2.3) suggests that as we approach Ɵ = -273°C
the volume of gas shrinks to zero. In reality, all gases liquefy before
reaching this value.
According
to eqn (2.2) the constant C in eqn (2.1) is related to a given temperature T0
for the volume V0:
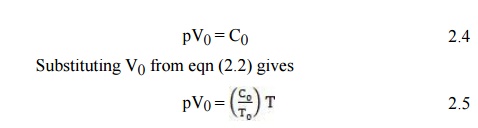
The ratio
C0/T0_ is called the universal gas constant and is
denoted by R. Equation (2.5) then becomes
pV = RT =
C 2.6
Numerically
R is equal to 8.314 joules/°Kmol. If we take n as the number of moles, i.e. the
mass m of the gas divided by it’s mol-mass, then for the general case eqn (2.1)
takes the form
pV = nC =
nRT, 2.7
Equation
(2.7) then describes the state of an ideal gas, since we assumed that R is a
constant
independent of the nature of the gas. Equation (2.7) may be written in terms of
gas density N in volume V containing N1 molecules.
Putting N
= NA where NA = 6.02 * 1023 molecules/mole, NA
is known as the Avogadro’s number. Then eqn (2.7) becomes
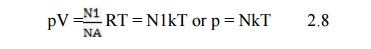
The
constant k = R/NA is the universal Boltzmann’s constant (=1.3804 *1023
joules/°K) and N is the number of molecules in the gas.
The
fundamental equation for the kinetic theory of gas is derived with the
following assumed conditions:
·
Gas consists of molecules of the same mass which
are assumed spheres.
·
Molecules are in continuous random motion.
·
Collisions are elastic – simple mechanical.
·
Mean distance between molecules is much greater
than their diameter.
Related Topics