Chapter: Clinical Anesthesiology: Anesthetic Management: Anesthesia for Patients with Kidney Disease
Anesthesia for Evaluating Renal Function
Evaluating Renal Function
Renal impairment can be due to
glomerular dys-function, tubular dysfunction, or obstruction of the urinary
tract. Because abnormalities of glomerular function cause the greatest
derangements and are most readily detectable, the most useful laboratory tests
utilized currently are those related to assess-ment of glomerular filtration
rate (GFR). Accurate clinical assessment of renal function is often difficult
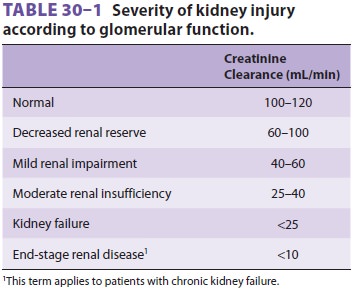
and relies heavily on laboratory determinations such as the creatinine
clearance (Table 30–1).
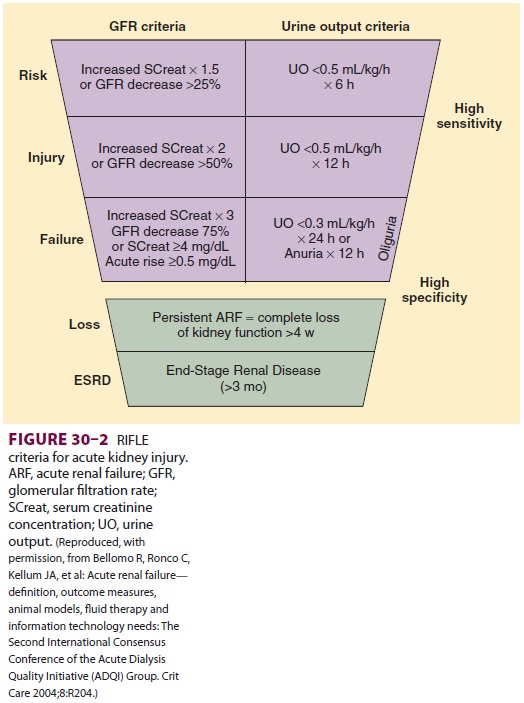
Two sys-tems for classification of AKI are helpful in defining and staging the
degree of renal dysfunction; these are the Acute Dialysis Quality Initiative
RIFLE cri-teria (Figure 30–2) and the Acute Kidney Injury Network
(AKIN) staging system (Table 30–2). A great deal of research is currently
evaluating plasma and urine biomarkers associated with AKI, such as cystatin C,
neutrophil gelatinase–associated lipo-calin, interleukin-18, and kidney injury
molecule-1. It is likely that biomarkers will play a prominent role in the near
future for diagnosis, staging, and prog-nostic assessment of AKI.

BLOOD UREA NITROGEN
The primary source of urea in the body is the liver. During
protein catabolism, ammonia is produced from the deamination of amino acids.
Hepatic con-version of ammonia to urea prevents the buildup of toxic ammonia
levels:
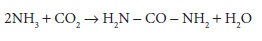
Blood urea nitrogen (BUN) is therefore
directly related to protein catabolism and inversely related to glomerular
filtration. As a result, BUN is not a reli-able indicator of the GFR unless
protein catabolism is normal and constant. Moreover, 40–50% of the urea
filtrate is normally reabsorbed passively by the renal tubules; hypovolemia
increases this fraction.
The normal BUN concentration is 10–20
mg/dL. Lower values can be seen with starvation or liver disease; elevations
usually result from decreases in GFR or increases in protein catabolism. The
latter may be due to a high catabolic state (trauma or sep-sis), degradation of
blood either in the gastrointesti-nal tract or in a large hematoma, or a
high-protein diet. BUN concentrations greater than 50 mg/dL are generally
associated with impairment of renal function.
SERUM CREATININE
Creatine is a product of muscle
metabolism that is nonenzymatically converted to creatinine. Creati-nine
production in most people is relatively constant and related to muscle mass,
averaging 20–25 mg/kg in men and 15–20 mg/kg in women. Creatinine is then
filtered (and to a minor extent secreted) but not reabsorbed in the kidneys.
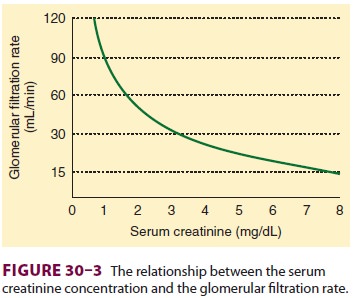
Serum creatinine con-centration is therefore directly related to body mus-cle
mass but inversely related to glomerular filtration (Figure 30–3).
Because body muscle mass is usually relatively constant, serum creatinine
measurements are generally reliable indices of GFR in the healthy patient. However, the utility of a
single serum creatinine measurement as an indicator of GFR is limited in critical illness: the rate of
creati-nine production, and its volume of distribution, may be abnormal in the
critically ill patient, and a single serum creatinine measurement often will
not accu-rately reflect GFR in the physiological disequilib-rium of AKI.
The normal serum creatinine concentration is 0.8–1.3 mg/dL in
men and 0.6–1 mg/dL in women. Note from Figure 30–3 that each doubling of the
serum creatinine represents a 50% reduction in GFR. Large meat meals,
cimetidine therapy, and increases in acetoacetate (as during ketoacido-sis) can
increase serum creatinine measurements without a change in GFR. Meat meals
increase the creatinine load, and high acetoacetate concentra-tions interfere
with the most common laboratory method for measuring creatinine. Cimetidine
appears to inhibit creatinine secretion by the renal tubules.
GFR declines with increasing age in most indi-viduals (5% per
decade after age 20), but because muscle mass also declines, the serum
creatinine remains relatively normal; creatinine production may decrease to 10
mg/kg. Thus, in elderly patients, small increases in serum creatinine may
represent large changes in GFR. Using age and lean body weight (in kilograms),
GFR can be estimated by the following formula for men:
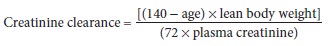
For women, this equation must be multiplied by 0.85 to
compensate for a smaller muscle mass. The serum creatinine concentration
requires 48–72 h to equilibrate at a new level following acute changes in GFR.
CREATININE CLEARANCE
Creatinine clearance measurement is the
most accurate method available for clinically assessing overall renal function
(actually, GFR). Although measurements are usually performed over 24 h, 2-h
creatinine clearance determinations are reasonably accurate and easier to
perform. Mild impairment of renal function generally results in creatinine
clear-ances of 40–60 mL/min. Clearances between 25 and 40 mL/min produce
moderate renal dysfunction and nearly always cause symptoms. Creatinine
clear-ances less than 25 mL/min are indicative of overt kidney failure.
Progressive kidney disease enhances
creati-nine secretion in the proximal tubule. As a result, with declining renal
function the creatinine clear-ance progressively overestimates the true GFR.
Moreover, relative preservation of GFR may occur early in the course of
progressive kidney disease due to compensatory hyperfiltration in the
remain-ing nephrons and increases in glomerular filtra-tion pressure. It is
therefore important to look for other signs of deteriorating renal function
such as hypertension, proteinuria, or other abnormalities in urine sediment.
BLOOD UREA NITROGEN:CREATININE RATIO
Low renal tubular flow rates enhance urea reab-sorption but do
not affect creatinine handling. As a result, the BUN to serum creatinine ratio
increases above 10:1. Decreases in
tubular flow can be caused by decreased renal perfusion or obstruction of the
urinary tract. BUN: creatinine ratios greater than 15:1 are therefore seen in
volume depletion and in edematous disorders associated with decreased tubular
flow (eg, congestive heart failure, cirrho-sis, nephrotic syndrome) as well as
in obstructive uropathies. Increases in protein catabolism can also increase
this ratio.
URINALYSIS
Urinalysis continues to be routinely
performed for evaluating renal function. Although its utility for that purpose
is justifiably questionable, urinaly-sis can be helpful in identifying some
disorders of renal tubular dysfunction as well as some nonrenal disturbances. A
routine urinalysis typically includes pH; specific gravity; detection and
quantification of glucose, protein, and bilirubin content; and micro-scopic
examination of the urinary sediment. Urinary pH is helpful only when arterial
pH is also known. A urinary pH greater than 7.0 in the presence of systemic
acidosis is suggestive of renal tubular aci-dosis . Specific gravity is related
to urinary osmolality; 1.010 usually corresponds to 290 mOsm/kg. A specific
gravity greater than 1.018 after an overnight fast is indicative of adequate
renal concentrating ability. A lower specific gravity in the presence of
hyperosmolality in plasma is con-sistent with diabetes insipidus.Glycosuria is
the result of either a low tubu-lar threshold for glucose (normally 180 mg/dL)
or hyperglycemia. Proteinuria detected by routine uri-nalysis should be
evaluated by means of 24-h urine collection. Urinary protein excretions greater
than 150 mg/d are significant. Elevated levels of bilirubin in the urine are
seen with biliary obstruction.Microscopic analysis of the urinary sedi-ment
detects the presence of red or white blood cells, bacteria, casts, and
crystals. Red cells may be indicative of bleeding due to tumor, stones,
infec-tion, coagulopathy, or trauma. White cells and bacteria are generally
associated with infection. Disease processes at the level of the nephron
pro-duce tubular casts. Crystals may be indicative of abnormalities in oxalic
acid, uric acid, or cystine metabolism.
Related Topics