Chapter: Obstetrics and Gynecology: Disorders of the Breast
Breast Cancer
BREAST CANCER
Breast
cancer is the second most common malignancy in women, ranking only behind skin
cancer. In addition, it is the secondleading cause of
cancer-related death in women. According to the National Cancer Institute,
there were 178,500 new cases and approximately 40,500 deaths related to breast
cancer in 2007. The steady increase in the incidence of breast cancer can be
attributed to the increased use of mammography screening, which has enabled the
detection of smaller invasive lesions and the earlier diagnosis of in-situ
lesions. Advances in treatment have also helped maintain the downward trend in
overall breast cancer mortality.
Nevertheless, breast cancer is a
serious health con-cern in the United States. It is estimated that the United
States spends approximately $8.1 billion annually on treat-ing breast cancer.
The lifetime risk of developing breast cancer in the United States is
approximately 12.5% (1 in 8), while the lifetime risk of dying from breast
cancer is 3.6% (1 in 28).
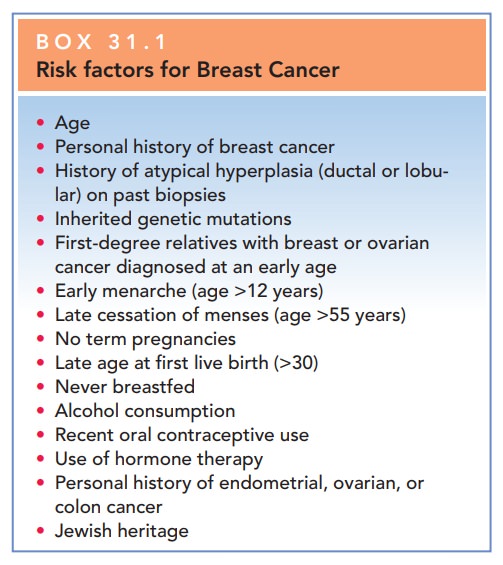
Risks Factors
Numerous studies have documented
factors that increase the relative risk for breast cancer (see Box 31.1).
AGE AND RACE
Age is the single largest risk factor for developing breast cancer.A majority of breast cancer cases occur in women over the age of 50. Stratified studies relate risk with age (by decades), and show that the risk for developing breast cancer increases as a woman gets older. For example, a woman has a 1.4% chance of being diagnosed with breast cancer between the ages of 40 and 49, compared to 3.7% between the ages of 60 and 69. When stratified by race, white women are more likely to be diagnosed with breast cancer compared to age-matched women of Latin, Asian, or African American descent.
FAMILY HISTORY AND GENETICS
Women who have first-degree
relatives (parent, sibling, or offspring) with breast cancer have a higher risk
than the general population.
If a
woman before the age of 40 is diagnosed with breast cancer, evaluating for
genetic mutations that predispose individuals to cancer is reasonable.
The two most commonly discussed
genetic mutations linked to breast cancer are the BRCA1 and BRCA2 gene
mutations.
BRCA1 is a gene located on the 17q21 chromosome.This mutation is associated with nearly half of the early-onset breast cancers and approximately 90% of hereditary ovarian cancers. BRCA2 is a gene located on the 13q12–13 chromosome. This mutation has a lower incidence of early-onset breast cancers (35%) and much lower risk of ovarian cancer, compared to BRCA1.
REPRODUCTIVE AND MENSTRUAL HISTORY
In general, women who have an
early age of menstrual onset (before the age of 12) and transition through
menopause after age 55 are at increased risk of breast cancer. Delayed
childbearing and nulliparity also increase the chance of breast cancer.
RADIATION EXPOSURE
Breast tissue of young women (along with the bone mar-row and infant thyroid) is highly susceptible to the cancer-causing effects of ionizing radiation. Women who have received a sufficiently large dose of radiation (radiation therapy to treat Hodgkin disease or an enlarged thymus gland) are at risk for radiation-induced breast cancer. The relationship between dose of radiation and risk of cancer is directly linear, although the threshold is unclear. Thus far, epidemiologic studies have not detected a significant increase in cancer risk below a cumulative dose of about 20 cGy. To put this dose into perspective, a typical mam-mogram results in a breast tissue dose of about 0.3 cGy. The time needed for a radiation-induced lesion to develop is about 5 to 10 years from exposure.
BREAST CHANGES
It is believed that women with
dense breast tissue are at increased risk for breast cancer. In addition,
histologic biopsies finding atypical hyperplasia or lobular carcinoma in situ
greatly increase the risk for breast cancer.
OTHER FACTORS
Being
overweight after menopause has been linked to an increased risk for breast
cancer. A possible mechanism in thisrelationship is that
the increased peripheral conversion of androstenedione to estrone stimulates
breast cancer development. Lack of exercise throughout life is linked to the
increased risk of breast cancer through the associated risk of obesity.
Women who
consume 2 to 4 alcoholic drinks per week have a 30% greater risk of dying from
breast cancer than women who never drink. The
exact mechanism of action is unclear, butresearchers speculate that alcohol
consumption stimulates the growth and progression of breast cancer by inducing
angiogenesis and increasing the expression of vascular endothelial growth
factor (VEGF).
Breast Cancer Risk Assessment Tool: The Gail Model
The National Cancer Institute
(NCI) has developed a computer-based tool to allow clinicians to estimate a
woman’s risk of developing invasive breast cancer over the next 5 years and in
their lifetime (up to age 90). Thetool is
based on a mathematical model of breast cancer risk cal-culation called the
Gail Model. Seven risk factors are usedin the calculations: a history of
LCIS or DCIS, age, age at onset of menstruation, age at time of the first live
birth, number of first-degree relatives with breast can-cer, history of breast
biopsy, and race/ethnicity. The use-fulness of the Gail model is limited in
patients with second-degree relatives with breast cancer (eg, paternal
transmission) and is falsely increased in patients with multiple breast biopsies.
Women at high risk, defined as a 5-year risk of 1.7% or more, can be referred for possible prophylactic therapy. Current prophylactic options include chemoprevention with the selective estrogen receptor modulators tamoxifen and raloxifene, and prophylactic mastectomy. Since all of the options areassociated with significant side effects, individualized risk assessment should be performed to determine whether a patient is a candidate for breast cancer risk reduction and, if so, which option is best.
Histologic Types of Breast Cancer
Malignant tumors of the breast
may arise from any of the major components of the breast. The American JointCommittee on Cancer classifies most breast
malignancies into one of three histologic categories according to their correspond-ing
cells of origin: ductal, lobular, and nipple. Seventy toeighty percent of
breast cancers are invasive ductal carcino-mas. These are most common among
women in their 50s and have a tendency to spread to regional lymph nodes.
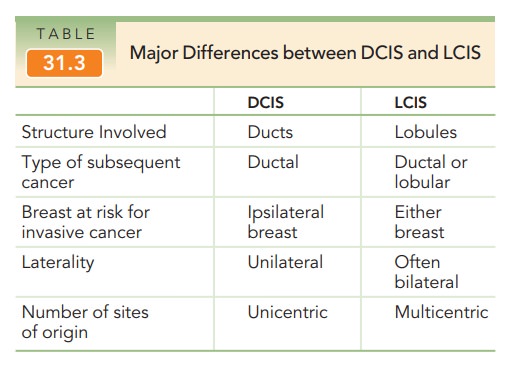
Invasive lobular carcinomas comprise 5% to 50% of breast cancers. This type is
often multifocal and bilat-eral. Table 31.3 summarizes the differences between
the two processes. Paget disease of the nipple presents as a superficial skin
lesion similar to eczema.
Breast Cancer Staging
The American Joint Committee on Cancer stages breast malignancies according to the TNM system that describes characteristics of the primary tumor, involvement of regional lymph nodes, and distant metastasis. Surgical stage helps determine the appropriate types of therapy (Tables 31.4 and 31.5).

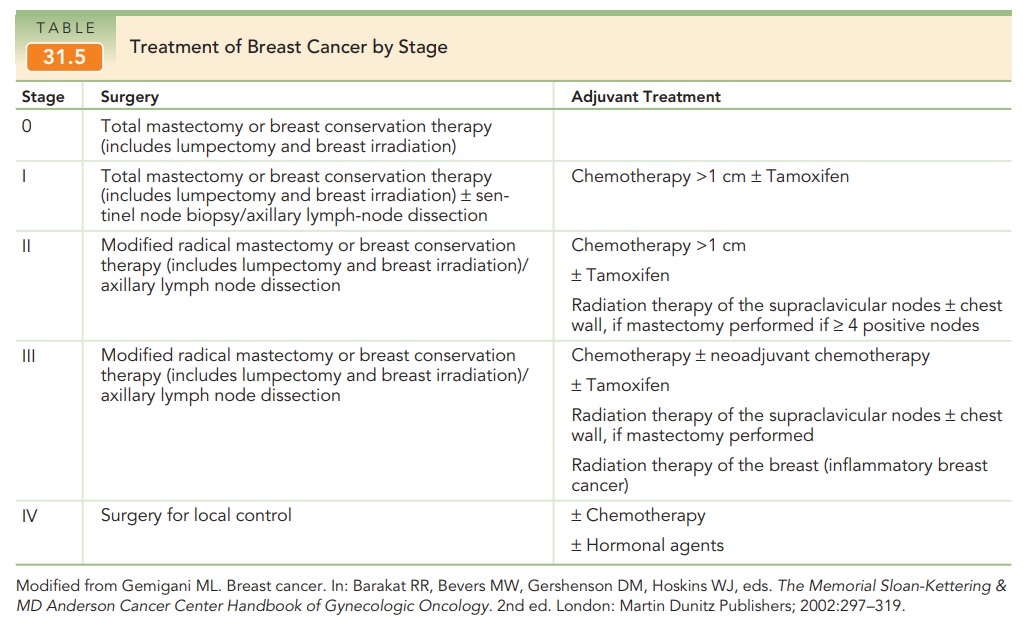
In addition to stage, receptor status is another impor-tant indicator of breast cancer prognosis. Expression ofestrogen or progesterone receptors positively affects prognosis. The Her2/neu (or c-erb-B2) is an oncogene encoding a membrane-bound growth-factor receptor. Overexpression confers a poor prognosis and is noted in 20% to 30% of invasive ductal cancers.
Breast Cancer Treatment
Breast cancer poses both a local
regional risk (i.e., to the breast and regional lymph nodes) and a systemic
risk. Thesurgical treatment is lumpectomy (breast conservation ther-apy) or mastectomy. Both procedures are aimed at achiev-ing local
control. Mastectomy is removal of all breast tissue and the nipple areolar
complex with preservation of the pectoralis muscles. A modified radical
mastectomy also includes removal of the axillary lymph nodes. Radiation therapy
is used in conjunction with mastectomy for later stages of breast cancer, and
to accompany lumpectomy and partial mastectomy for early stages of breast
cancer. Radiation is an essential component of lumpectomy. This combination
yields outcomes that are equal to those of radical mastectomy.
Breast
reconstruction should be an option for all women who desire it. Reconstruction
can be achieved by several methods,including the insertion of a saline implant
under the pectoral muscle or by using a rectus muscle to replace the lost
tissue. To prepare for a saline implant, a tissue expander is placed beneath
the muscle. Saline is injected into the expander over a period of weeks to
months until the space is large enough to accommodate the implant. Breast
reconstruction can take place immediately after surgery, or it can be delayed
for sev-eral months. Radiation therapy can be given if breast recon-struction
has taken place.
Adjuvant
(systemic) therapy is used in the treatmentof all
stages of breast cancer, regardless of lymph node sta-tus. Adjuvant therapy includes chemotherapeutic drugs that killcancer cells
and hormonal therapies such as tamoxifen that act as
Tamoxifen is used to treat women withestrogen
receptor-positive breast cancer. It can be used in conjunction with
chemotherapy. It is also given as a 5-year course of preventive treatment
following surgery. Aromatase inhibitors (AIs)
prevent the production ofestrogen in postmenopausal women.
AIs are used to extend survival in women with metastatic cancer, as primary adjuvant therapy, and in conjunction with tamoxifen to prevent cancer recurrence.
Another drug used to treat breast
cancer is trastuzumab. It acts on membrane-bound protein produced by Her2/neu. If a patient’s cancer is found to
overexpress the Her2/neu protein, trastuzumab can be given as adjuvant therapy.
Trastuzumabis associated with significant side effects, including heart
failure, respiratory problems, and life-threatening allergic reactions.
Obstetrician-gynecologists are in
the unique position of providing care for women who have been treated for
breast cancer. For some women, the continuation of care years, follow-up
appointments occur every 3 to 6 months and then annually after that. Annual
mammography and physical exams should continue indefinitely. Most breast cancer
recurrence will occur within 5 years of primary therapy.
Related Topics